1 Part 2 DEVICE ASSOCIATED INFECTIONS BIOFILM FAST

1 Part 2
DEVICE ASSOCIATED INFECTIONS

BIOFILM FAST FACTS Biofilm is bacteria that adheres to implanted/indwelling devices ( i. e. ; Endotracheal Tubes, Central Lines, Urinary Catheters, Implants) or damaged tissue forming a slimy layer resulting in infection. Biofilm forms on living and non-living surfaces; where is there is moisture in support of bacterial growth Bacteria in biofilm are highly resistant to antibiotics (1, 000 X more resistant than the same bacteria not growing in a biofilm). Indwelling devices when inserted, may readily acquire biofilms on the inner or outer surfaces. The organisms commonly associated with indwelling devices and biofilms are Staph epidermidis, Enterococcus faecalis, E. coli, Proteus mirabilis, Pseudomonas aeruginosa, Klebsiella pneumoniae, and other gram- negative organisms. The best way to prevent biofilm…remove the device. Take a proactive approach Query the physician Get the device out!
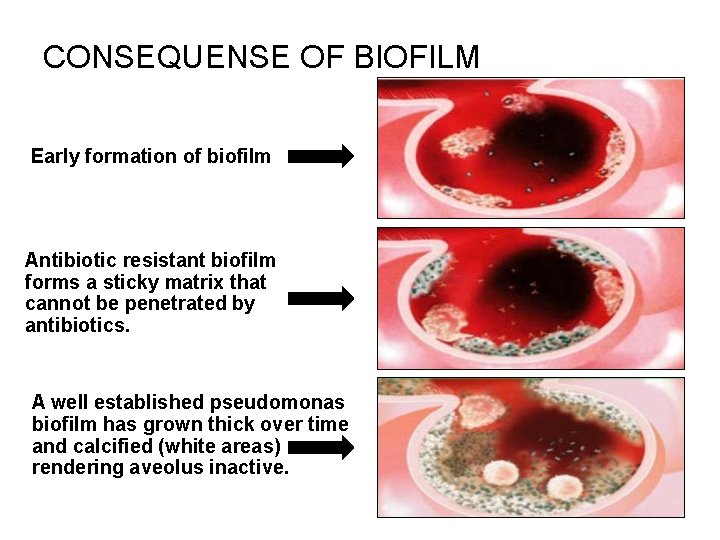
CONSEQUENSE OF BIOFILM Early formation of biofilm Antibiotic resistant biofilm forms a sticky matrix that cannot be penetrated by antibiotics. A well established pseudomonas biofilm has grown thick over time and calcified (white areas) rendering aveolus inactive.
VAP VENTILATOR ASSOCIATED PNEUMONIA

PREVENTION STRATEGIES HOB 30 degrees prevents the pooling of secretions Oral care every 2 hours reduces oral / subglottic bacteria Sedation Vacations in preparation for weaning from the vent Deep Vein Thrombosis prophylaxis to prevent venous stasis Peptic ulcer prophylaxis to decrease risk of stress ulcers
CLABSI CENTRAL LINE ASSOCIATED BLOOD STREAM INFECTION
WHAT IS CONSIDERED A CENTRAL LINE? For the purposes of mandated reporting, the followi ng are considered central lines: • Hemodialysis Catheters • Portacaths • PICC Lines • Umbilical Catheters • Central Venous Catheters

CLABSI PREVENTION STRATEGIES • Aseptic techn ique when changing the dressing • Appropriate disinfection of hubs and ports • Adherence to all elements of Central Line Insertion Practices(CL IP) • Use of isopropyl port protectors to decrease risk of inoculat.
CENTRAL LINE INSERTION PRACTICES (CLIP) Standardizing best practice for insertion techniques leads to better patient outcomes Insertion Practices Proper Hand Hygiene Full barrier precautions including — — — Gown Gloves Mask Eyeshield Cap Full Drape Avoid use of the femoral vein Chlorhexidine-based Antiseptic Prep (betadine is permitted on neonates)

DISINFECTION OF PORTS CUROS caps, ALL ports, ALL the time Cap must be in place for 3 minutes in order to be effective “If the cap is green, the port is clean. ” Excludes pediatric population (risk of foreign body ingestion) Between each infused medication the hub must be scrubbed 10 seconds Discard any left over strands of CUROS prior to disinfection of IV poles and replace with new strands of caps to prevent cross contamination between patients.

CATHETER CHECKLIST “CLIP FORM” Prior to Insertion Provide CLABSI prevention education prior to line insertion. Obtain “Central Line Packet” (with patient name/MRN) from forms fast. Asterisked sections are state required information and as such must be completed. After Insertion Fax Central Line Checklist to Infection Control at 1334. Place checklist in medical record. Ensure all required information is documented to avoid the following: Delays in mandated reporting Timely reporting to stakeholders Citation by the state.
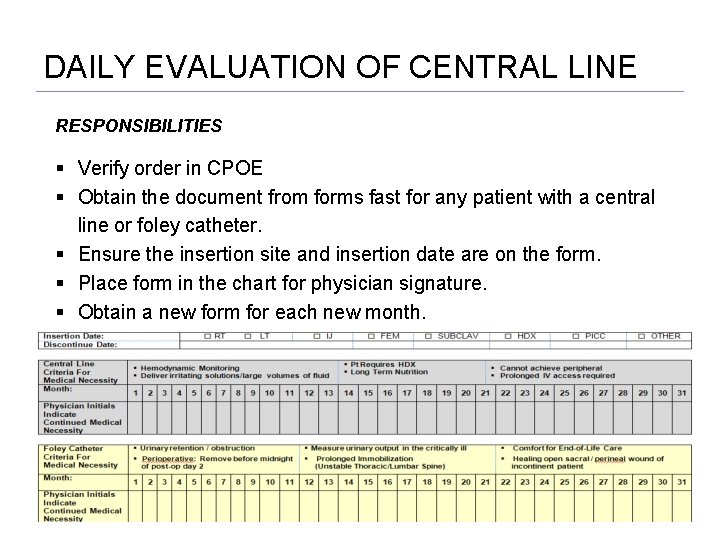
DAILY EVALUATION OF CENTRAL LINE RESPONSIBILITIES Verify order in CPOE Obtain the document from forms fast for any patient with a central line or foley catheter. Ensure the insertion site and insertion date are on the form. Place form in the chart for physician signature. Obtain a new form for each new month.
CAUTI CATHETER ASSOCIATED URINARY TRACT INFECTIONS
INDICATIONS FOR URINARY CATHETERS Urinary Retention Urinary Obstruction Strict I & O Surgeries involving the genitourinary tract Surgical Patients (Discontinue by post-op day 2) Acute genitalia wounds End of life comfort care Stage 3 or 4 pressure ulcers (skin cannot be kept dry otherwise) When is a urinary catheter NOT indicated? To prevent decubiti Incontinence Patient is bedbound Altered mental status

DOCUMENTATION
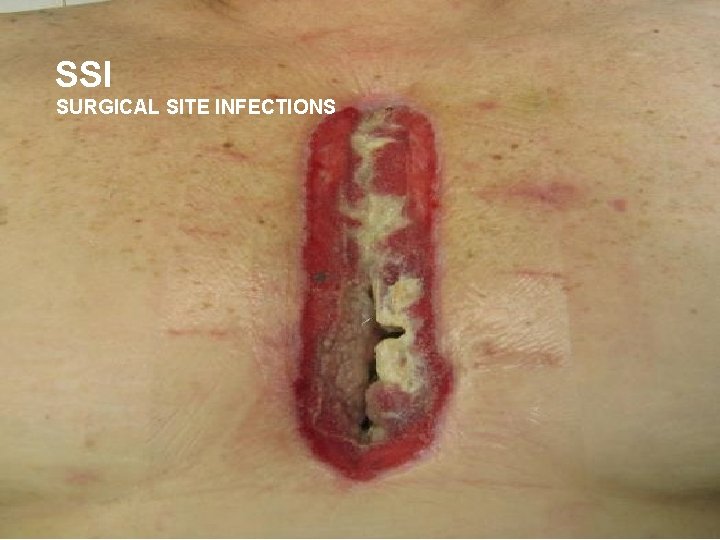
SSI SURGICAL SITE INFECTIONS

O. R. Excellence Surgical Care Improvement Project (SCIP) Centers for Medicare & Medicaid Services (CMS) in collaboration with: American Hospital Association (AHA) Centers for Disease Prevention and Control (CDC) Institute for Healthcare Improvement (IHI) The Joint Commission (TJC) Goal: Reduce the incidence of surgical complications by following a specific set of evidence-based practices aimed at reducing SSIs.

PREVENTION STRATEGIES - SSI Preoperative Bath with Chlorhexidine (CHG) removes surface bacteria from skin Postoperative Normal temperature & glucose promotes the tissue healing Hair removal with clippers decreases the risk of impairing the skin integrity Antibiotics discontinued within 24 hours post op reduces risk of developing a drug resistance Antibiotics are given within 60 minutes of incision The antibiotic is more effective, resulting in better patient outcomes Remove urinary catheters 24 -48 hours post op reduces risk of Catheter Associated Urinary Tract Infection (CAUTI)

PATIENT / FAMILY EDUCATION BEFORE a procedure or the insertion of any device, provide the patient with printed education from KRAMES: CLABSI – NPSG>Central line associated infections print “Central Line Infections” VAP-NPSG print “Healthcare Associated Pneumonia” SSI print “Preventing Surgical Site Infections” CAUTI – Search CAUTI DOCUMENT ALL EDUCATION

REFERENCES Carolyn V. Gould, MD, MSCR; Craig A. Umscheid, MD, MSCE; Rajender K. Agarwal, MD, MPH; Gretchen Kuntz, MSW, MSLIS; David A. Pegues, MD and the Healthcare Infection Control Practices Advisory Committee (HICPAC) (2009), Guideline for Prevention of Catheter Associated Urinary Tract Infections. Atlanta GA. Association for Professionals in Infection Control and Epidemiology (2009), Guideline to the Elimination of Ventilator Associated Pneumonia, APIC Washington DC. Society for Healthcare Epidemiology of America (SHEA) / Infectious Diseases Society America (IDSA) (2008), Strategies to prevent Ventilator-Associated Pneumonia in Acute Care Hospitals, Infection Control and Hospital Epidemiology, vol. 29. supplement 1. Centers for Disease Control and prevention /Healthcare Infection Control Practices Advisory Committee (2003), Guideline for Prevention of Healthcare Associated Pneumonia. Retrieved from: http: //www. cdc. gov/mmwr/preview/mmwrhtml/rr 5303 a 1. htm Guidelines for the Prevention of Intravascular Catheter-Related Infections, Centers for Disease Control and Prevention 2011. http: //www. cdc. gov/hicpac/pdf/guidelines/bsi-guidelines-2011. pdf
- Slides: 21