1 of 8 Boardworks Ltd 2007 Patterns metals
1 of 8 © Boardworks Ltd 2007

Patterns: metals and non-metals Where are these different types of elements grouped together in the periodic table? l metals on the left and centre l non-metals on the right (except hydrogen) l semi-metals between metals and non-metals Semi-metals have some properties similar to metals and other properties similar to non-metals. Can you name a semi-metal element? 2 of 8 © Boardworks Ltd 2007

Patterns: physical state Where are these elements of different states grouped together in the periodic table? l solids on the left, in the centre and on the right l liquids in the middle and on the right l gases on the far right (except hydrogen) Only two elements are liquids at room temperature. What are they? l bromine and mercury 3 of 8 © Boardworks Ltd 2007

Metals to non-metals, solids to gases 4 of 8 © Boardworks Ltd 2007

Patterns: reactivity of metals increase in reactivity What happens to the reactivity of metals along a period? What happens to the reactivity of metals down a group? Which is the most reactive metal? Li Be Na Mg Al K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po Fr Ra Ac Rf Db Sg Bh Hs Mt Ds Rg increase in reactivity 5 of 8 © Boardworks Ltd 2007

Which metal is more reactive? 6 of 8 © Boardworks Ltd 2007
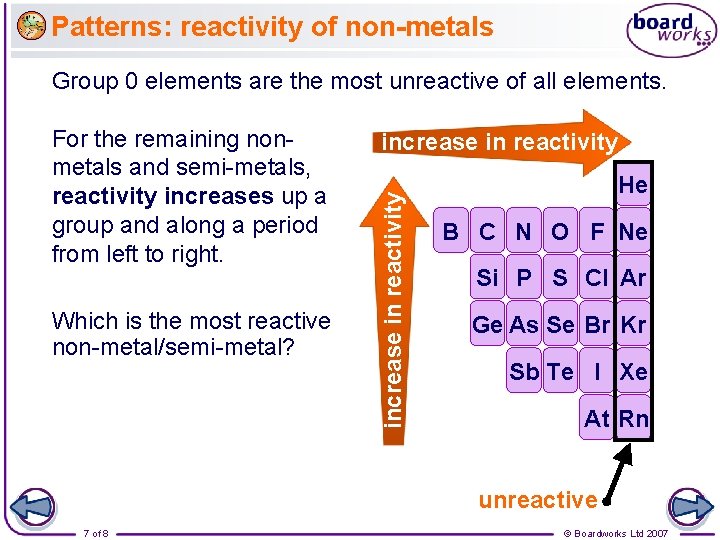
Patterns: reactivity of non-metals Group 0 elements are the most unreactive of all elements. Which is the most reactive non-metal/semi-metal? increase in reactivity For the remaining nonmetals and semi-metals, reactivity increases up a group and along a period from left to right. He B C N O F Ne Si P S Cl Ar Ge As Se Br Kr Sb Te I Xe At Rn unreactive 7 of 8 © Boardworks Ltd 2007
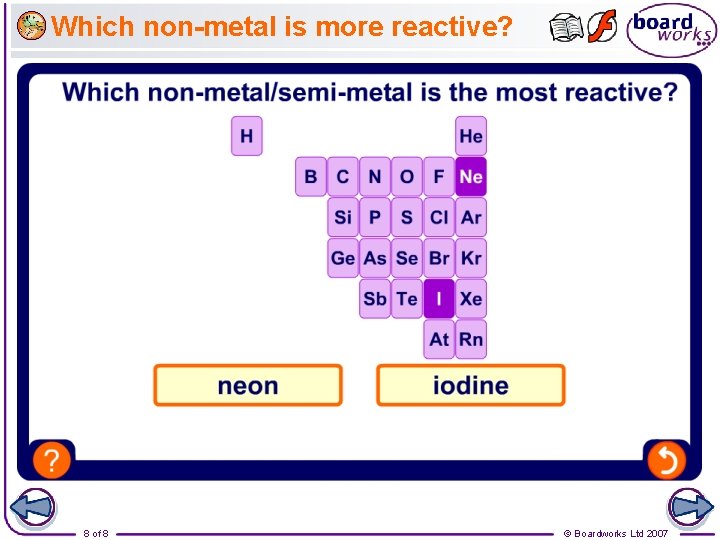
Which non-metal is more reactive? 8 of 8 © Boardworks Ltd 2007
- Slides: 8