1 of 5 Boardworks Ltd 2007 What are
1 of 5 © Boardworks Ltd 2007

What are bonds? Elements are the simplest substances that exist on Earth. Each element is made up of just one type of atom, usually joined to other atoms of the same element by bonds. This forms molecules such as chlorine (Cl 2). Compounds are formed when different elements chemically react and form bonds with each other. Some compounds, like water (H 20), have small, simple structures with just a few atoms bonded together. Others compounds, like DNA, have large, complex structures containing thousands or even millions of bonded atoms. 2 of 5 © Boardworks Ltd 2007
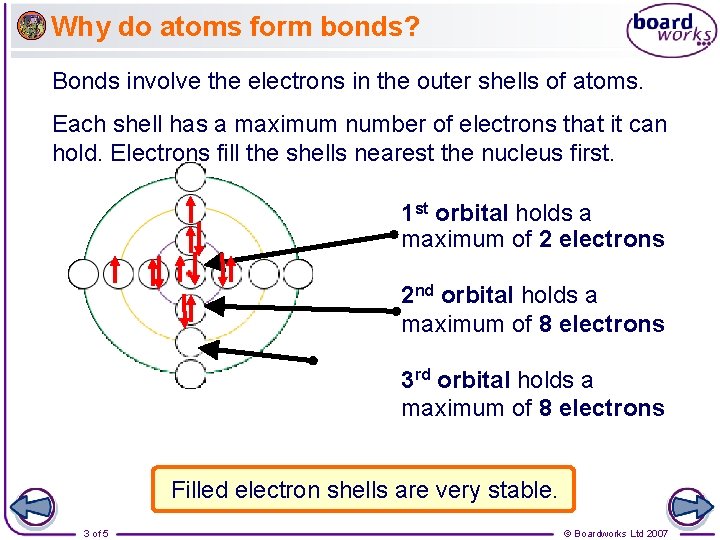
Why do atoms form bonds? Bonds involve the electrons in the outer shells of atoms. Each shell has a maximum number of electrons that it can hold. Electrons fill the shells nearest the nucleus first. 1 st orbital holds a maximum of 2 electrons 2 nd orbital holds a maximum of 8 electrons 3 rd orbital holds a maximum of 8 electrons Filled electron shells are very stable. 3 of 5 © Boardworks Ltd 2007

Why do atoms form bonds? The atoms of noble gases have completely full outer orbitals (or shells) and so are stable. This makes the noble gases very unreactive and so they do not usually form bonds. The atoms of other elements have incomplete outer electron orbitals (or shells) and so are unstable. By forming bonds, the atoms of these elements are able to have filled outer orbitals (or shells) and become stable. 4 of 5 © Boardworks Ltd 2007

What are the types of bonding? Different types of bonds are formed depending on the types of atoms involved: l ionic bonding – occurs between metal and non-metal atoms. l covalent bonding – occurs between non-metals atoms only. l metallic bonding – occurs between metal atoms only. All bonds involve electrons and all bonding involve changes to the number of electrons in the outer shells of atoms. How do you think electrons are involved in ionic bonding? 5 of 5 © Boardworks Ltd 2007

How do atoms form ions? An ion is an atom or group of atoms that has an electrical charge, either positive or negative. Atoms have an equal number of protons and electrons and so do not have an overall charge. Atoms with incomplete outer electron orbitals (shells) are unstable. By either gaining or losing electrons, atoms can obtain full outer electron shells and become stable. When this happens, atoms have an unequal number of protons and electrons and so have an overall charge. This is how atoms become ions. How does an atom become a positive or negative ion? 6 of 5 © Boardworks Ltd 2007

Positive and negative ions? An atom that loses electrons has more protons than electrons and so has a positive overall charge. This is called a positive ion. An atom that gains electrons has more electrons than protons and so has a negative overall charge. This is called a negative ion. The electron configuration of an atom shows how many electrons it must lose or gain to have a filled outer orbital/shell. l Atoms with a nearly empty outer shell, will lose electrons to obtain a full outer orbital/shell. l Atoms with a nearly full outer shell, will gain electrons to obtain a full outer orbital/shell. 7 of 5 © Boardworks Ltd 2007

How do atoms form positive ions? An atom that loses one or more electrons forms a positive ion. Metal atoms, such as sodium, magnesium and iron, form positive ions. Positive ions have a small ‘+’ symbol and a number by this to indicate how many electrons have been lost. This number is usually the same as the number of electrons in the atom’s outer shell. For example: lithium atom 2. 1 lithium ion [ 2 ] = Li+ magnesium atom 2. 8. 2 magnesium ion [ 2. 8 ] = Mg 2+ aluminium atom 2. 8. 3 aluminium ion [ 2. 8 ] = Al 3+ 8 of 5 © Boardworks Ltd 2007
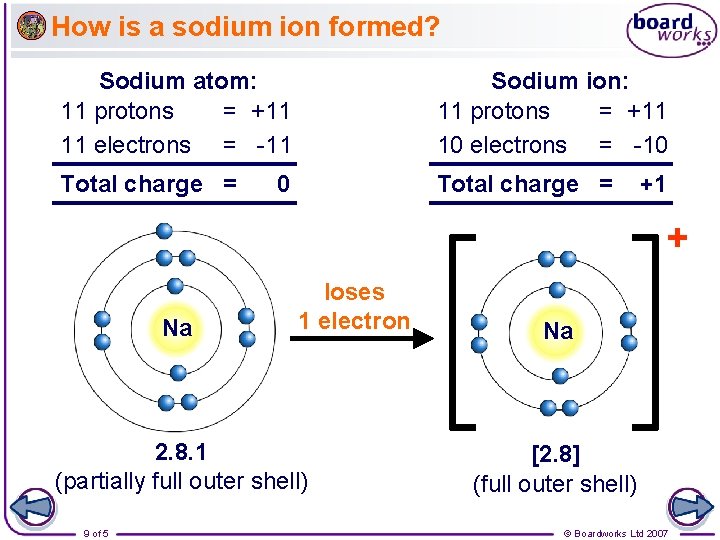
How is a sodium ion formed? Sodium atom: 11 protons = +11 11 electrons = -11 Sodium ion: 11 protons = +11 10 electrons = -10 Total charge = 0 +1 + Na loses 1 electron 2. 8. 1 (partially full outer shell) 9 of 5 Na [2. 8] (full outer shell) © Boardworks Ltd 2007
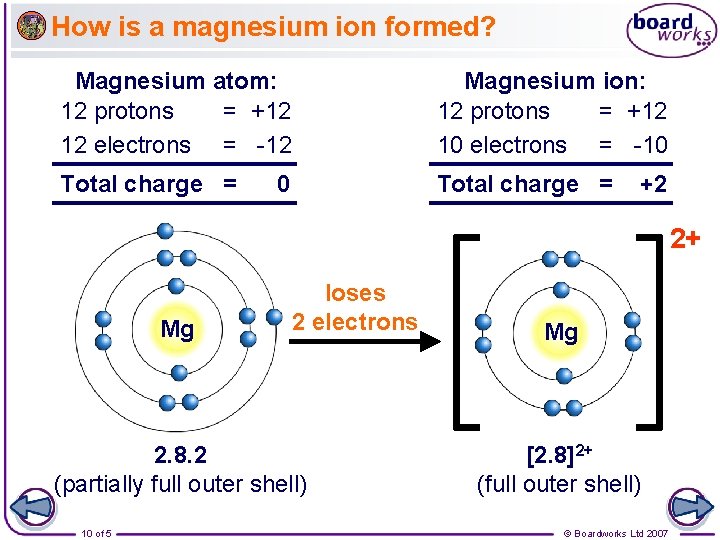
How is a magnesium ion formed? Magnesium atom: 12 protons = +12 12 electrons = -12 Magnesium ion: 12 protons = +12 10 electrons = -10 Total charge = 0 +2 2+ Mg loses 2 electrons 2. 8. 2 (partially full outer shell) 10 of 5 Mg [2. 8]2+ (full outer shell) © Boardworks Ltd 2007

How do atoms form negative ions? An atom that gains one or more electrons forms a negative ion. Non-metal atoms, such as chlorine, oxygen and nitrogen, form negative ions. Negative ions have a small ‘-’ symbol and a number by this to indicate how many electrons have been gained to fill their outer shell. For example: chlorine atom 2. 8. 7 chloride ion [ 2. 8. 8 ] = Cl- oxygen atom 2. 6 oxide ion nitrogen atom 2. 5 nitride ion [ 2, 8 ] = O 2= N 3 - The name of the ion is slightly different to the atom’s name. 11 of 5 © Boardworks Ltd 2007
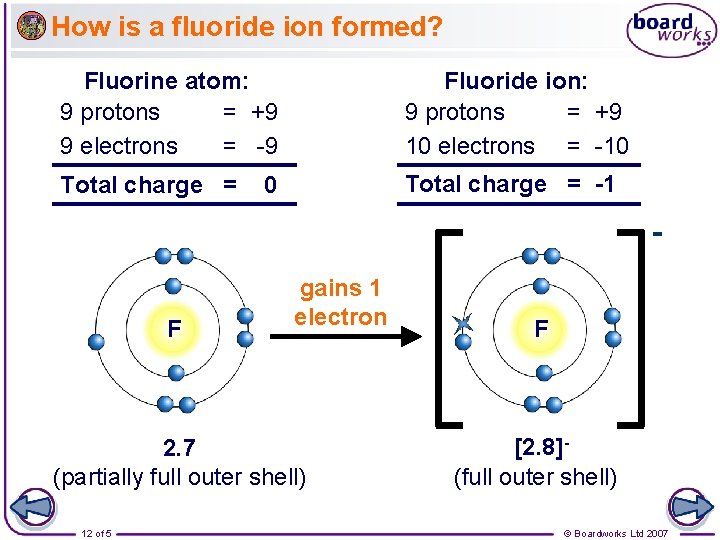
How is a fluoride ion formed? Fluorine atom: 9 protons = +9 9 electrons = -9 Fluoride ion: 9 protons = +9 10 electrons = -10 Total charge = -1 0 F gains 1 electron 2. 7 (partially full outer shell) 12 of 5 F [2. 8](full outer shell) © Boardworks Ltd 2007
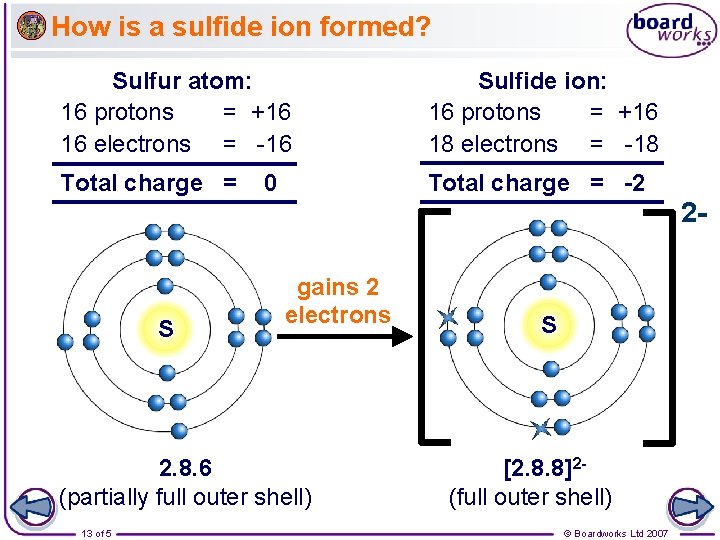
How is a sulfide ion formed? Sulfur atom: 16 protons = +16 16 electrons = -16 Sulfide ion: 16 protons = +16 18 electrons = -18 Total charge = -2 0 2 - S gains 2 electrons 2. 8. 6 (partially full outer shell) 13 of 5 S [2. 8. 8]2(full outer shell) © Boardworks Ltd 2007

Activity • Complete worksheek 9 14 of 5 © Boardworks Ltd 2007

Electron configuration of ions When different elements gain or lose electrons to complete their outer electron shell, they can create ions that have the same electron configuration. For example, oxygen and fluorine both gain electrons to become negative ions. The resulting ions have the same electron configuration: - 2 O F The number of protons and neutrons however, remain different for each element. This means that each ion has different properties. 15 of 5 © Boardworks Ltd 2007

What is ionic bonding? Compounds that contain ions are called ionic compounds. These compounds are usually formed by a reaction between a metal and a non-metal. Why do these substances react together and form bonds? The metal and non-metal atoms have incomplete outer electron shells and so are unstable. Electrons are transferred from each metal atom to each non-metal atom. The metal and the non-metal atoms form ions with completely full outer orbitals/ shells and become stable. The positive and negative ions are strongly attracted to each other. This electrostatic attraction is called ionic bonding. 16 of 5 © Boardworks Ltd 2007
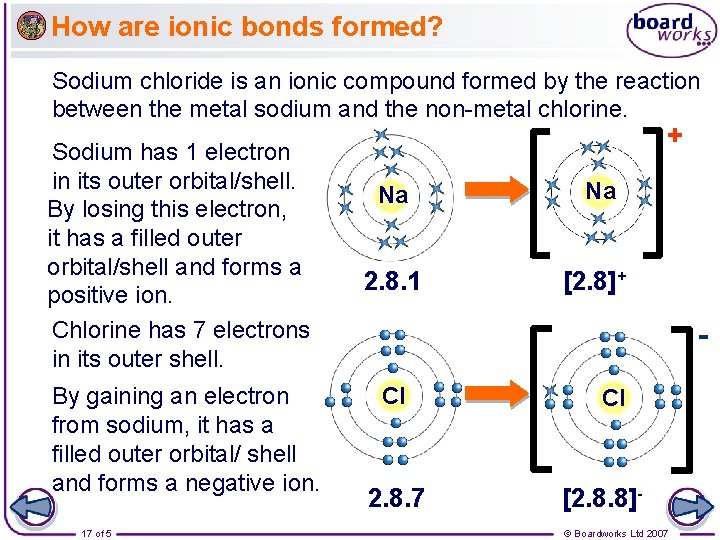
How are ionic bonds formed? Sodium chloride is an ionic compound formed by the reaction between the metal sodium and the non-metal chlorine. Sodium has 1 electron in its outer orbital/shell. By losing this electron, it has a filled outer orbital/shell and forms a positive ion. Chlorine has 7 electrons in its outer shell. By gaining an electron from sodium, it has a filled outer orbital/ shell and forms a negative ion. 17 of 5 + Na Na 2. 8. 1 [2. 8]+ Cl 2. 8. 7 Cl [2. 8. 8]© Boardworks Ltd 2007
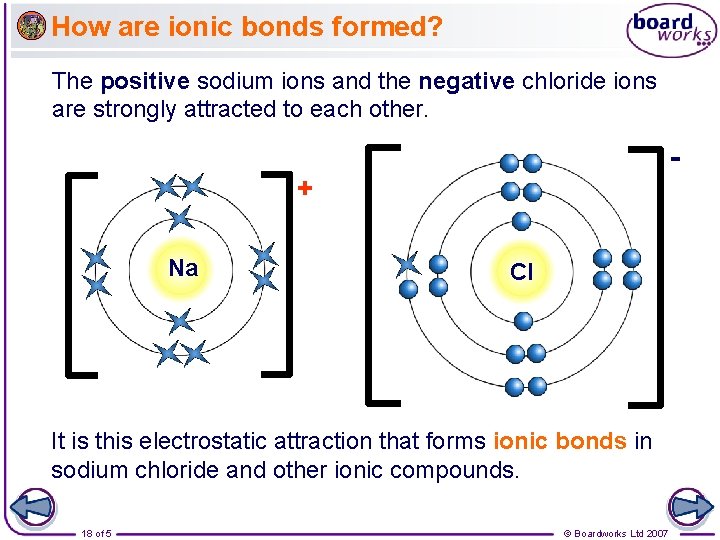
How are ionic bonds formed? The positive sodium ions and the negative chloride ions are strongly attracted to each other. + Na Cl It is this electrostatic attraction that forms ionic bonds in sodium chloride and other ionic compounds. 18 of 5 © Boardworks Ltd 2007
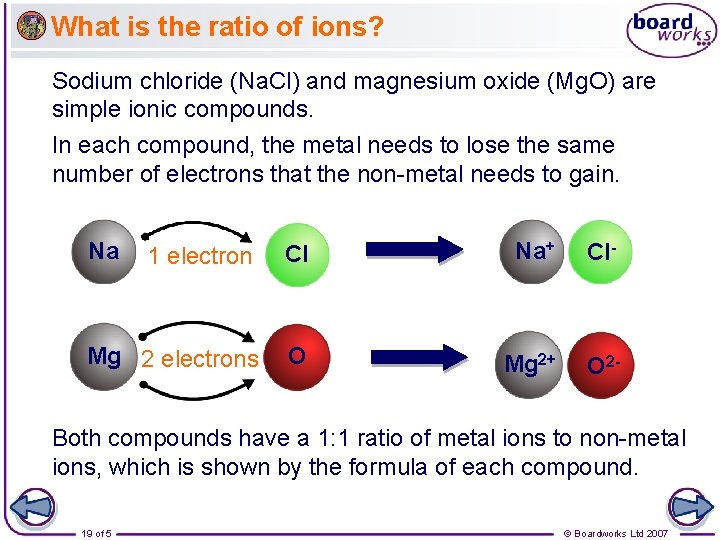
What is the ratio of ions? Sodium chloride (Na. Cl) and magnesium oxide (Mg. O) are simple ionic compounds. In each compound, the metal needs to lose the same number of electrons that the non-metal needs to gain. Na 1 electron Cl Na+ Cl- Mg 2 electrons O Mg 2+ O 2 - Both compounds have a 1: 1 ratio of metal ions to non-metal ions, which is shown by the formula of each compound. 19 of 5 © Boardworks Ltd 2007
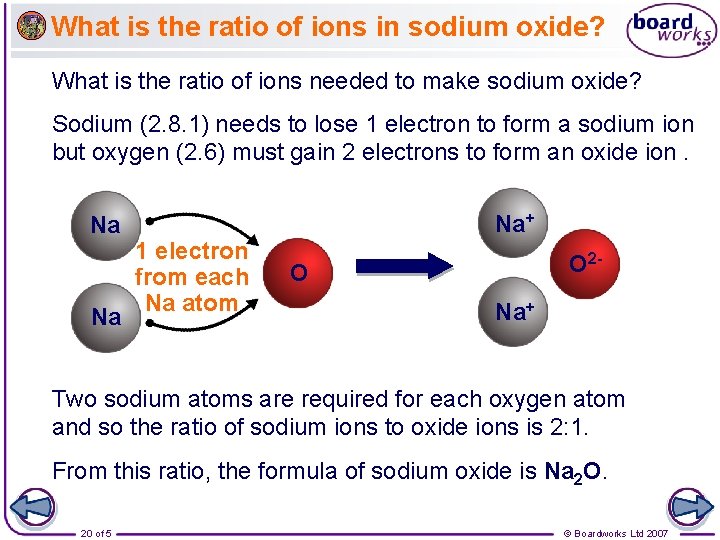
What is the ratio of ions in sodium oxide? What is the ratio of ions needed to make sodium oxide? Sodium (2. 8. 1) needs to lose 1 electron to form a sodium ion but oxygen (2. 6) must gain 2 electrons to form an oxide ion. Na Na Na+ 1 electron from each Na atom O 2 - O Na+ Two sodium atoms are required for each oxygen atom and so the ratio of sodium ions to oxide ions is 2: 1. From this ratio, the formula of sodium oxide is Na 2 O. 20 of 5 © Boardworks Ltd 2007
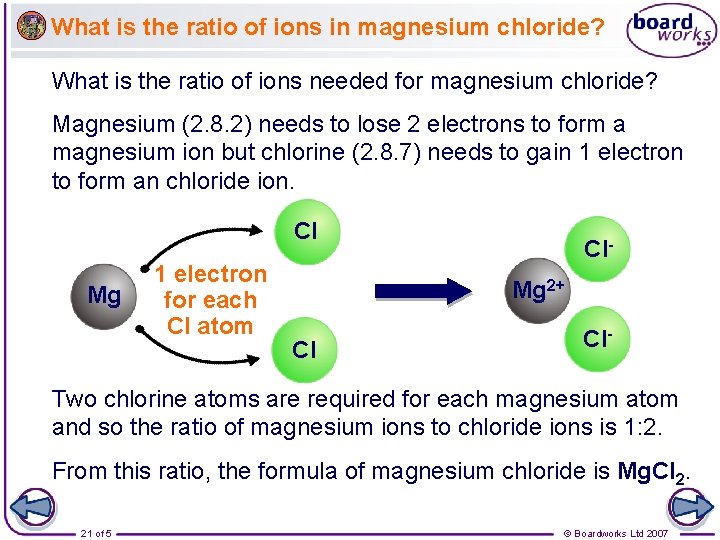
What is the ratio of ions in magnesium chloride? What is the ratio of ions needed for magnesium chloride? Magnesium (2. 8. 2) needs to lose 2 electrons to form a magnesium ion but chlorine (2. 8. 7) needs to gain 1 electron to form an chloride ion. Cl Mg 1 electron for each Cl atom Cl. Mg 2+ Cl Cl- Two chlorine atoms are required for each magnesium atom and so the ratio of magnesium ions to chloride ions is 1: 2. From this ratio, the formula of magnesium chloride is Mg. Cl 2. 21 of 5 © Boardworks Ltd 2007

How to write the formula of an ionic compound The formula of a compound uses chemical symbols and numbers to show the ratio of atoms of each element present. To work out the formula of an ionic compound: 1. Write down the symbol for each element – the metal is always written first. 2. Calculate the charge for each type of ion. 3. Balance the number of ions so that the positive and negative charges are balanced and equal zero. This gives the ratio of ions. 4. Use the ratio to write down the formula of the ionic compound. 22 of 5 © Boardworks Ltd 2007
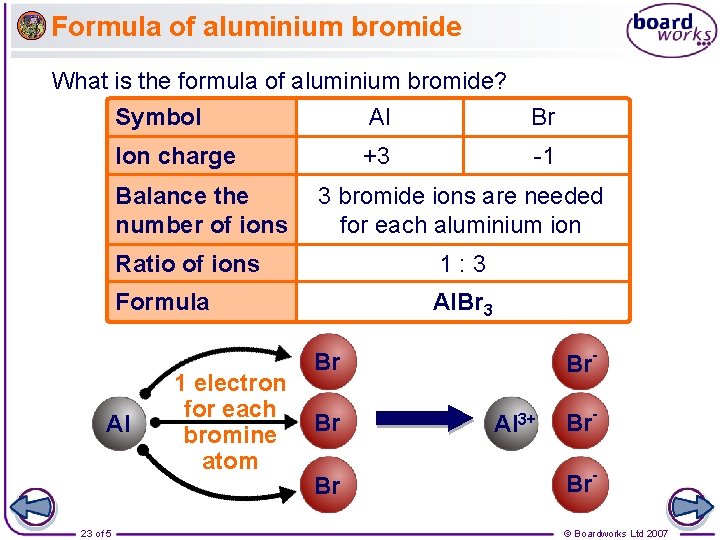
Formula of aluminium bromide What is the formula of aluminium bromide? Symbol Al Br Ion charge +3 -1 Balance the number of ions Ratio of ions 1: 3 Formula Al. Br 3 Al 23 of 5 3 bromide ions are needed for each aluminium ion 1 electron for each bromine atom Br- Br Br Br Al 3+ Br. Br© Boardworks Ltd 2007
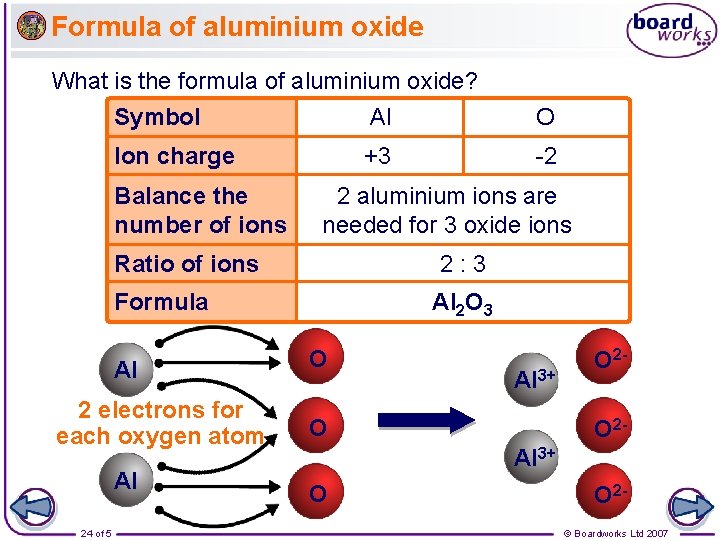
Formula of aluminium oxide What is the formula of aluminium oxide? Symbol Al O Ion charge +3 -2 Balance the number of ions 2 aluminium ions are needed for 3 oxide ions Ratio of ions 2: 3 Formula Al 2 electrons for each oxygen atom Al 24 of 5 Al 2 O 3 O Al 3+ O O 2 O 2 - Al 3+ O O 2© Boardworks Ltd 2007
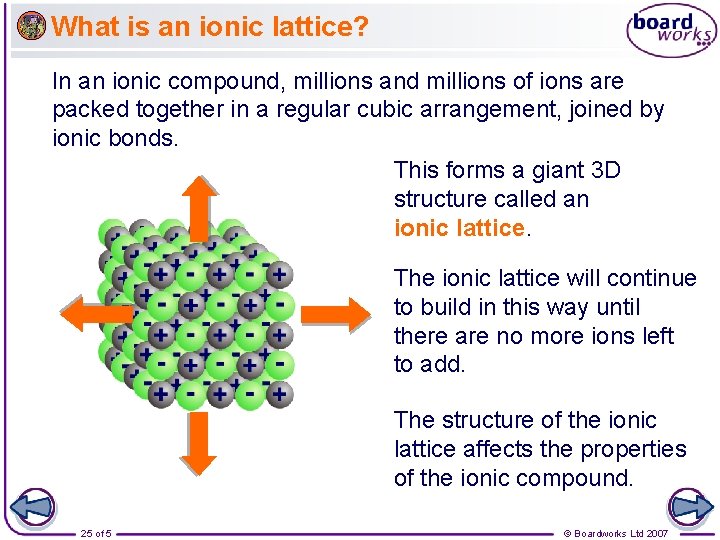
What is an ionic lattice? In an ionic compound, millions and millions of ions are packed together in a regular cubic arrangement, joined by ionic bonds. This forms a giant 3 D structure called an ionic lattice. The ionic lattice will continue to build in this way until there are no more ions left to add. The structure of the ionic lattice affects the properties of the ionic compound. 25 of 5 © Boardworks Ltd 2007

Why do ionic compounds form crystals? Ionic compounds such as sodium chloride, form crystals, with a cubic shape. This is due to the structure of the ionic lattice. All ionic compounds form lattices and crystals when solid. 26 of 5 © Boardworks Ltd 2007

Heating ionic compounds Why are ionic compounds solid at room temperature and have high melting points and boiling points? Compound Ion charges Melting point (o. C) sodium chloride 1+ and 1 - 801 1, 413 magnesium oxide 2+ and 2 - 2, 852 3, 600 Ionic bonds are strong and a lot of heat is needed to break them. Boiling point (o. C) strong ionic bonds hold ions together Larger ionic charges produce stronger ionic bonds and so much more heat is required to break the ionic bonds in magnesium oxide than in sodium chloride. 27 of 5 © Boardworks Ltd 2007
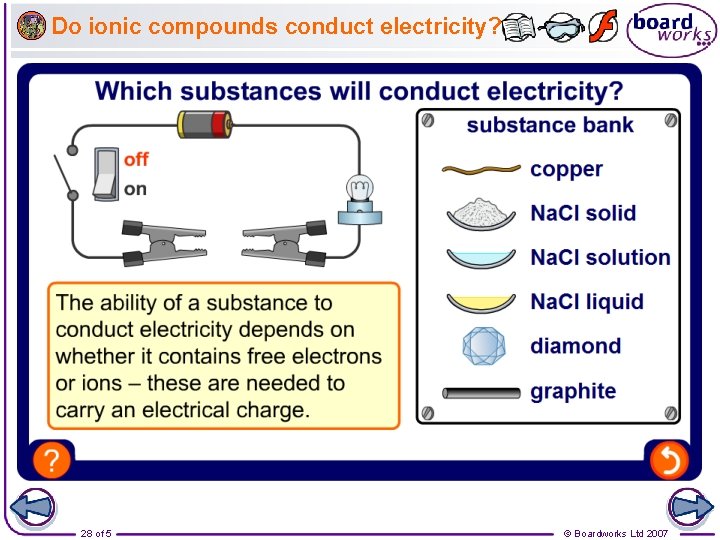
Do ionic compounds conduct electricity? 28 of 5 © Boardworks Ltd 2007

How can ionic compounds conduct electricity? As solids, ionic compounds cannot conduct electricity because their ions are bonded together in the lattice. When liquid (molten), the ions can break free of the lattice and are able to move. The ions are charged particles and so can carry an electric current. ions in solid state cannot move ions in molten state can move and conduct electricity Ionic compounds are usually soluble in water because water molecules have a slight electrical charge and so can attract the ions away from the lattice. When dissolved, the ions are free to move and can carry an electric current. 29 of 5 © Boardworks Ltd 2007
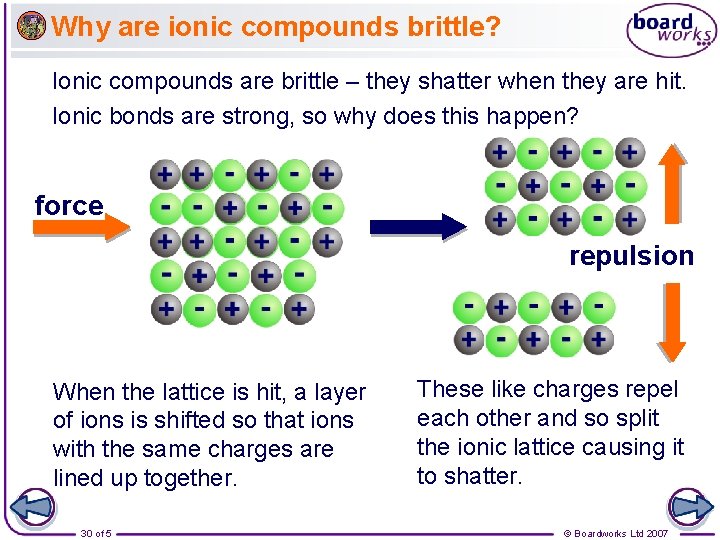
Why are ionic compounds brittle? Ionic compounds are brittle – they shatter when they are hit. Ionic bonds are strong, so why does this happen? force repulsion When the lattice is hit, a layer of ions is shifted so that ions with the same charges are lined up together. 30 of 5 These like charges repel each other and so split the ionic lattice causing it to shatter. © Boardworks Ltd 2007

Activity • Complete worksheet 8 31 of 5 © Boardworks Ltd 2007
- Slides: 31