1 of 47 Boardworks Ltd 2008 2 of
1 of 47 © Boardworks Ltd 2008

2 of 47 © Boardworks Ltd 2008

Acids and alkalis How many examples of acids and alkalis can you spot? 3 of 47 © Boardworks Ltd 2008

Acids: true or false? 4 of 47 © Boardworks Ltd 2008

Alkalis: true or false? 5 of 47 © Boardworks Ltd 2008
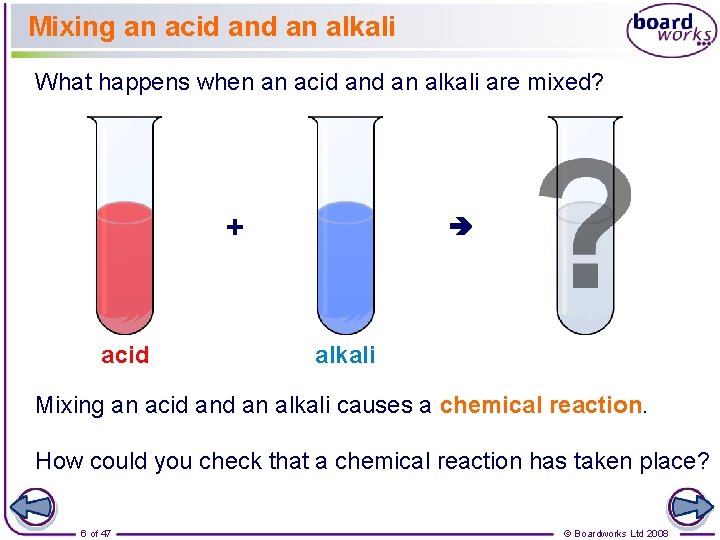
Mixing an acid an alkali What happens when an acid an alkali are mixed? + acid alkali Mixing an acid an alkali causes a chemical reaction. How could you check that a chemical reaction has taken place? 6 of 47 © Boardworks Ltd 2008

Mixing acids and alkalis 7 of 47 © Boardworks Ltd 2008

What is neutralization? The chemical reaction between an acid an alkali is called neutralization. What is the p. H value of the mixture of salt and water? + acid alkali salt + water The mixture of salt and water is neutral, so its p. H is 7. The chemical reaction between an acid an alkali can be written as: acid 8 of 47 + alkali salt + water © Boardworks Ltd 2008

Neutralizing stings Bee stings are acidic. Which safe household substances could you use to treat a bee sting? 9 of 47 Wasp stings are alkaline. Which safe household substances could you use to treat a wasp sting? © Boardworks Ltd 2008

Separating a salt 10 of 47 © Boardworks Ltd 2008

Making salts The salt produced by a neutralization reaction depends on the acid and the alkali used. Sodium chloride is produced by the reaction between hydrochloric acid and sodium hydroxide. hydrochloric acid + sodium hydroxide chloride + water Which acid and which alkali would be needed to make the salt potassium chloride? hydrochloric acid 11 of 47 + potassium hydroxide potassium chloride + water © Boardworks Ltd 2008

Naming salts l When an alkali reacts with hydrochloric acid, the salt produced is a chloride. l When an alkali reacts with sulfuric acid, the salt produced is a sulfate. l When an alkali reacts with nitric acid, the salt produced is a nitrate. Which acid would be needed to make these salts? 1. copper nitrate 2. magnesium chloride 3. sodium sulfate 12 of 47 © Boardworks Ltd 2008

Making salts: word equations Complete these word equations: hydrochloric acid sulfuric acid nitric acid 13 of 47 + potassium hydroxide potassium chloride + water + sodium hydroxide sodium sulfate + water + potassium hydroxide potassium nitrate + water © Boardworks Ltd 2008
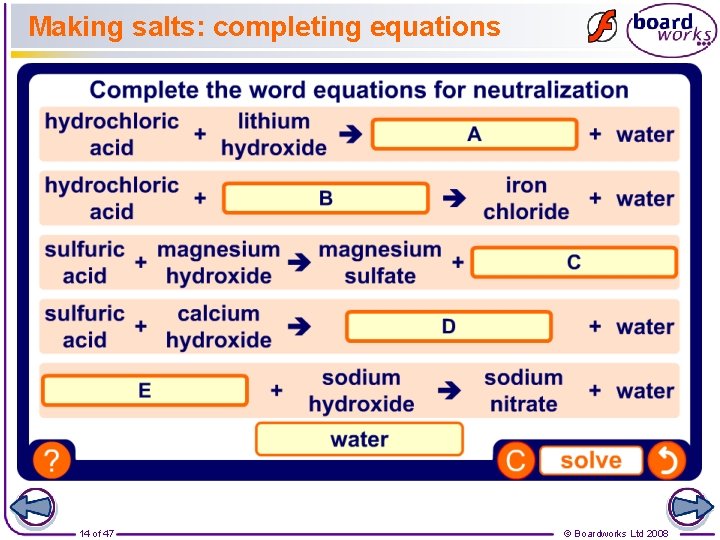
Making salts: completing equations 14 of 47 © Boardworks Ltd 2008
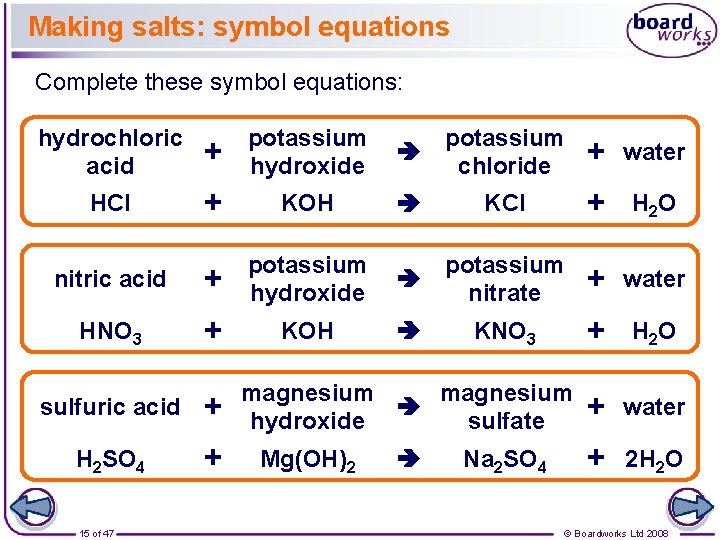
Making salts: symbol equations Complete these symbol equations: hydrochloric acid + potassium hydroxide potassium chloride + water HCl + KOH KCl + H 2 O nitric acid + potassium hydroxide potassium nitrate + water HNO 3 + KOH KNO 3 + H 2 O + water + 2 H 2 O sulfuric acid + H 2 SO 4 + 15 of 47 magnesium hydroxide sulfate Mg(OH)2 Na 2 SO 4 © Boardworks Ltd 2008

Balancing equations When a chemical reaction occurs, it can be described by a a symbol equation. The balanced symbol equation for a chemical reaction shows the ratio of reactants and products involved. For example: sulfuric acid H 2 SO 4 + sodium hydroxide sodium sulfate + water + 2 Na. OH Na 2 SO 4 + 2 H 2 O An ‘unbalanced’ equation can be balanced by multiplying the different atoms and molecules on each side by different amounts, so that they match. 16 of 47 © Boardworks Ltd 2008

Balancing equations If you count the number of atoms of each type on each side of this equation, you will see that they do not match. H 2 SO 4 + Na. OH Na 2 SO 4 + H 2 O So, multiply the sodium atoms on the left by 2: H 2 SO 4 + 2 Na. OH Na 2 SO 4 Now that the number of sodium atoms on each side is equal the number of hydrogen atoms on the left side has increased. So multiply the number of hydrogen atoms on the right by 2. H 2 SO 4 17 of 47 + 2 Na. OH Na 2 SO 4 + 2 H 2 O © Boardworks Ltd 2008
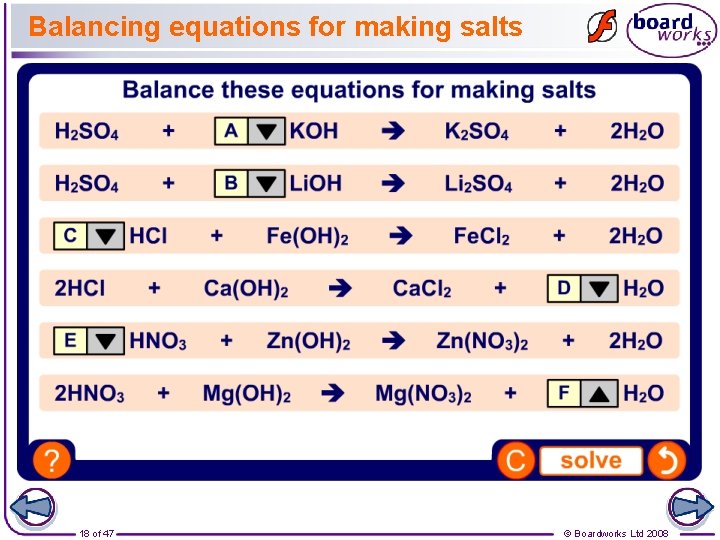
Balancing equations for making salts 18 of 47 © Boardworks Ltd 2008

Indigestion remedies The acid in your stomach that helps you break down your food is called hydrochloric acid. It has a p. H of between 1 and 2. Indigestion and heartburn are caused by an excess of acid in your stomach. Indigestion remedies such as Milk of Magnesia contain antacids, like magnesium oxide and calcium carbonate. What do you think antacids do to the acid in your stomach? 19 of 47 © Boardworks Ltd 2008

20 of 47 © Boardworks Ltd 2008

Metal oxides are compounds made from a metal and oxygen. For example, burning magnesium in oxygen produces magnesium oxide. magnesium + oxygen magnesium oxide Acids react with metal oxides to make a salt and water. For example, sulfuric acid reacts with copper oxide to make copper sulfate and water. sulfuric acid 21 of 47 + copper oxide copper sulfate + water © Boardworks Ltd 2008

Base or alkali? Bases are substances that react with acids, which include metals oxides, hydroxides and carbonates. Alkalis are substances that react with acids and which are soluble in water. bases: react with acids alkalis: soluble in water Copper oxide is a base, but not an alkali, because it reacts with acids, but does not dissolve in water. Sodium hydroxide is an alkali (and a base), because it reacts with acids and it is soluble in water. 22 of 47 © Boardworks Ltd 2008

Acids and metal oxides 23 of 47 © Boardworks Ltd 2008

Making copper sulfate 24 of 47 © Boardworks Ltd 2008
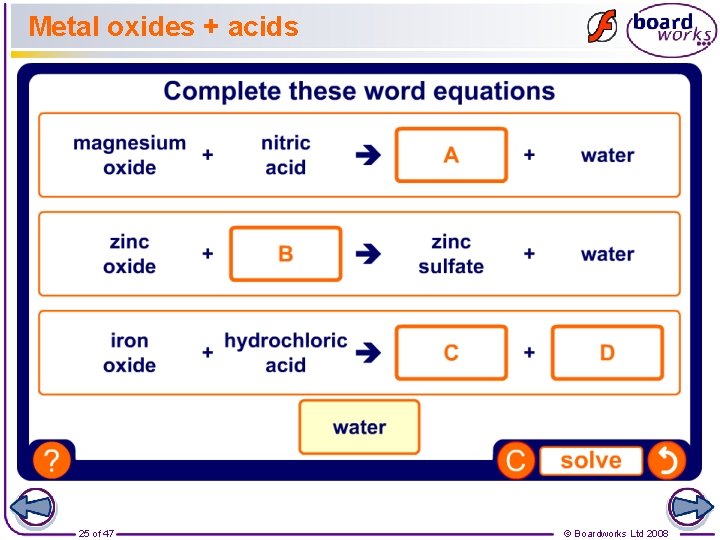
Metal oxides + acids 25 of 47 © Boardworks Ltd 2008

What is the formula? 26 of 47 © Boardworks Ltd 2008

Formulae How do we know that the formula of sodium chloride is Na. Cl, while the formula of copper chloride is Cu. Cl 2? It is because of the combining power of the atoms. l Sodium has a combining power of 1, so it bonds with one other atom. l Chlorine also has a combining power of 1, so it bonds with one other atom. l Copper, though, has a combining power of 2, so it needs to bond to two chlorine atoms. The formula of magnesium chloride is Mg. Cl 2, so what do you think is the combining power of magnesium? 27 of 47 © Boardworks Ltd 2008
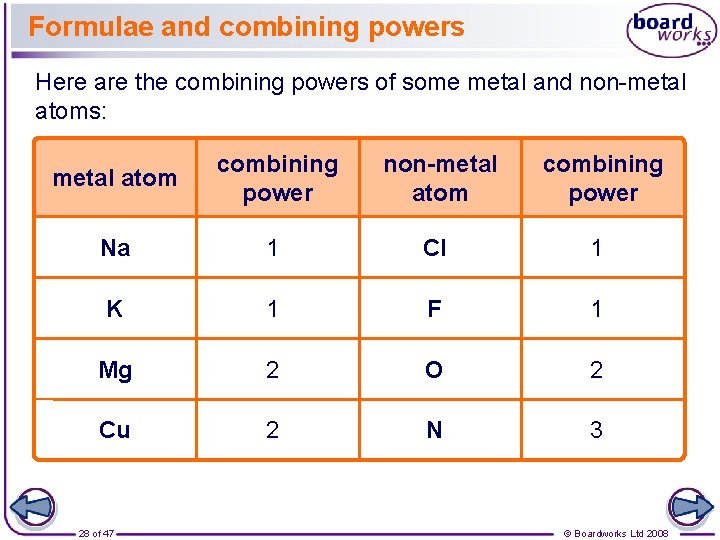
Formulae and combining powers Here are the combining powers of some metal and non-metal atoms: metal atom combining power non-metal atom combining power Na 1 Cl 1 K 1 F 1 Mg 2 O 2 Cu 2 N 3 28 of 47 © Boardworks Ltd 2008

Formulae and combining powers 29 of 47 © Boardworks Ltd 2008

30 of 47 © Boardworks Ltd 2008
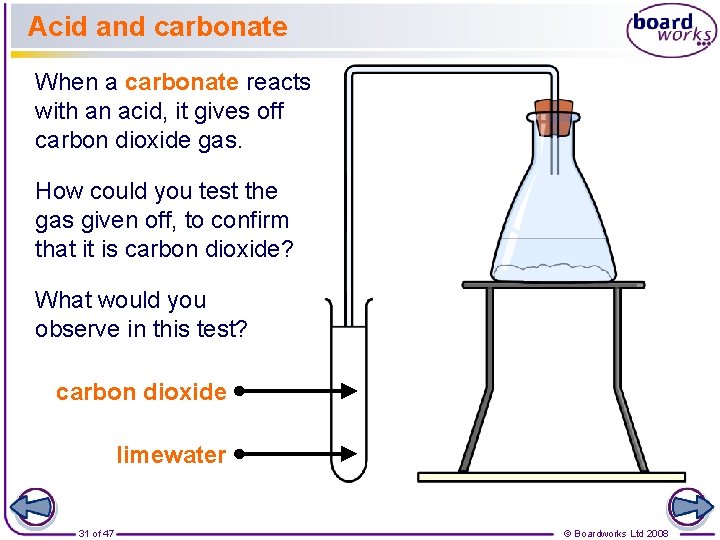
Acid and carbonate When a carbonate reacts with an acid, it gives off carbon dioxide gas. How could you test the gas given off, to confirm that it is carbon dioxide? What would you observe in this test? carbon dioxide limewater 31 of 47 © Boardworks Ltd 2008

Acid and carbonate The products of the reaction between a carbonate and an acid are a salt, carbon dioxide and water. Calcium carbonate is a substance that is naturally found in the shells of sea creatures and snails. What salt do you think will be produced by the reaction of hydrochloric acid and calcium carbonate? hydrochloric acid 32 of 47 + calcium carbonate chloride + carbon dioxide + water © Boardworks Ltd 2008
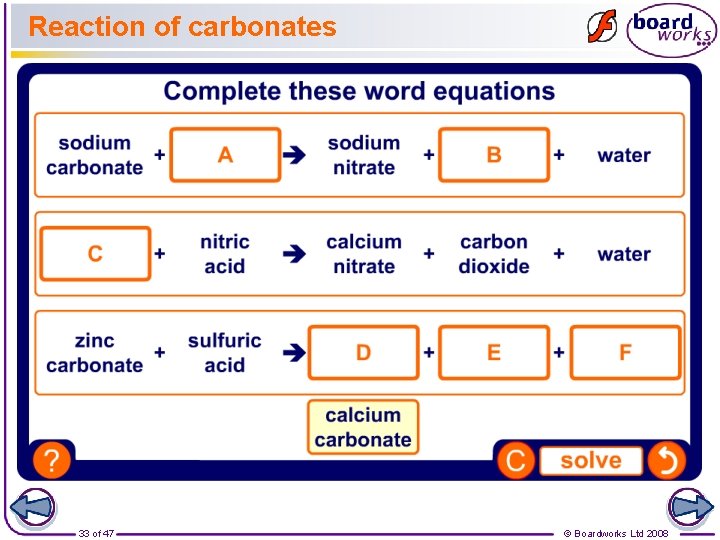
Reaction of carbonates 33 of 47 © Boardworks Ltd 2008
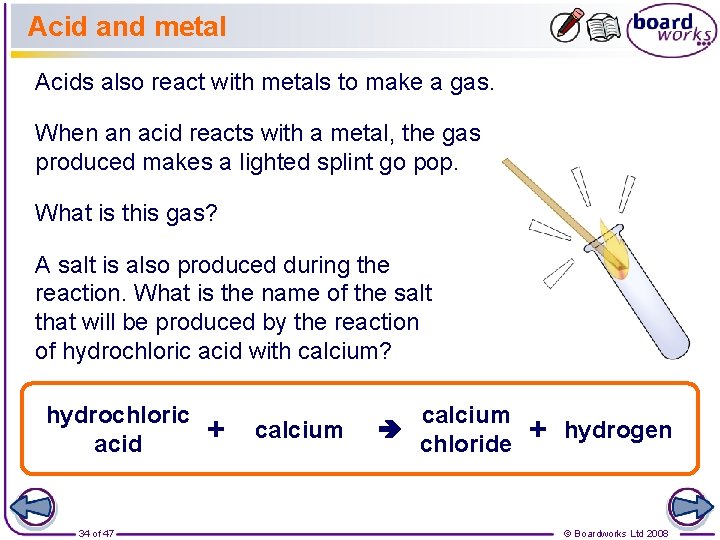
Acid and metal Acids also react with metals to make a gas. When an acid reacts with a metal, the gas produced makes a lighted splint go pop. What is this gas? A salt is also produced during the reaction. What is the name of the salt that will be produced by the reaction of hydrochloric acid with calcium? hydrochloric acid 34 of 47 + calcium chloride + hydrogen © Boardworks Ltd 2008

Choose your reaction Some metals react violently with acids, so it wouldn’t be safe to use them to make a salt in the classroom. However, if you still wanted to make that salt, you could use the metal carbonate instead. Sodium reacts very violently with sulfuric acid, so should not be uses to make sodium sulfate. However, you could safely use sodium carbonate. sulfuric acid + sodium sulfate carbonate + carbon dioxide + water Could you safely use sodium hydroxide to make sodium sulfate? What about sodium oxide? 35 of 47 © Boardworks Ltd 2008

Choose your reaction 36 of 47 © Boardworks Ltd 2008

What is acid rain? Rainwater has a p. H of about 5. 6, which means that it is naturally slightly acidic. This natural acidity is due to carbon dioxide in the air dissolving into the rain, forming a weak acid called carbonic acid. Rainwater with a lower p. H than normal is called acid rain. Burning of fossil fuels in power stations and cars forms pollutants such as sulfur dioxide and nitrogen oxides. These gases dissolve in rainwater to make sulfuric acid and nitric acid, which are strong acids, so also form acid rain. Why is acid rain a serious environmental problem? 37 of 47 © Boardworks Ltd 2008

How does acid rain work? 38 of 47 © Boardworks Ltd 2008

Effects of acid rain Acid rain has many damaging effects on the environment: l The acids in acid rain speed up the chemical weathering of rocks and buildings. l Rivers, lakes and streams become more acidic so that animals and plants cannot live in them. l Soil that becomes more acidic has fewer nutrients so trees and other plants cannot grow. Acid rain also damages trees by breaking down the waxy coating of leaves. What can be done to reduce acid rain and its effects? 39 of 47 © Boardworks Ltd 2008

Reducing acid rain Governments are working to reduce the emissions of the acidic pollutants produced by burning fossil fuels. l Sulfur dioxide is formed when coal containing sulfur is burned in power stations. This acidic pollutant can be removed from the gases before they are released into the atmosphere. l Nitrogen oxides are formed when petrol burns in a car’s engine. Exhaust systems can be fitted with a catalytic converter which turns harmful gases into harmless gases. How do these changes help to reduce acid rain? 40 of 47 © Boardworks Ltd 2008

Acid rain – a global problem Forests in Scandinavian countries such as Sweden have suffered greatly from the damaging effects of acid rain. However, scientists have shown that the acid rain that falls in Sweden is not produced there. The source of the acidic pollutants is in fact more likely to be UK factories and power stations. This happens because air pollution can be carried high up into the sky, where it is pushed by strong winds towards other countries. Is it right that a country can directly cause pollution in another? 41 of 47 © Boardworks Ltd 2008
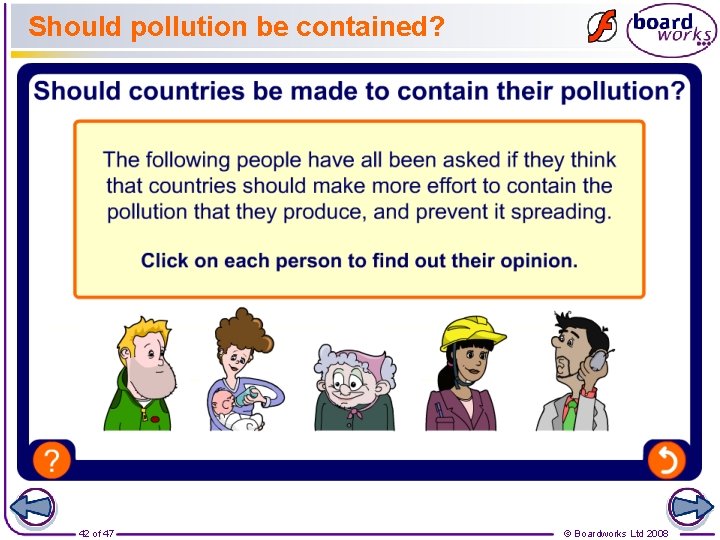
Should pollution be contained? 42 of 47 © Boardworks Ltd 2008

Limiting the effects of acid rain: opinions 43 of 47 © Boardworks Ltd 2008

44 of 47 © Boardworks Ltd 2008

Glossary 45 of 47 © Boardworks Ltd 2008
Anagrams 46 of 47 © Boardworks Ltd 2008

Multiple-choice quiz 47 of 47 © Boardworks Ltd 2008
- Slides: 47