1 of 45 Boardworks Ltd 2009 2 of

1 of 45 © Boardworks Ltd 2009

2 of 45 © Boardworks Ltd 2009

How many particles? Scientists often need to know how many particles of a substance they are dealing with. Unfortunately, atoms and molecules are tiny and it is impossible to count them directly. Instead of counting, scientists use the relative masses of different atoms and molecules to measure how many are present in a given sample. 3 of 45 …thirty-four million and one… © Boardworks Ltd 2009

How many particles? For example: One carbon atom has a relative atomic mass of 12. One magnesium atom has a relative atomic mass of 24. The quantity of atoms in 24 g of pure graphite can be calculated as 24 = 2 12 The quantity of atoms in 24 g of pure magnesium can be calculated as 24 = 1 24 So, 24 g of carbon contains twice as many atoms as 24 g of magnesium, because magnesium atoms are twice as heavy. The unit for these results is called a mole. 4 of 45 © Boardworks Ltd 2009

What are moles? The term mole is used to describe how many atoms or molecules of a substance are present in a sample. One mole of a substance is equal to its relative atomic mass, or relative formula mass, in grams. relative atomic mass For example, the relative atomic mass of carbon is 12, so one mole of carbon atoms weighs 12 g. 24 g of pure graphite therefore contains 2 moles of carbon atoms. 5 of 45 © Boardworks Ltd 2009
How many moles? 6 of 45 © Boardworks Ltd 2009

How much is in a mole? Scientists have calculated that one mole of any substance contains 602, 000, 000, 000 (6. 02 × 1023) particles. One mole of carbon weighs 12 g, so 12 g of carbon contains 6. 02 × 1023 carbon atoms. One mole of sodium weighs 23 g, so 23 g of sodium contains 6. 02 × 1023 carbon atoms. One mole of water weighs 18 g, so 18 g of water contains 6. 02 × 1023 water molecules. How many carbon atoms are in 6 g of carbon? 7 of 45 3. 01 × 1023 © Boardworks Ltd 2009
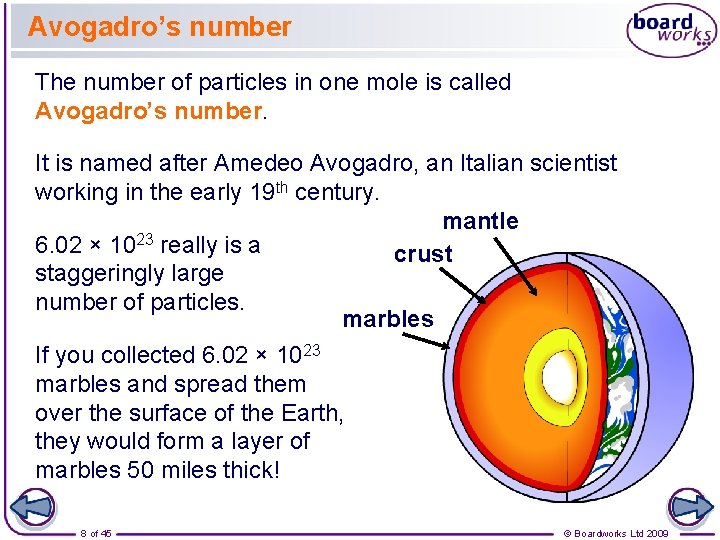
Avogadro’s number The number of particles in one mole is called Avogadro’s number. It is named after Amedeo Avogadro, an Italian scientist working in the early 19 th century. mantle 6. 02 × 1023 really is a crust staggeringly large number of particles. marbles If you collected 6. 02 × 1023 marbles and spread them over the surface of the Earth, they would form a layer of marbles 50 miles thick! 8 of 45 © Boardworks Ltd 2009

Calculating moles The number of moles in a sample is calculated using this equation: number of moles = mass of substance molar mass Remember: l mass of substance is measured in grams. l some elements exist as molecules (for example, O 2). You will need to use the molecular rather than atomic mass for these. l for compounds, the molar mass is the sum of the relative atomic masses of all the atoms in the formula. 9 of 45 © Boardworks Ltd 2009

Calculating molar mass 10 of 45 © Boardworks Ltd 2009

Calculating moles: questions number of moles = mass of substance molar mass 1. How many moles of iron are in 28 g of pure iron? Number of moles = 28 = 0. 5 moles of iron 56 2. How many moles of CO 2 are in 11 g of carbon dioxide? Relative molecular mass = 12 + 16 = 44 Number of moles = 11 = 0. 25 moles of CO 2 44 11 of 45 © Boardworks Ltd 2009

Using moles in calculations Moles are very useful when reacting substances together. They provide us with a quick way of calculating how many particles of each substance are present. Chemists use moles to calculate: l how much of each reactant they will need l how much of a substance they are likely to make in the end. 12 of 45 © Boardworks Ltd 2009

Using moles: example 1 If you reacted 56 g of iron with excess copper sulfate solution, what mass of copper would you expect to make? Step 1: Write a balanced equation for the reaction: Fe + Cu. SO 4 Cu + Fe. SO 4 Step 2: Turn the mass of iron into moles of iron: mass 56 moles = = = 1 mole of iron molar mass 56 Step 3: Use the balanced equation to work out how many moles of copper you should make: 1 mole of Fe makes 1 mole of Cu Step 4: Turn moles of copper into mass of copper: mass = moles × RAM = 1 × 63. 5 = 63. 5 g of Cu 13 of 45 © Boardworks Ltd 2009

Using moles: example 2 What mass of iron oxide would you need to react with excess aluminium to make 1120 g of iron? Step 1: Write a balanced equation for the reaction: Fe 2 O 3 + 2 Al Al 2 O 3 + 2 Fe Step 2: Turn mass of iron into moles of iron: mass 1, 120 moles = = = 20 moles of iron RAM 56 Step 3: Use the balanced equation to work out how many moles of iron oxide you would need: 1 mole of iron oxide makes 2 moles of iron, so 10 moles of iron oxide are needed to make 20 moles of iron. Step 4: Turn moles of iron oxide into mass of iron oxide: mass = moles × RAM = 10 × 160 = 1, 600 g of Fe 2 O 3 14 of 45 © Boardworks Ltd 2009
Using moles in calculations 15 of 45 © Boardworks Ltd 2009

16 of 45 © Boardworks Ltd 2009
Amedeo Avogadro 17 of 45 © Boardworks Ltd 2009

Avogadro’s law states that: Equal volumes of gases, at the same temperature and pressure, contain the same number of molecules. This means that one mole of any gas at a given temperature and pressure will always have the same volume. At room temperature and pressure (RTP), one mole of any gas has a volume of 24 dm 3 (24 litres). What would happen to the volume of a mole of gas if you: l increased the temperature? l increased the pressure? 18 of 45 © Boardworks Ltd 2009

Gases and moles 19 of 45 © Boardworks Ltd 2009

Gases and moles 20 of 45 © Boardworks Ltd 2009

Gases and moles 21 of 45 © Boardworks Ltd 2009

Gas calculations: example 1 Hydrogen gas reacts with oxygen gas to make water vapour. If 24 dm 3 of hydrogen gas is burned with excess oxygen, what volume of water vapour is produced? Step 1: Write a balanced equation for the reaction: 2 H 2 (g) + O 2 (g) 2 H 2 O (g) Step 2: Turn volume of hydrogen into moles of hydrogen: moles of H 2 = volume = 24 = 1 mole of H 2 24 24 Step 3: Use the balanced equation to work out how many moles of water vapour you should make: 1 mole of hydrogen makes 1 mole of water vapour Step 4: Turn moles of water into volume of water vapour: volume = moles × 24 = 1 × 24 = 24 dm 3 of water vapour 22 of 45 © Boardworks Ltd 2009

Gas calculations: example 2 Nitrogen and hydrogen react together to make ammonia. What is the maximum volume of ammonia that can be made from 2, 400 cm 3 of nitrogen gas? Step 1: Write a balanced equation for the reaction: N 2 (g) + 3 H 2 (g) 2 NH 3 (g) Step 2: Turn volume of nitrogen into moles of nitrogen: moles of N 2 = volume 24 = 2. 4 24 NOTE: To convert cm 3 to dm 3, divide by 1, 000. = 0. 1 moles of N 2 23 of 45 © Boardworks Ltd 2009

Gas calculations: example 2 continued Step 3: Use the balanced equation to work out how many moles of ammonia you can make: 1 mole of N 2 makes 2 moles of NH 3, so 0. 1 moles of N 2 makes 0. 2 moles of NH 3. Step 4: Turn moles of ammonia into volume of ammonia: volume = moles × 24 = 0. 2 × 24 = 4. 8 dm 3 (4, 800 cm 3) of ammonia 24 of 45 © Boardworks Ltd 2009
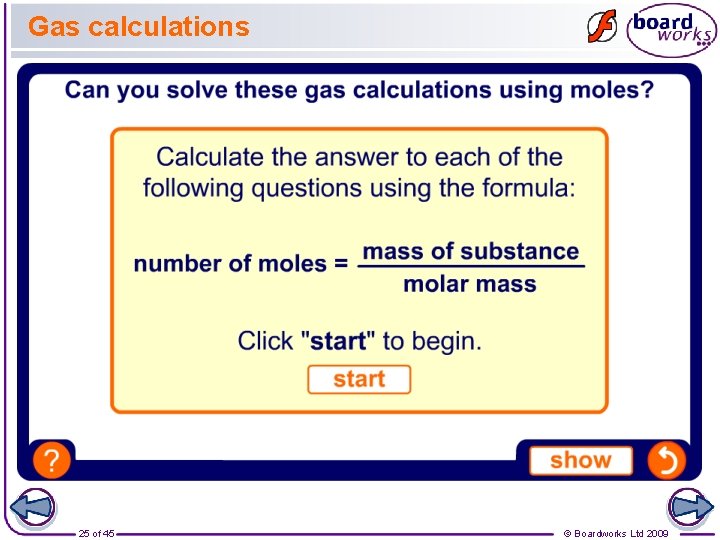
Gas calculations 25 of 45 © Boardworks Ltd 2009

26 of 45 © Boardworks Ltd 2009

Using gases to follow reactions Many chemical reactions produce gases. Scientists can measure how much gas is produced and use it to follow the progress of a reaction. The volume of gas produced can also be converted into moles and used to determine the amount of reactants present at the start of the reaction. Which gas is produced by each of these reactions? Magnesium + hydrochloric acid hydrogen Calcium carbonate + hydrochloric acid Decomposition of hydrogen peroxide 27 of 45 carbon dioxide oxygen © Boardworks Ltd 2009

Measuring gases in reactions One way of measuring how much gas is produced in a reaction is to use an upturned measuring cylinder filled with water. As the gas bubbles into the measuring cylinder, it displaces the water. This technique works well for gases which are less dense than air and not very soluble in water. What would be the problem with using this method for a gas which is very soluble in water? 28 of 45 © Boardworks Ltd 2009
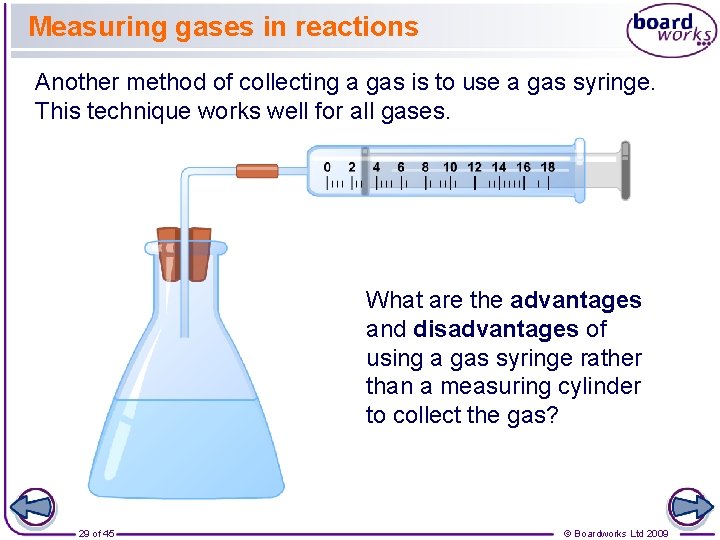
Measuring gases in reactions Another method of collecting a gas is to use a gas syringe. This technique works well for all gases. What are the advantages and disadvantages of using a gas syringe rather than a measuring cylinder to collect the gas? 29 of 45 © Boardworks Ltd 2009

Measuring gases in reactions One other method of measuring how much gas is produced in a reaction is to measure the mass of the reaction mixture. What will happen to the reading on the balance? Why is cotton wool placed in the top of the flask? 30 of 45 © Boardworks Ltd 2009

Limiting reactants The amount of gas produced in a reaction will depend on the limiting reactant. This is the reactant that runs out first, bringing the reaction to an end. The other reactant(s) are said to be “in excess”. This means that there will be some left after the reaction. This table shows hydrogen gas production during a reaction between a small amount of magnesium and excess dilute hydrochloric acid. After how many seconds was all the magnesium used up? 31 of 45 time (s) volume H 2 (cm 3) 0 30 60 90 120 150 180 210 240 270 300 0 150 300 450 600 780 850 895 910 © Boardworks Ltd 2009
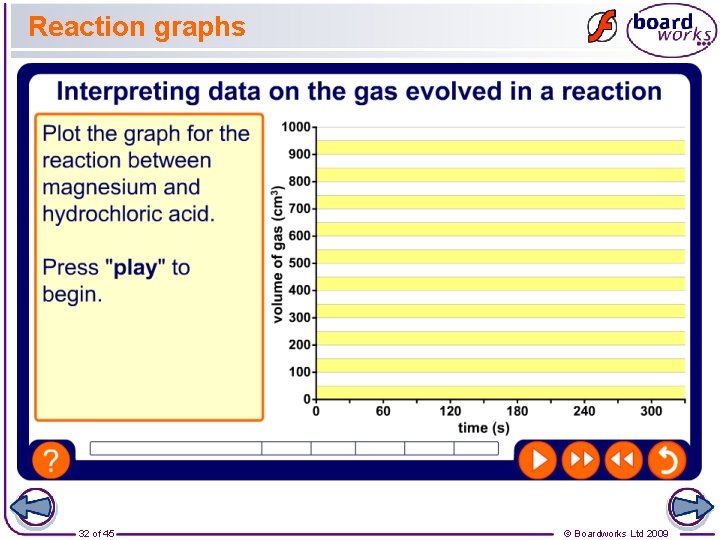
Reaction graphs 32 of 45 © Boardworks Ltd 2009
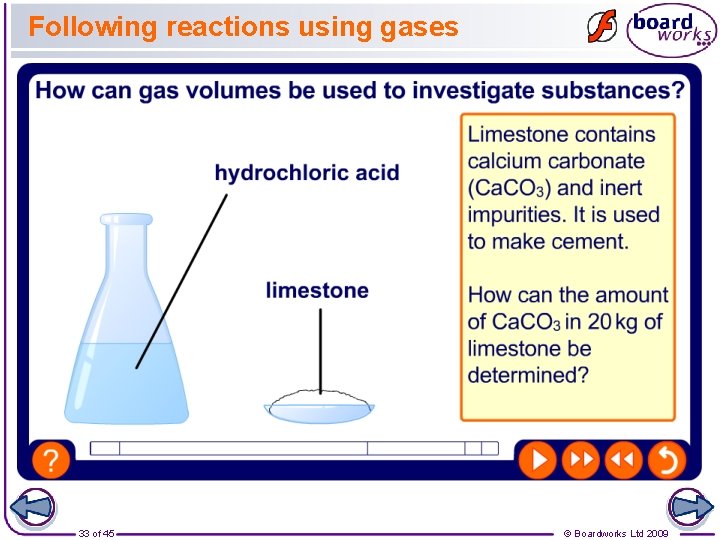
Following reactions using gases 33 of 45 © Boardworks Ltd 2009

34 of 45 © Boardworks Ltd 2009
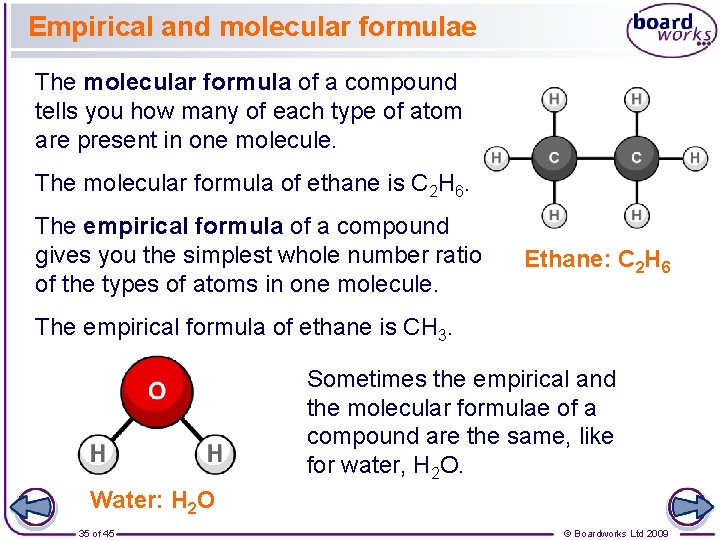
Empirical and molecular formulae The molecular formula of a compound tells you how many of each type of atom are present in one molecule. The molecular formula of ethane is C 2 H 6. The empirical formula of a compound gives you the simplest whole number ratio of the types of atoms in one molecule. Ethane: C 2 H 6 The empirical formula of ethane is CH 3. Sometimes the empirical and the molecular formulae of a compound are the same, like for water, H 2 O. Water: H 2 O 35 of 45 © Boardworks Ltd 2009

Empirical and molecular formulae 36 of 45 © Boardworks Ltd 2009

Why are empirical formulae useful? Chemists sometimes make compounds whose identity they are unsure of. Finding the empirical formula of an unknown compound is an important step in identifying it. The empirical formula can often be determined by studying the reactions of the unknown compound. For example, an unknown hydrocarbon can be burned in oxygen to produce carbon dioxide and water vapour. Chemists can measure the volumes of the gases produced by the reaction and use them to work out the empirical formula. 37 of 45 © Boardworks Ltd 2009
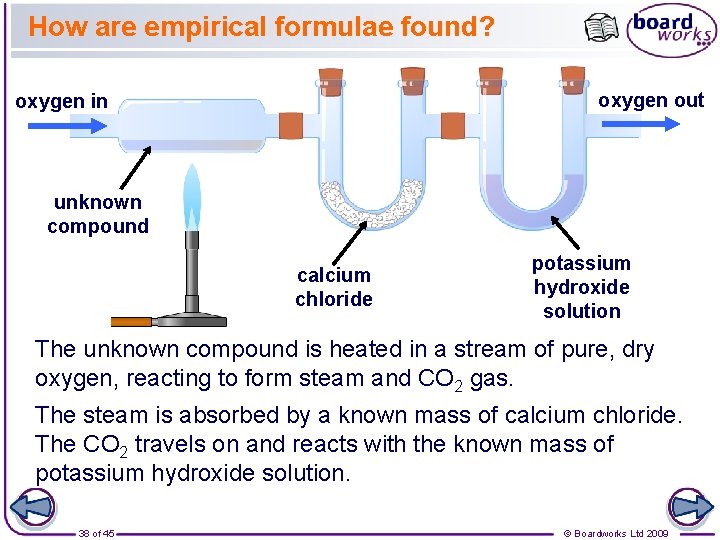
How are empirical formulae found? oxygen out oxygen in unknown compound calcium chloride potassium hydroxide solution The unknown compound is heated in a stream of pure, dry oxygen, reacting to form steam and CO 2 gas. The steam is absorbed by a known mass of calcium chloride. The CO 2 travels on and reacts with the known mass of potassium hydroxide solution. 38 of 45 © Boardworks Ltd 2009

How are empirical formulae found? The change in mass of the calcium chloride and the potassium hydroxide can be used to calculate the relative masses of carbon and hydrogen in the original compound. For example: If the calcium chloride increased in mass by 27 grams and the potassium hydroxide solution increased by 66 grams… 27 g of H 2 O was made. H 2 O = 1 + 16 = 18. There are 2 g of hydrogen in every 18 g of water. 2 × 27 = 3 g of hydrogen were produced. 18 CO 2 = 12 + 16 = 44. 66 g of CO 2 was made. There are 12 g of carbon in every 44 g of carbon dioxide. 12 × 66 = 18 g of carbon were produced. 44 39 of 45 © Boardworks Ltd 2009

Empirical formulae from masses 40 of 45 © Boardworks Ltd 2009
Calculating from masses or percentages 41 of 45 © Boardworks Ltd 2009

42 of 45 © Boardworks Ltd 2009

Glossary 43 of 45 © Boardworks Ltd 2009

Anagrams 44 of 45 © Boardworks Ltd 2009

Multiple-choice quiz 45 of 45 © Boardworks Ltd 2009
- Slides: 45