1 of 40 Boardworks Ltd 2008 2 of
1 of 40 © Boardworks Ltd 2008

2 of 40 © Boardworks Ltd 2008

Energy changes in chemical reactions Some chemical reactions give out energy. For example: l Magnesium burning – this reaction gives out heat and light energy. l A candle burning – this reaction gives out heat and light energy. l A battery – this reaction gives out electrical energy. l A firework or bomb – this reaction gives out light, heat and sound energy. 3 of 40 © Boardworks Ltd 2008

Exothermic reactions Reactions that give out heat energy are called exothermic. Combustion is an example of an exothermic reaction – for example, when a piece of wood is burned, heat is produced. Reactions that take in heat energy are called endothermic. Sherbet dissolving on your tongue is an example of an endothermic reaction. 4 of 40 © Boardworks Ltd 2008

An exothermic reaction 5 of 40 © Boardworks Ltd 2008

An endothermic reaction 6 of 40 © Boardworks Ltd 2008

Endothermic or exothermic? 7 of 40 © Boardworks Ltd 2008

Types of chemical reactions There are many different types of chemical reaction. For example, oxidation is the reaction of a substance with oxygen. What is the word equation for the oxidation of copper? copper + oxygen copper oxide Reduction is the opposite of oxidation. What do you think is formed in the reduction of magnesium oxide? magnesium oxide 8 of 40 magnesium + oxygen © Boardworks Ltd 2008

Heating copper carbonate When you heat copper carbonate, it breaks up to form copper oxide and carbon dioxide. What is the word equation for this reaction? copper carbonate copper oxide + carbon dioxide This reaction is called thermal decomposition. Can you explain why? Thermal means heat, and decomposition means breaking into smaller pieces. So in a thermal decomposition reaction, heat is used to break a substance into smaller pieces. 9 of 40 © Boardworks Ltd 2008

Combustion A common combustion reaction is petrol burning with oxygen in a car engine. The products of this reaction are carbon dioxide and water. What is the word equation for this combustion reaction? petrol + oxygen carbon dioxide + water Combustion involves reacting with oxygen, so it can also be classified as another type of reaction. Which one? 10 of 40 © Boardworks Ltd 2008

Types of chemical reaction Displacement reactions are commonly used to obtain pure metals from their ores. Displacement means replacing something with something else. For example, copper can be displaced from copper sulfate by reacting it with zinc. copper sulfate + zinc sulfate + copper What would be displaced in the reaction between zinc sulfate and magnesium? zinc sulfate 11 of 40 + magnesium sulfate + zinc © Boardworks Ltd 2008

Types of chemical reaction What is neutralization? Neutralization is the reaction between an acid an alkali to make a neutral substance. In the neutralization reaction between hydrochloric acid and sodium hydroxide, the products are sodium chloride and water. What is the word equation for this reaction? hydrochloric acid 12 of 40 + sodium hydroxide chloride + water © Boardworks Ltd 2008

Which type of reaction? 13 of 40 © Boardworks Ltd 2008

14 of 40 © Boardworks Ltd 2008
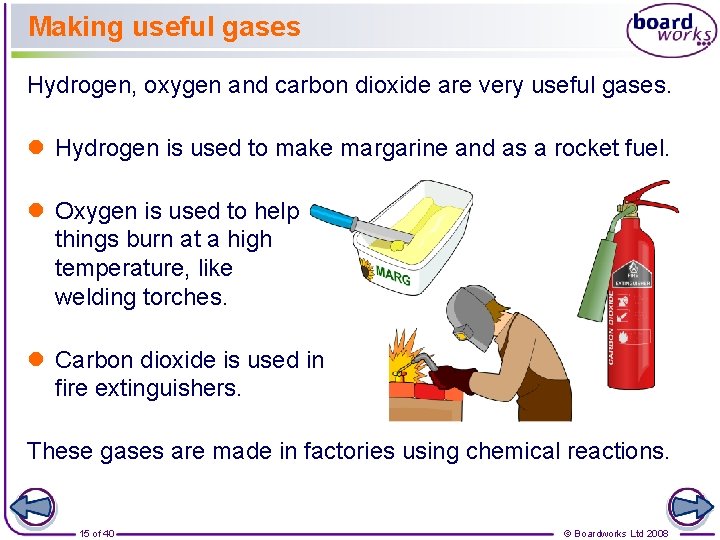
Making useful gases Hydrogen, oxygen and carbon dioxide are very useful gases. l Hydrogen is used to make margarine and as a rocket fuel. l Oxygen is used to help things burn at a high temperature, like welding torches. l Carbon dioxide is used in fire extinguishers. These gases are made in factories using chemical reactions. 15 of 40 © Boardworks Ltd 2008

Making and identifying hydrogen 16 of 40 © Boardworks Ltd 2008

Making and identifying oxygen 17 of 40 © Boardworks Ltd 2008
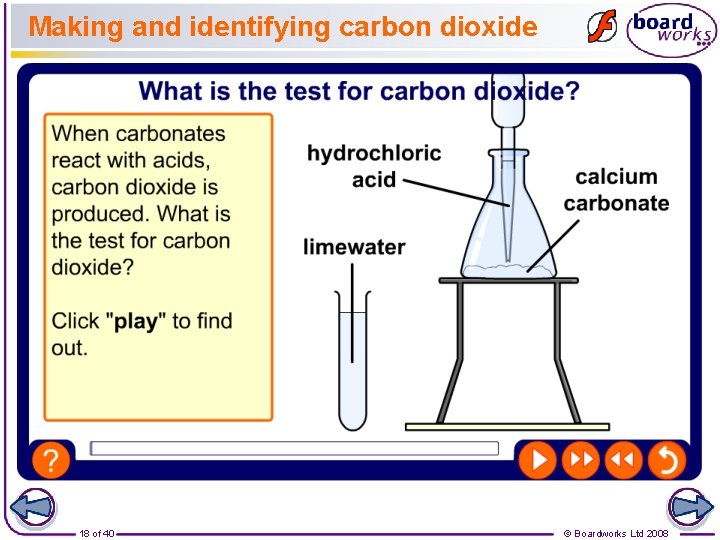
Making and identifying carbon dioxide 18 of 40 © Boardworks Ltd 2008

Word equations for making gases 19 of 40 © Boardworks Ltd 2008

Which gas? 20 of 40 © Boardworks Ltd 2008
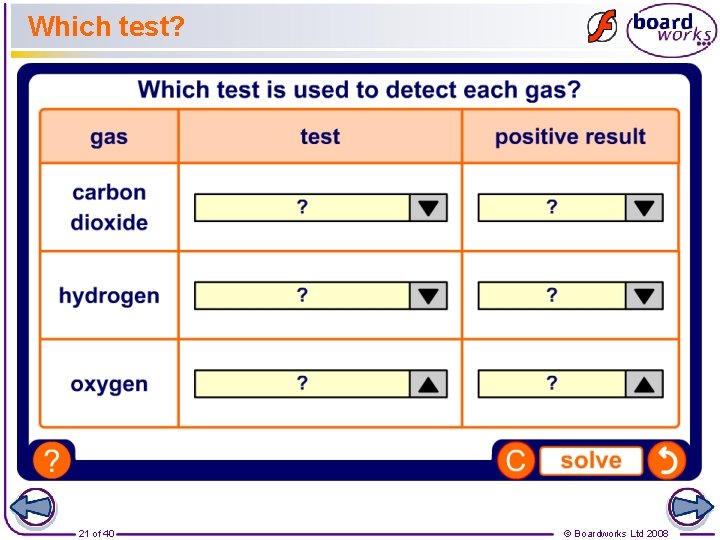
Which test? 21 of 40 © Boardworks Ltd 2008

22 of 40 © Boardworks Ltd 2008

What is combustion? Combustion is the scientific word for burning. It is the chemical reaction that takes place when a substance burns and reacts with oxygen to produce heat and light energy. 23 of 40 © Boardworks Ltd 2008

Bell jar experiment 24 of 40 © Boardworks Ltd 2008
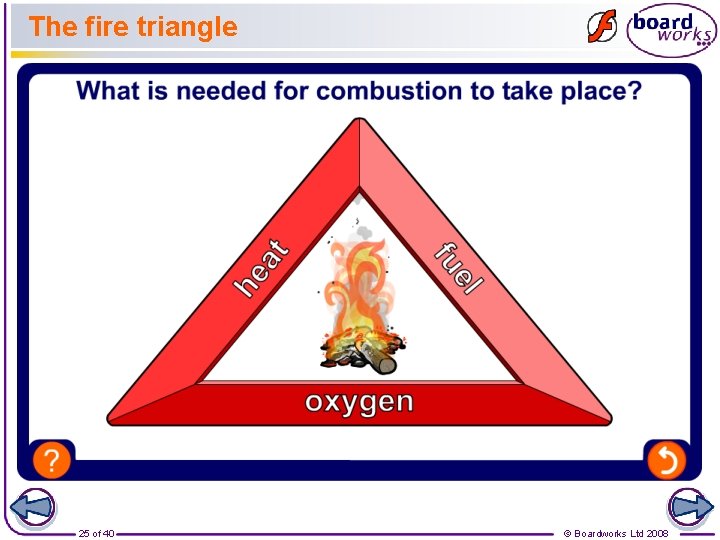
The fire triangle 25 of 40 © Boardworks Ltd 2008

Putting out fires If a fire starts accidentally, it is important to be able to put it out quickly and safely. To put out a fire, you can cut off the supply of any side of the fire triangle. For example: l putting water on burning wood cuts off the heat. l putting sand on burning oil cuts off the oxygen. l cutting down trees in a forest fire cuts off the supply of fuel. 26 of 40 © Boardworks Ltd 2008

Products of combustion 27 of 40 © Boardworks Ltd 2008

Combustion of methane Natural gas, methane, is often burned for cooking. Methane is made up of carbon and hydrogen. l What gas does methane burn with? oxygen l What substance will the carbon in methane change into when it burns? carbon dioxide l What substance will the hydrogen in methane change into when it burns? water l What is the word equation for the combustion of methane? methane 28 of 40 + oxygen carbon dioxide + water © Boardworks Ltd 2008

Word equations for combustion When substances combust, the new substances formed are called ‘oxides’. What are the word equations for these combustion reactions? 1. Coal (made from carbon) burns and forms carbon dioxide. carbon + oxygen carbon dioxide 2. Hydrogen burns and forms dihydrogen oxide (i. e water!). hydrogen + oxygen water 3. Methane burns and forms carbon dioxide and water. methane 29 of 40 + oxygen carbon dioxide + water © Boardworks Ltd 2008

Symbol equations for combustion What are the symbol equations for these combustion reactions? 1. Coal (made from carbon) burns and forms carbon dioxide. + + carbon C oxygen carbon dioxide O 2 CO 2 2. Hydrogen burns and forms dihydrogen oxide (i. e water!). hydrogen 2 H 2 + + oxygen water O 2 2 H 2 O 3. Methane burns and forms carbon dioxide and water. methane CH 4 30 of 40 + + oxygen carbon dioxide 2 O 2 CO 2 + + water 2 H 2 O © Boardworks Ltd 2008

Complete and incomplete combustion Fuels like methane, butane and petrol are hydrocarbons. Hydrocarbons are compounds made of carbon and hydrogen. When these hydrocarbon fuels burn in a good supply of oxygen, they burn completely to make carbon dioxide and water. If there is a lack of oxygen, incomplete combustion takes place. This means that the fuel burns to produce water, carbon monoxide and carbon particles. 31 of 40 © Boardworks Ltd 2008

Incomplete combustion is a hazardous problem because the products of the process are toxic. Carbon monoxide prevents people’s blood from carrying oxygen. Carbon particles make city buildings very dirty, and they can get into people’s lungs and cause breathing problems. What precautions do you think people take in their homes to prevent incomplete combustion? 32 of 40 © Boardworks Ltd 2008

Using fossil fuels Coal, oil and natural gas are called fossil fuels. Burning fossil fuels, like coal, petrol and natural gas, provides more than 90% of the energy we need for transport, factories and in our homes. How different would life be without fossil fuels? 33 of 40 © Boardworks Ltd 2008

Crude oil and fractional distillation The oil that comes out of the ground is called crude oil. It cannot be used as it is, because it is a mixture of many different substances with different boiling points. How can you separate a mixture of substances with different boiling points? To separate out the useful substances, a process called fractional distillation is carried out. big molecules boil at a high temperature 34 of 40 small molecules boil at a lower temperature © Boardworks Ltd 2008
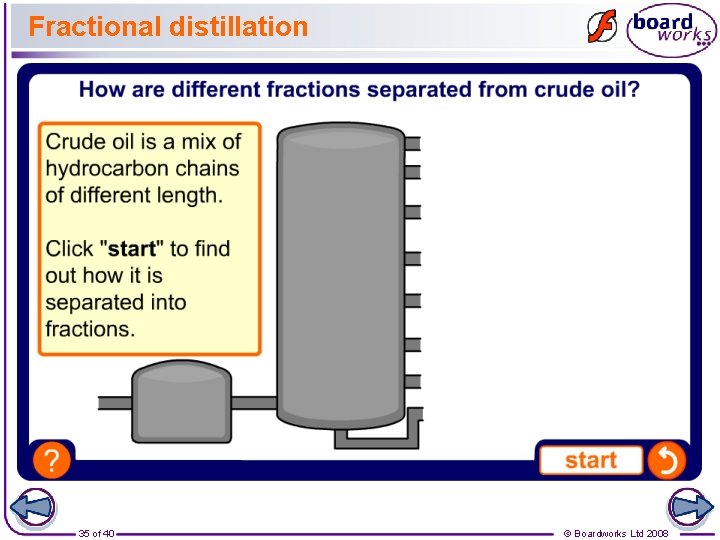
Fractional distillation 35 of 40 © Boardworks Ltd 2008
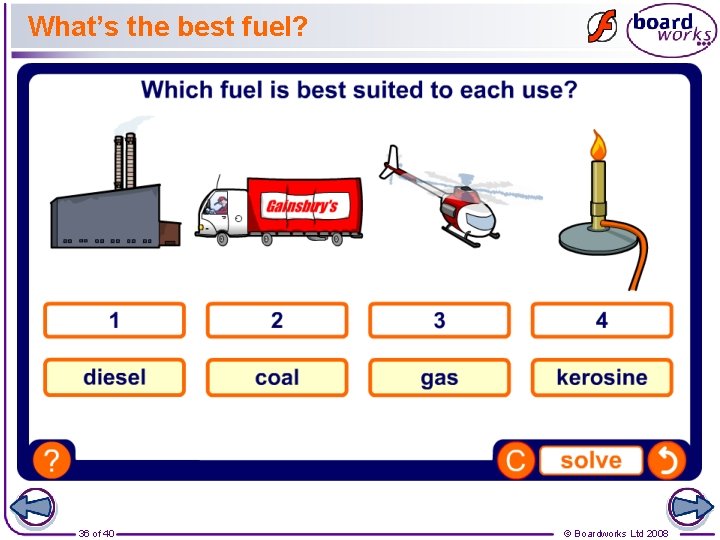
What’s the best fuel? 36 of 40 © Boardworks Ltd 2008

37 of 40 © Boardworks Ltd 2008

Glossary 38 of 40 © Boardworks Ltd 2008

Anagrams 39 of 40 © Boardworks Ltd 2008
Multiple-choice quiz 40 of 40 © Boardworks Ltd 2008
- Slides: 40