1 of 40 Boardworks Ltd 2007 2 of
1 of 40 © Boardworks Ltd 2007

2 of 40 © Boardworks Ltd 2007

What are atoms made of? Atoms are made of three basic building blocks called protons, neutrons and electrons. There are two properties of protons, neutrons and electrons that are especially important: mass and charge. Particle Mass Charge proton 1 +1 neutron 1 0 electron almost 0 -1 In any atom, the number of electrons is equal to the number of protons and so the overall charge of an atom is zero. 3 of 40 © Boardworks Ltd 2007

Particles in the modern model 4 of 40 © Boardworks Ltd 2007

What makes a carbon atom carbon? The atoms of any particular element always contains the same number of protons. Carbon atoms always have six protons. Atoms with different numbers of protons must be other elements. For example: l all atoms with 1 proton are hydrogen atoms; l all atoms with 17 protons are chlorine atoms. In the periodic table, there are two numbers found with each element. What do these numbers represent? Mass number is the number of protons + the number of neutrons. Atomic number (or proton number) is the number of protons 5 of 40 © Boardworks Ltd 2007

What are isotopes? All carbon atoms have the same number of protons, but not all carbon atoms are identical. Although atoms of the same element always have the same number of protons, they can have different numbers of neutrons. Atoms that differ in this way are called isotopes. For example, carbon exists as three different isotopes: carbon-12, carbon-13 and carbon-14: mass number is different atomic number is the same Potassium is another element that exists as three different isotopes: potassium-39, potassium-40 and potassium-41. 6 of 40 © Boardworks Ltd 2007
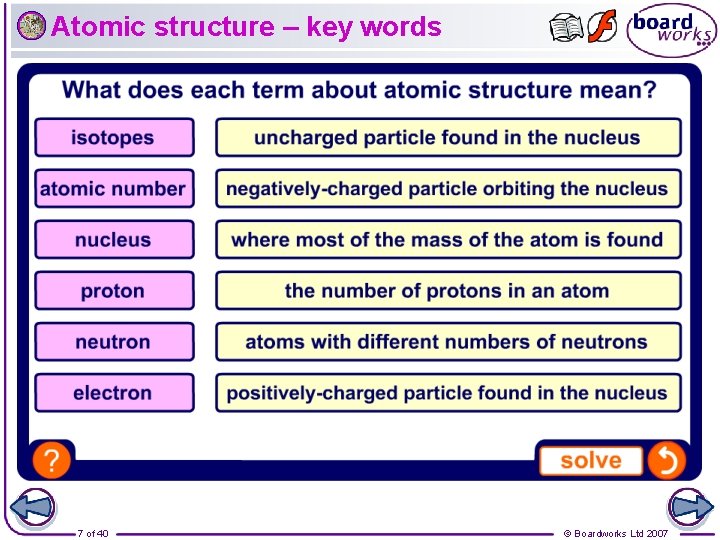
Atomic structure – key words 7 of 40 © Boardworks Ltd 2007
8 of 40 © Boardworks Ltd 2007

Types of radioactive decay 9 of 40 © Boardworks Ltd 2007

What happens during alpha decay? An alpha particle consists of two protons and two neutrons. It is the same as a helium nucleus. When an atom’s nucleus decays and releases an alpha particle, it loses two protons and two neutrons. mass number decreases by 4 238 U 92 234 Th 90 + 4 α 2 atomic number decreases by 2 The number of protons has changed, so the decayed atom has changed into a new element. 10 of 40 © Boardworks Ltd 2007

What happens during beta decay? An beta particle consists of a high energy electron, which is emitted by the nucleus of the decaying atom. When an atom’s nucleus decays and releases a beta particle, a neutron turns into a proton, which stays in the nucleus, and a high energy electron, which is emitted. mass number remains the same 14 C 6 14 N 7 + β atomic number increases by 1 The decayed atom has gained a proton and so has changed into a new element. 11 of 40 © Boardworks Ltd 2007

What happens during gamma decay? Gamma radiation is a form of electromagnetic radiation, not a type of particle. When an atom’s nucleus decays and emits gamma radiation, it releases energy in the form of electromagnetic radiation. There is no change to the make up of the nucleus and so a new element is not formed. Gamma rays are usually emitted with alpha or beta particles. For example, cobalt-60 decays releasing a beta particle. The nickel formed is still not stable and so emits gamma radiation. 60 27 Co 12 of 40 60 28 Ni* + β 60 28 Ni + The nickel does not change into a new element. © Boardworks Ltd 2007

Radioactive decay – true or false? 13 of 40 © Boardworks Ltd 2007

14 of 40 © Boardworks Ltd 2007

How can radioactivity be measured? Radioactivity cannot be seen, it has no smell and does not make any sound so how can it be detected? Radioactivity can be detected with a Geiger counter, which is a Geiger-Müller (GM) tube connected to a ratemeter. It can also be used to measure the amount of radiation. GM tube ratemeter The ratemeter gives a reading in ‘counts per second’ and a loudspeaker ‘clicks’ for each particle, or burst of radiation, detected by the GM tube. 15 of 40 © Boardworks Ltd 2007

What happens to radioactivity? 16 of 40 © Boardworks Ltd 2007

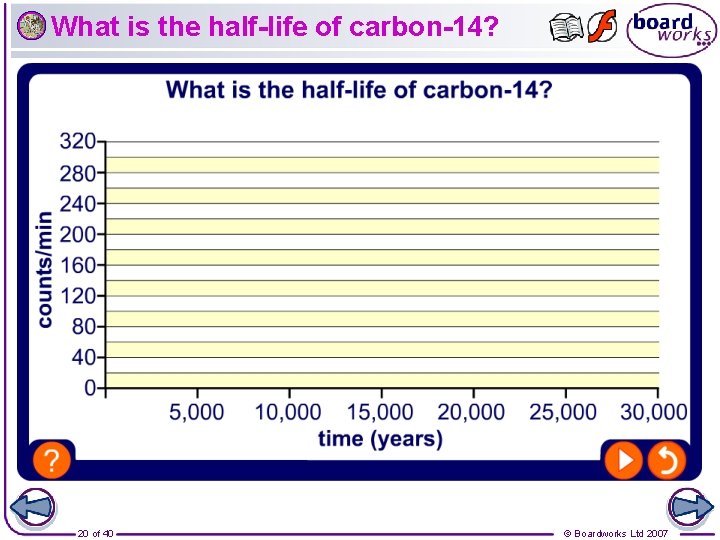
What is half-life? Radioactive decay is a spontaneous process that cannot be controlled and is not affected by temperature. However, each radioactive element has its own particular decay rate, which is called the half-life. The half-life of a radioactive element is the time that it takes half the atoms in a sample to decay. For example, the half-life of the isotope iodine-131 is 8 days. This means that after 8 days half the atoms in a sample of iodine-131 have decayed. 8 days later half the remaining atoms have decayed and so on. 17 of 40 © Boardworks Ltd 2007
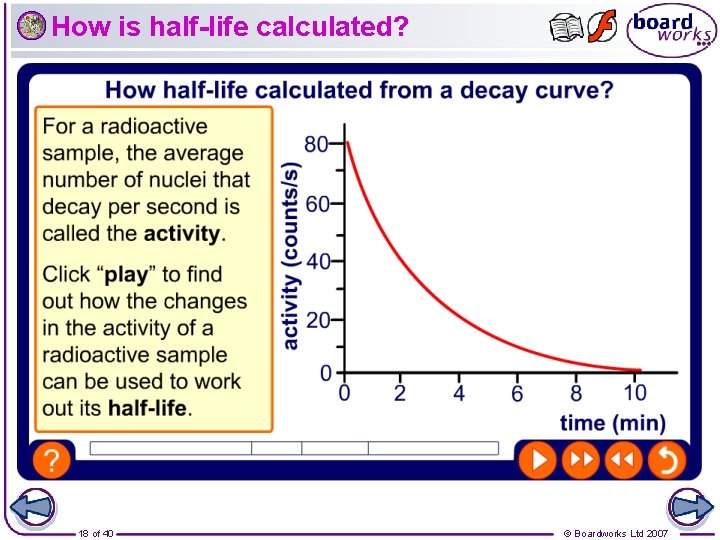
How is half-life calculated? 18 of 40 © Boardworks Ltd 2007

How long are half-lives? Half-lives range from millionths of a second to millions of years. Some types of nuclei are more unstable than others and decay at a faster rate. Radioisotope Half-life boron-12 0. 02 seconds radium-226 uranium-235 1602 years 710 million years Xenon-133 is a radioactive isotope used for studying lung function. Why does its half-life of 5. 2 days make it suitable for this use? Uranium-235, which is used in nuclear reactors and nuclear weapons, has a half-life of 710 million years. Why is the use of uranium-235 considered controversial? 19 of 40 © Boardworks Ltd 2007
What is the half-life of carbon-14? 20 of 40 © Boardworks Ltd 2007

How does carbon dating work? 21 of 40 © Boardworks Ltd 2007

What are the problems of using carbon dating? What are some of the problems with using carbon dating to predict the age of a sample? l The half-life of carbon-14 is 5, 700 years. If the sample is older than 60, 000 years, the amount of carbon-14 is too small to measure accurately. Instead, radioactive isotopes with longer half-lives, such as uranium-235 with a half-life of 710 million years, can be used to date older samples. l Samples can become contaminated with materials of a different age which may confuse the readings of carbon-14. l Carbon dating anything that died after the 1940 s, when nuclear bombs, nuclear reactors and open-air nuclear tests began, is harder to date precisely due to contamination from this increased background radioactivity. 22 of 40 © Boardworks Ltd 2007

Using half-life to date a sample Half-life can be used to do many useful calculations. For example, the half-life of carbon-14 is 5, 700 years. If a fossil bone has a count of 25, and a piece of bone from a living body has a count of 200, how old is the fossil? l After one half-life, the count will decrease by half to 100. l After the second half-life, the count decreases by half again to 50. l After the third half-life, the count decreases to 25. l Three half-lives of carbon-14 have passed, so 3 x 5, 700 years makes the fossil 17, 100 years old. 23 of 40 © Boardworks Ltd 2007

Using half-life in calculations 24 of 40 © Boardworks Ltd 2007

25 of 40 © Boardworks Ltd 2007

Glossary l atomic number – The number of protons in the nucleus of an atom, which is the same for all isotopes of an element. l half-life – The time taken for the number of radioactive atoms in a sample, or the count rate, to decrease by half. l isotopes – Different forms of the same element, with the same number of protons but different numbers of neutrons. l mass number – The total number of protons and neutrons in the nucleus of an atom, which differs for each isotope of an element. l radioactive decay – The breakdown of unstable radioactive nuclei by releasing radiation. l radioisotope – A radioactive isotope of an element, which may be naturally occurring or artificially created. 26 of 40 © Boardworks Ltd 2007
Anagrams 27 of 40 © Boardworks Ltd 2007
Multiple-choice quiz 28 of 40 © Boardworks Ltd 2007
- Slides: 28