1 of 36 Boardworks Ltd 2009 2 of
1 of 36 © Boardworks Ltd 2009

2 of 36 © Boardworks Ltd 2009

What are acids and alkalis? 3 of 36 © Boardworks Ltd 2009

Naming acids and alkalis 4 of 36 © Boardworks Ltd 2009

What are acids? Acids can be defined as substances that release H+ ions when they dissolve in water. When hydrogen chloride dissolves in water, it releases H+ ions and Cl– ions: HCl H+ + Cl– When ethanoic acid dissolves in water, it releases H+ ions and CH 3 COO– ions: CH 3 COOH H+ + CH 3 COO– Since acids release H+ ions, we call them H+ donors. 5 of 36 © Boardworks Ltd 2009

What are H+ ions? Hydrogen atoms contain one proton and one electron. A H+ ion is a hydrogen atom that has lost its single electron. This leaves a lone, positively charged proton. Because acids donate protons, they are sometimes called proton donors. When dissolved in water, the H+ ion is hydrated. This is represented in formulae by adding a state symbol, H+ (aq). 6 of 36 © Boardworks Ltd 2009

What are bases and alkalis? Bases are substances that react with acids. They absorb hydrogen ions (H+). Bases are therefore H+ acceptors, or proton acceptors. The oxides, hydroxides and carbonates of metals, such as sodium carbonate, are bases. Some bases are soluble in water: these bases are called alkalis. Alkalis release hydroxide ions (OH–) in solution. All alkalis are bases, but not all bases are alkalis. 7 of 36 bases: react with acids alkalis: soluble bases © Boardworks Ltd 2009
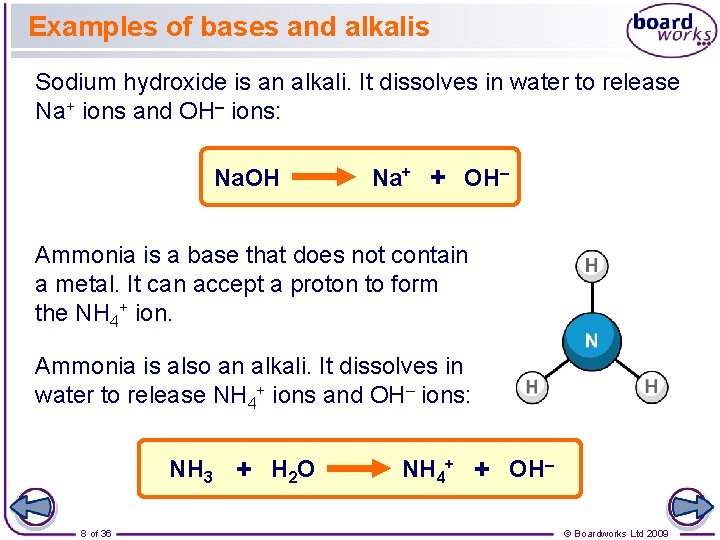
Examples of bases and alkalis Sodium hydroxide is an alkali. It dissolves in water to release Na+ ions and OH– ions: Na. OH Na+ + OH– Ammonia is a base that does not contain a metal. It can accept a proton to form the NH 4+ ion. Ammonia is also an alkali. It dissolves in water to release NH 4+ ions and OH– ions: NH 3 + H 2 O 8 of 36 NH 4+ + OH– © Boardworks Ltd 2009
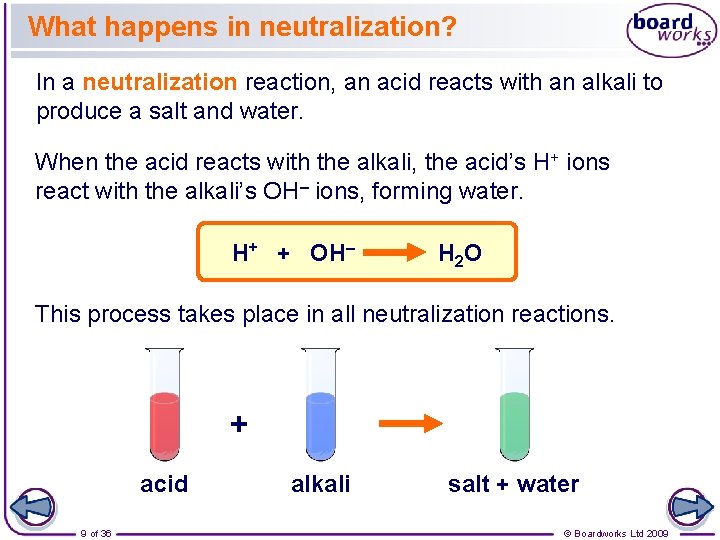
What happens in neutralization? In a neutralization reaction, an acid reacts with an alkali to produce a salt and water. When the acid reacts with the alkali, the acid’s H+ ions react with the alkali’s OH– ions, forming water. H+ + OH– H 2 O This process takes place in all neutralization reactions. + acid 9 of 36 alkali salt + water © Boardworks Ltd 2009
The history of acids 10 of 36 © Boardworks Ltd 2009

Theories about acids and bases When the 25 -year-old Svante Arrhenius submitted his dissertation to Uppsala University in 1884, he was disappointed to receive only a fourth-class degree. His theories relied on the idea that compounds disassociate in water to form ions. This was a new idea that had not been proposed before. In contrast Brønsted and Lowry’s ideas were immediately accepted and embraced by other scientists. Their theory was based on Arrhenius’s ideas. Both were among many respected scientists working on acids and bases at the time. Why do you think the scientific community responded differently to the two theories? 11 of 36 © Boardworks Ltd 2009

Acids and alkalis – true or false? 12 of 36 © Boardworks Ltd 2009

13 of 36 © Boardworks Ltd 2009

Strong acids Acids can be strong or weak. Strong acids dissociate fully into solution, so that all their H+ ions are released into the mixture. HA H+ + A– Hydrochloric acid is a typical strong acid, so the dissociation reaction is complete: HCl 14 of 36 H+ + Cl– © Boardworks Ltd 2009

Weak acids do not dissociate fully, some of their H+ ions stay attached to the acid molecule. The dissociation of a weak acid in water is a reversible reaction: HA ⇌ H+ + A– Ethanoic acid is a typical weak acid, with its ions in dynamic equilibrium with the un-dissociated acid. The reaction is moving both directions at the same rate. CH 3 COOH 15 of 36 ⇌ H+ + CH 3 COO– © Boardworks Ltd 2009

Strong or weak? 16 of 36 © Boardworks Ltd 2009

Sulfuric acid (H 2 SO 4) contains two hydrogen ions per sulphate ion. Both hydrogen ions can dissociate in solution. It is therefore called a diprotic acid. In general the first H+ ion of a diprotic acid will dissociate more readily than the second. H 2 SO 4 is a strong acid, so it fully dissociates to release its first H+ ion: H+ + HSO 4– H 2 SO 4 However, HSO 4– is a weak acid, so it only dissociates partially to release the second H+ ion: HSO 4– 17 of 36 ⇌ H+ + SO 42– © Boardworks Ltd 2009
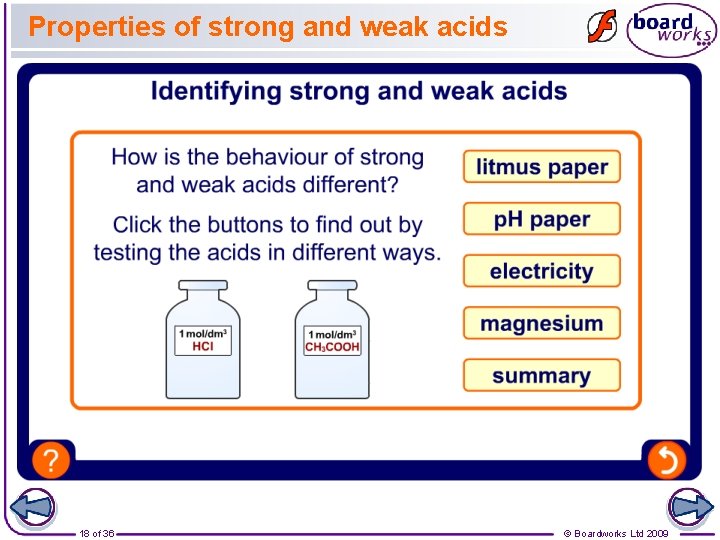
Properties of strong and weak acids 18 of 36 © Boardworks Ltd 2009

p. H of strong and weak acids p. H is a measure of the number of H+ ions in solution, with a lower p. H meaning more H+ ions. Because strong acids dissociate fully in solution, they contain more H+ ions per molecule of acid, producing a lower p. H. Compared to weak acids of the same concentration, strong acids: l have lower p. H values l are better conductors of electricity l react more quickly. high H+ 19 of 36 low H+ © Boardworks Ltd 2009

p. H or concentration? 20 of 36 © Boardworks Ltd 2009

Properties of strong and weak alkalis Alkalis can be classified as strong and weak in the same way as acids. A strong alkali, such as sodium hydroxide, fully dissociates in solution. Na+ + OH– Na. OH A weak alkali, such as ammonia, does not fully dissociate, and some of the OH– ions are not released into solution. NH 3 + H 2 O ⇌ NH 4+ + OH– Comparable levels of ion dissociation mean that strong and weak alkalis have similar properties to strong and weak acids. 21 of 36 © Boardworks Ltd 2009
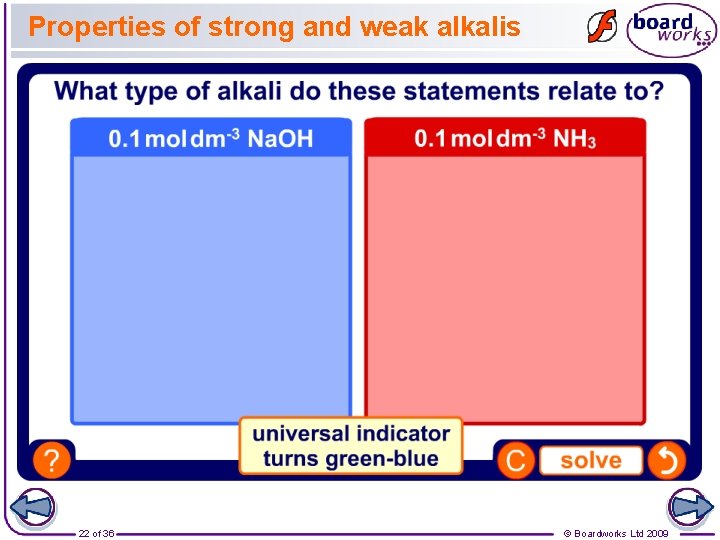
Properties of strong and weak alkalis 22 of 36 © Boardworks Ltd 2009

Strong and weak acids and alkalis 23 of 36 © Boardworks Ltd 2009

24 of 36 © Boardworks Ltd 2009
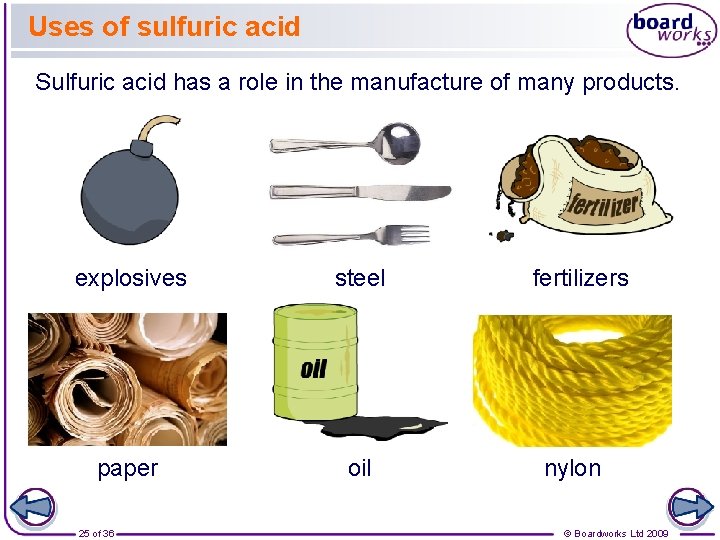
Uses of sulfuric acid Sulfuric acid has a role in the manufacture of many products. explosives steel paper oil 25 of 36 fertilizers nylon © Boardworks Ltd 2009

Dangers of sulfuric acid Concentrated sulfuric acid is a very powerful dehydrating agent. Dehydrating agents remove water from a compound, in a vigorous exothermic reaction. An example is the reaction of sulfuric acid with glucose. conc. H 2 SO 4 glucose carbon + water The sugar is rapidly broken down, producing an expanding mass of steaming carbon. Sulfuric acid will remove water from your skin, leading to bad burns. 26 of 36 © Boardworks Ltd 2009
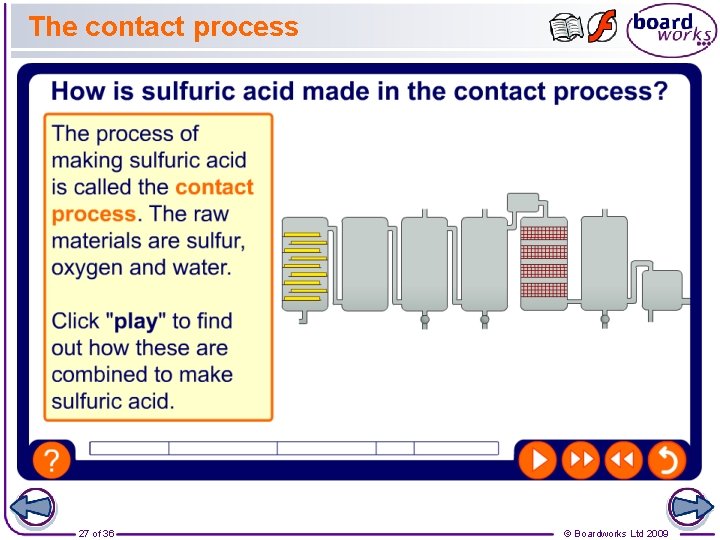
The contact process 27 of 36 © Boardworks Ltd 2009

Making sulfuric acid 28 of 36 © Boardworks Ltd 2009

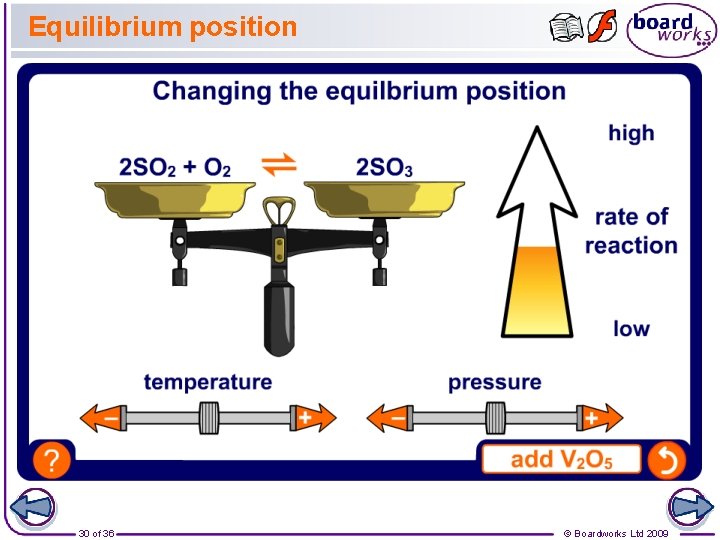
Conditions for the contact process The contact process involves a reversible reaction which reaches an equilibrium: 2 SO 2 + O 2 ⇌ 2 SO 3 This reaction is exothermic in the forward direction. What conditions would you suggest to produce the highest and most economical yield of sulfur trioxide? Remember: Whenever a change is made to a reversible reaction in dynamic equilibrium, the equilibrium position will shift to try and oppose that change. 29 of 36 © Boardworks Ltd 2009
Equilibrium position 30 of 36 © Boardworks Ltd 2009

Compromise conditions The conditions used during this stage of the contact process are: 2 SO 2 + O 2 ⇌ 2 SO 3 l 400 to 450 °C l 10 atmospheres l vanadium (V) oxide (V 2 O 5) catalyst These provide a compromise in which the cost of making the sulfuric acid is balanced by the yield and speed of production. What costs are involved in the manufacture of sulfuric acid? 31 of 36 l raw materials l energy l equipment l wages © Boardworks Ltd 2009

Making sulfuric acid 32 of 36 © Boardworks Ltd 2009

33 of 36 © Boardworks Ltd 2009

Glossary 34 of 36 © Boardworks Ltd 2009
Anagrams 35 of 36 © Boardworks Ltd 2009

Multiple-choice quiz 36 of 36 © Boardworks Ltd 2009
- Slides: 36