1 of 36 Boardworks Ltd 2008 2 of
1 of 36 © Boardworks Ltd 2008
2 of 36 © Boardworks Ltd 2008
Odd-one-out 3 of 36 © Boardworks Ltd 2008
Combining elements There are many different materials in the world, but only around 100 elements. These elements can be combined in many different ways to make thousands of compounds. 4 of 36 © Boardworks Ltd 2008
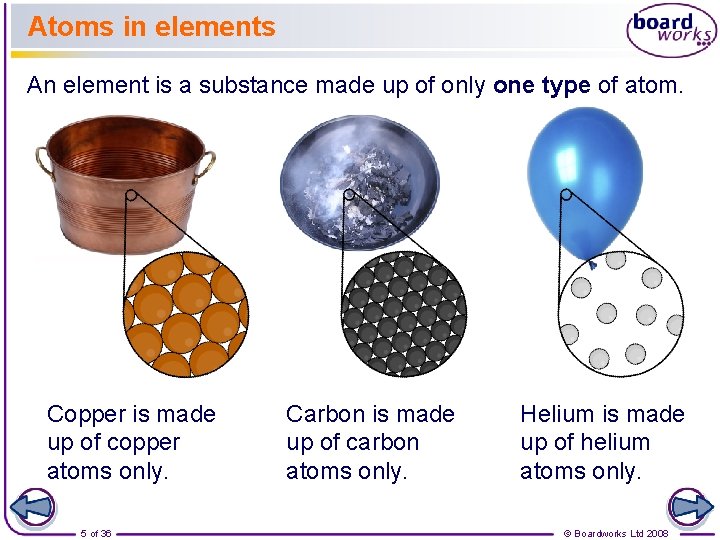
Atoms in elements An element is a substance made up of only one type of atom. Copper is made up of copper atoms only. 5 of 36 Carbon is made up of carbon atoms only. Helium is made up of helium atoms only. © Boardworks Ltd 2008

Molecules of elements The atoms in some elements join together to make molecules. One of the elements that exists like this is hydrogen. Each hydrogen atom joins up with one other atom to make a hydrogen molecule. = hydrogen atom = hydrogen molecule The formula of a hydrogen molecule is H 2. Other elements such as nitrogen, oxygen, and chlorine also exist as molecules. What are the formulae of these elements? 6 of 36 © Boardworks Ltd 2008
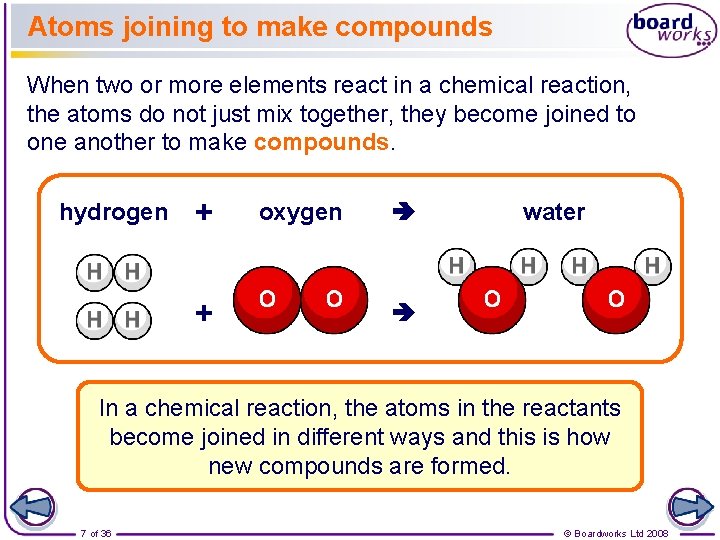
Atoms joining to make compounds When two or more elements react in a chemical reaction, the atoms do not just mix together, they become joined to one another to make compounds. hydrogen + + oxygen water In a chemical reaction, the atoms in the reactants become joined in different ways and this is how new compounds are formed. 7 of 36 © Boardworks Ltd 2008
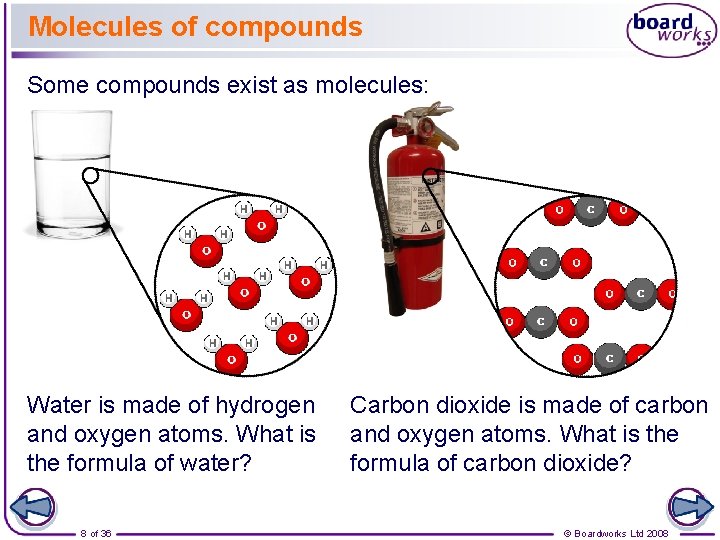
Molecules of compounds Some compounds exist as molecules: Water is made of hydrogen and oxygen atoms. What is the formula of water? 8 of 36 Carbon dioxide is made of carbon and oxygen atoms. What is the formula of carbon dioxide? © Boardworks Ltd 2008

Element or compound? 9 of 36 © Boardworks Ltd 2008
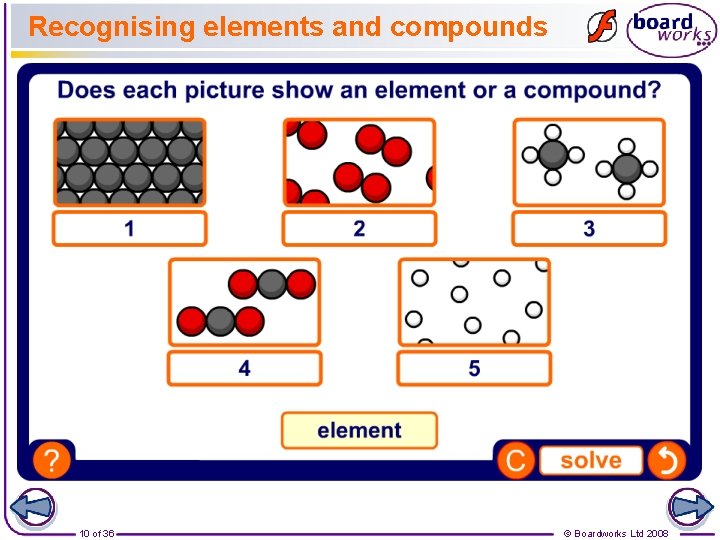
Recognising elements and compounds 10 of 36 © Boardworks Ltd 2008

A compound is not a mixture The molecules in a mixture of hydrogen gas and oxygen gas look like this: But the molecules in water look like this… …because water is a compound, made when hydrogen and oxygen react and their atoms become chemically joined to each other. 11 of 36 © Boardworks Ltd 2008
12 of 36 © Boardworks Ltd 2008

Properties of elements and compounds Why is it safe to put sodium chloride on fish and chips… …but not safe to use sodium and chlorine? 13 of 36 © Boardworks Ltd 2008

Properties of a compound Compounds have very different properties to the elements from which they are made. This is because the atoms are joined together differently. carbon (element) 14 of 36 + oxygen (element) carbon dioxide (compound) © Boardworks Ltd 2008
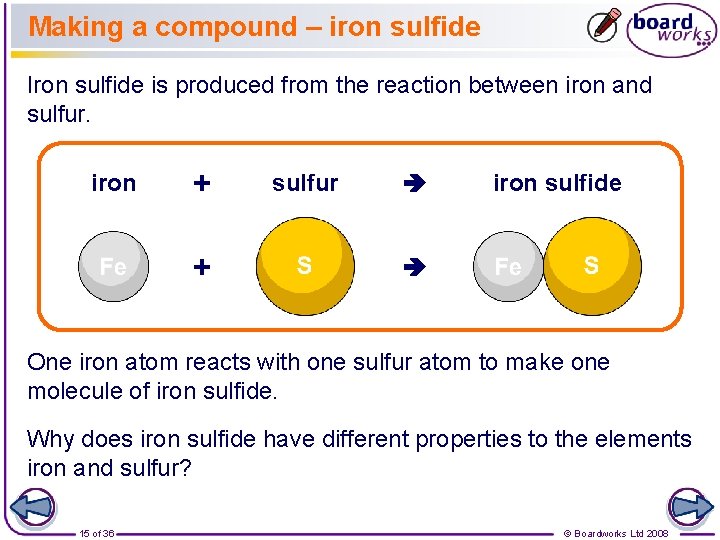
Making a compound – iron sulfide Iron sulfide is produced from the reaction between iron and sulfur. iron + + sulfur iron sulfide One iron atom reacts with one sulfur atom to make one molecule of iron sulfide. Why does iron sulfide have different properties to the elements iron and sulfur? 15 of 36 © Boardworks Ltd 2008
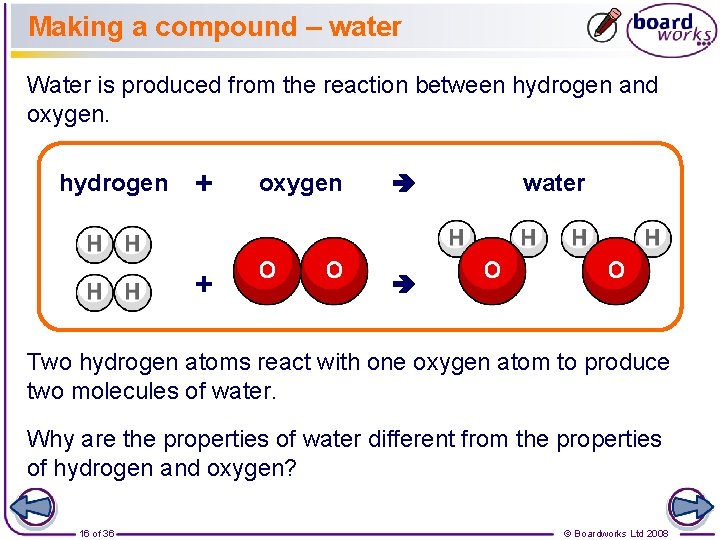
Making a compound – water Water is produced from the reaction between hydrogen and oxygen. hydrogen + + oxygen water Two hydrogen atoms react with one oxygen atom to produce two molecules of water. Why are the properties of water different from the properties of hydrogen and oxygen? 16 of 36 © Boardworks Ltd 2008

Making a compound – magnesium oxide Magnesium oxide is produced from the reaction between magnesium and oxygen. magnesium + + oxygen magnesium oxide Two atoms of magnesium react with one molecule of oxygen to make two molecules of magnesium oxide. What are the differences in the properties of magnesium, oxygen and magnesium oxide? 17 of 36 © Boardworks Ltd 2008
18 of 36 © Boardworks Ltd 2008

Naming simple compounds To name simple compounds of metals and non-metals: 1. Write down the name of the metal. 2. Write down the name of the non-metal, changing the ending of the word to “-ide”. What is the name of the compound made when the following elements combine? l magnesium and oxygen magnesium oxide l sodium and chlorine sodium chloride l oxygen and iron oxide 19 of 36 © Boardworks Ltd 2008

Naming simple compounds What is the name of each compound formed by these metal and non-metal elements? element 1 element 2 compound iron (Fe) sulfur (S) iron sulfide (Fe. S) magnesium (Mg) nitrogen (N) magnesium nitride (Mg 3 N 2) sodium (Na) chlorine (Cl) sodium chloride (Na. Cl) tin (Sn) oxygen (O) tin oxide (Sn. O) aluminium (Al) bromine (Br) aluminium bromide (Al. Br 3) nickel (Ni) iodine (I) nickel iodide (Ni. I 2) zinc (Zn) sulfur (S) zinc sulfide (Zn. S) lithium (Li) nitrogen (N) lithium nitride (Li 3 N) 20 of 36 © Boardworks Ltd 2008

Naming compounds containing oxygen Many compounds contain more than two elements. For compounds containing two elements plus oxygen, the ending of the other non-metal usually changes to “-ate”. element 1 element 2 element 3 compound nickel sulfur oxygen nickel sulfate magnesium nitrogen oxygen magnesium nitrate sodium nitrogen oxygen sodium nitrate copper sulfur oxygen copper sulfate aluminium bromine oxygen aluminium bromate 21 of 36 © Boardworks Ltd 2008
22 of 36 © Boardworks Ltd 2008

Writing a word equation A word equation can be used to describe any chemical reaction. The steps for writing a word equation are: 1. On the right-hand side, put the name(s) of the reactant(s). If there are two or more reactants, link them with a + sign. 2. In the middle, draw an arrow ( ). 3. On the right-hand side, put the name(s) of the product(s). If there are two or more products, link them with a + sign. reactant 1 23 of 36 + reactant 2 product 1 + product 2 © Boardworks Ltd 2008

Completing word equations 24 of 36 © Boardworks Ltd 2008
25 of 36 © Boardworks Ltd 2008

Composition of compounds A compound always contains a particular amount of each element. It has a fixed composition. For example, water molecules always contain twice as much hydrogen as oxygen. two hydrogen atoms This is shown in its formula, H 2 O. What is the formula of carbon dioxide? How many of each atom does each carbon dioxide molecule contain? one oxygen atom 26 of 36 © Boardworks Ltd 2008

Match the names and the formulae 27 of 36 © Boardworks Ltd 2008

Writing a formula A formula uses the symbols of the elements in a compound. When there is more than one atom of an element, the number is always written after the symbol. carbon atoms = 1 carbon dioxide oxygen atoms = 2 hydrogen atoms = 2 water oxygen atoms =1 formula = CO 2 formula = H 2 O The formula shows the ratio of atoms in a compound. 28 of 36 © Boardworks Ltd 2008

What is the formula? What is the formula of each of these compounds? (remember when you write a formula, put the metal first) 1. Titanium oxide For every titanium atom there are two oxygen atoms. Formula = Ti. O 2 2. Lithium oxide For every two lithium atoms there is one oxygen atom. Formula = Li 2 O 3. Aluminium chloride For every aluminium atom there are three chlorine atoms. Formula = Al. Cl 3 29 of 36 © Boardworks Ltd 2008

What does a formula show? 30 of 36 © Boardworks Ltd 2008

What is the ratio of atoms? 31 of 36 © Boardworks Ltd 2008

Why do scientists use formulae? Elements and compounds have different names in different languages: For example, sodium chloride is called: l Cloruro sódico in Spanish l Chlorek sodu in Polish l Хлорид натрия in Russian. This can lead to problems when scientists from different countries try to explain what they are investigating. However, because formulae are the same in every language, scientists can communicate their ideas easily using these instead of names. 32 of 36 © Boardworks Ltd 2008
33 of 36 © Boardworks Ltd 2008

Glossary 34 of 36 © Boardworks Ltd 2008
Anagrams 35 of 36 © Boardworks Ltd 2008
Multiple-choice quiz 36 of 36 © Boardworks Ltd 2008
- Slides: 36