1 of 35 Boardworks Ltd 2008 2 of
1 of 35 © Boardworks Ltd 2008

2 of 35 © Boardworks Ltd 2008

What are three states of matter? At any given temperature, all substances exist in one of the three states of matter. solid 3 of 35 liquid gas © Boardworks Ltd 2008

What state is it? 4 of 35 © Boardworks Ltd 2008
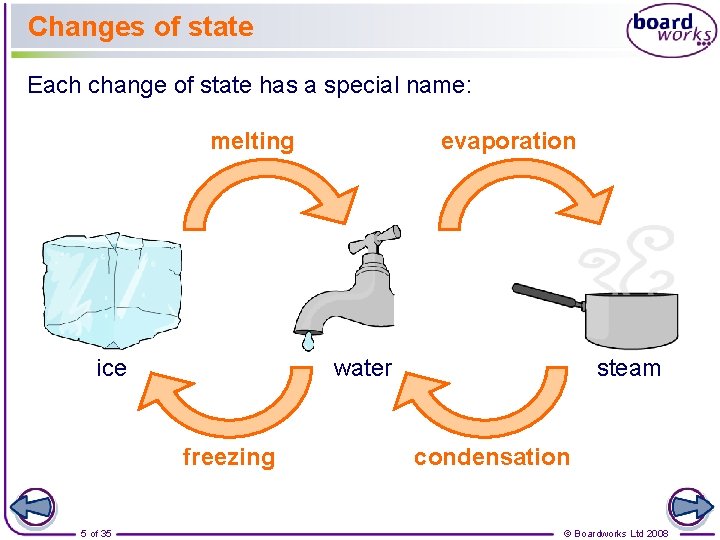
Changes of state Each change of state has a special name: melting ice water freezing 5 of 35 evaporation steam condensation © Boardworks Ltd 2008

Sublimation Some substances sublime. This means that they go straight from solid to gas without passing through the liquid state. sublimation solid gas Substances that sublime include: l iodine l carbon dioxide (dry ice) l plug-in air fresheners l ink in printers and photocopiers l comets as they approach the Sun. 6 of 35 © Boardworks Ltd 2008

Melting point and boiling point The temperature at which a substance changes from a solid to a liquid is called its melting point (or freezing point). What is the melting point of pure water? The temperature at which a substance changes from a liquid to a gas is called its boiling point. What is the boiling point of pure water? What state is water at 25 °C, 300 °C and 100 °C? 7 of 35 © Boardworks Ltd 2008
Temperature scales 8 of 35 © Boardworks Ltd 2008

What effect does salt have on ice? 9 of 35 © Boardworks Ltd 2008

Reducing the freezing point of water Salt reduces the freezing point of water. This means that the temperature has to be much colder than 0 °C for it to freeze. Can you explain why people put salt on roads and paths during icy weather? In what other situations would it be useful to reduce the freezing point of water? How about in making ice-cream? 10 of 35 © Boardworks Ltd 2008

11 of 35 © Boardworks Ltd 2008

Particles The differences between solids, liquids and gases can be explained by looking at the particles. l All substances are made up of particles. l The particles are attracted to each other. Some particles are attracted strongly to each other, and others weakly. l The particles move around. They are described as having kinetic energy. l The kinetic energy of the particles increases with temperature. 12 of 35 © Boardworks Ltd 2008

Particles and properties 13 of 35 © Boardworks Ltd 2008

Properties of solids, liquids and gases 14 of 35 © Boardworks Ltd 2008

How do particles move? 15 of 35 © Boardworks Ltd 2008

What are the properties of solids? Solids: l have a high density, as the particles are packed very closely together l cannot be compressed because there is very little empty space between particles l have a fixed shape because the particles are held tightly together l cannot diffuse because the particles are not able to move. 16 of 35 © Boardworks Ltd 2008

Cornflour paste: solid or liquid? 17 of 35 © Boardworks Ltd 2008

What are the properties of liquids? Liquids: l have a fairly high density because the particles are close together l cannot be compressed because there is very little empty space between particles l take up the shape of its container because the particles can move l can diffuse because the particles are able to change places. 18 of 35 © Boardworks Ltd 2008

What are the properties of gases? Gases: l have a low density because the particles are spaced far apart l can be compressed because there is space between particles l have no fixed shape because the particles move about rapidly in all directions l can diffuse because the particles are able to move in all directions. 19 of 35 © Boardworks Ltd 2008

Gas pressure 20 of 35 © Boardworks Ltd 2008

Solids, liquids and gases: which one? 21 of 35 © Boardworks Ltd 2008

22 of 35 © Boardworks Ltd 2008

How do smells spread out? 23 of 35 © Boardworks Ltd 2008

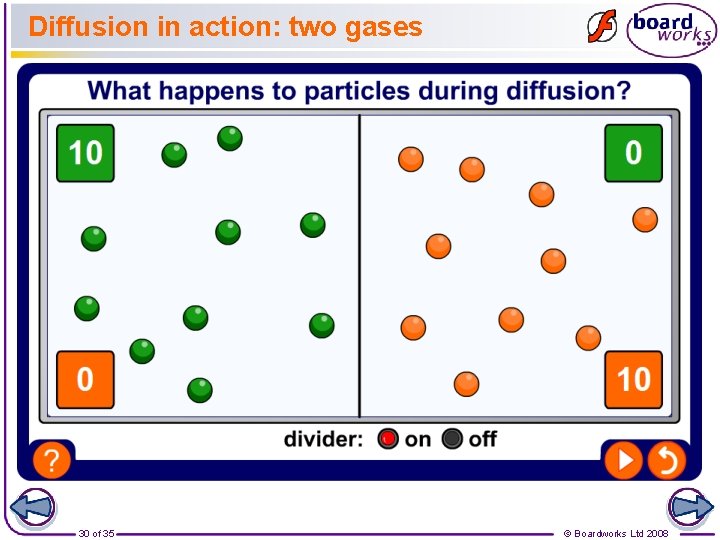
What is diffusion? Diffusion is the random movement of particles. When particles diffuse, they spread out and mix with other particles. For example, the smell of aftershave or perfume can be detected by people on the other side of a room because the perfume particles diffuse in the air. Can you think of some more everyday examples of diffusion? 24 of 35 © Boardworks Ltd 2008
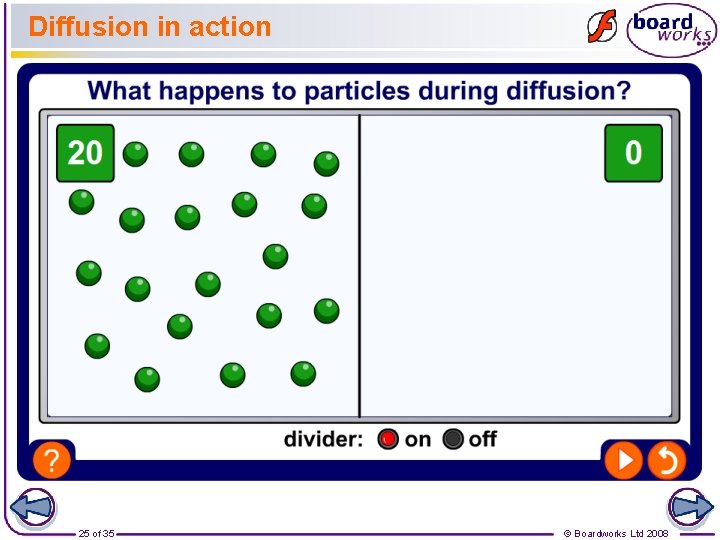
Diffusion in action 25 of 35 © Boardworks Ltd 2008

Diffusion and temperature 26 of 35 © Boardworks Ltd 2008

Diffusion in solids The particles in liquids and gases diffuse easily because they are free to move around. However, it is possible for solids to diffuse as well. Scientists have found that if they put two metals next to each other and leave them for a long time, eventually a few particles of one metal will diffuse into the other. This is because the particles in a solid vibrate, and some of them have enough energy to move around a little bit as well. Eventually, a few particles move around enough to diffuse into the other metal. 27 of 35 © Boardworks Ltd 2008

Diffusion in liquids and gases Which will diffuse faster, a gas or a liquid? The particles of a gas will diffuse faster. Why? The gas particles are moving around faster than the particles in a liquid because they have more energy. This means that the gas particles will spread out more quickly. 28 of 35 © Boardworks Ltd 2008

Rate of diffusion 29 of 35 © Boardworks Ltd 2008
Diffusion in action: two gases 30 of 35 © Boardworks Ltd 2008

Summary: diffusion 31 of 35 © Boardworks Ltd 2008

32 of 35 © Boardworks Ltd 2008

Glossary 33 of 35 © Boardworks Ltd 2008

Anagrams 34 of 35 © Boardworks Ltd 2008

Multiple-choice quiz 35 of 35 © Boardworks Ltd 2008
- Slides: 35