1 of 34 Boardworks Ltd 2009 2 of
1 of 34 © Boardworks Ltd 2009

2 of 34 © Boardworks Ltd 2009

Energy in food Energy is measured in joules. One joule is the energy needed to lift a weight of one newton through one metre. A small apple has a weight of about 1 N. How many apples would you need to lift through one metre to use up all the energy in this chocolate bar? Energy (k. J) 1075 Energy (kcal) 255 Protein 3. 7 g Carbohydrate 27. 9 g Fat 14. 6 g Fibre 0. 3 g Sodium 0. 04 g 3 of 34 © Boardworks Ltd 2009

Energy in food is usually measured in calories. This comes from calor, the Latin word for heat. 1 calorie = 4. 2 joules 1 kilocalorie (kcal) = 1000 calories How many joules of energy does this cheeseburger contain? Energy (k. J) ? Energy (kcal) 295 Protein 16 g Carbohydrate 31 g Fat 12 g Fibre 2 g Sodium 1. 5 g 4 of 34 © Boardworks Ltd 2009
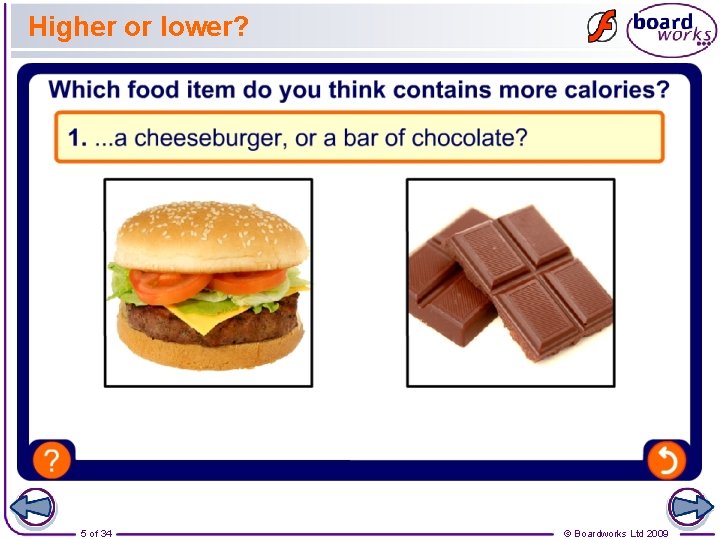
Higher or lower? 5 of 34 © Boardworks Ltd 2009
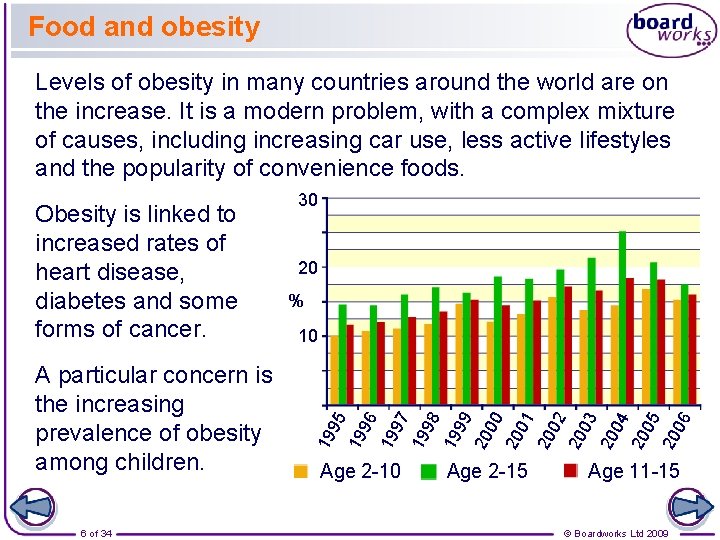
Food and obesity Levels of obesity in many countries around the world are on the increase. It is a modern problem, with a complex mixture of causes, including increasing car use, less active lifestyles and the popularity of convenience foods. 6 of 34 % 19 96 19 97 19 98 19 99 20 00 20 01 20 02 20 03 20 04 20 05 20 06 10 95 A particular concern is the increasing prevalence of obesity among children. 20 19 Obesity is linked to increased rates of heart disease, diabetes and some forms of cancer. 30 Age 2 -15 Age 11 -15 © Boardworks Ltd 2009

The obesity debate 7 of 34 © Boardworks Ltd 2009

Treatments for obesity The recommended treatment for obesity is a reduction in calorie intake and increased exercise. Medical treatments have been developed that work alongside these measures. The treatments act in different ways: l Blocking body enzymes which digest fat. The undigested fat is not absorbed by the body. l Affecting chemicals in the brain which control appetite. Gastric banding is a surgical procedure in which a band is fitted around the upper part of the stomach. It reduces the amount of food that can be held in the stomach. 8 of 34 © Boardworks Ltd 2009

The obesity debate 9 of 34 © Boardworks Ltd 2009
10 of 34 © Boardworks Ltd 2009

Calorimetry The energy content of food is measured using a technique called calorimetry. Calorimetry involves burning a known mass of food and measuring the energy released. This is done by transferring the energy to a measured volume of water and working out the temperature change. Knowing the temperature change and the specific heat capacity of water, it is possible to calculate the energy released by the burning food. 11 of 34 © Boardworks Ltd 2009

Using calorimetry The energy content of food is found using this equation: Q = mc. T l Q = energy given out by burning food (J) l m = mass of water heated by burning food (g) l c = specific heat capacity of water = 4. 18 Jg-1°C-1 l T = change in temperature of the water (°C) Specific heat capacity is a measure of how much energy it takes to heat up a given mass of a substance. It takes 4. 18 J of energy to heat up 1 cm 3 of water by 1 °C. 12 of 34 © Boardworks Ltd 2009

Performing a calorimetry experiment 13 of 34 © Boardworks Ltd 2009

Finding the energy content of food The 1. 5 g crisp that was burned gave out 33 k. J of energy. How much energy was given out per gram of crisp? The mass of crisp that burned was 1. 5 g, so to find the amount of energy per gram, divide the energy given out by the mass that burned: Energy per gram = 33 / 1. 5 = 22 k. J/g One bag of crisps has a mass of 25 g. How much energy does one bag of crisps contain? How likely do you think it would be to obtain this answer experimentally? 14 of 34 © Boardworks Ltd 2009
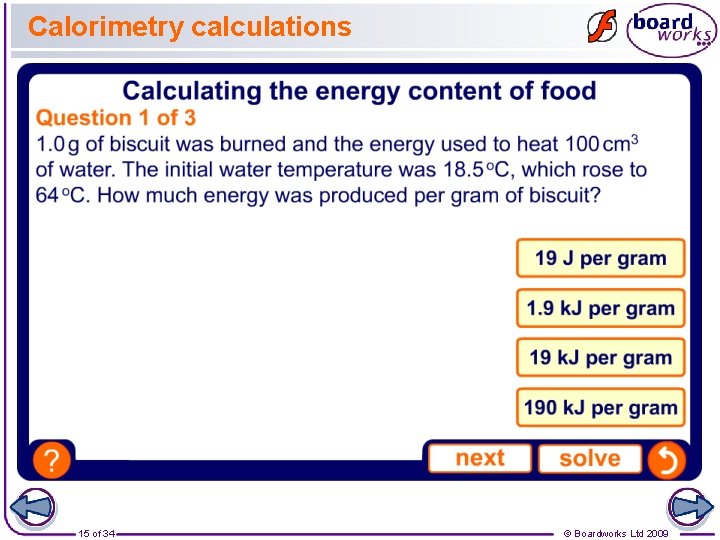
Calorimetry calculations 15 of 34 © Boardworks Ltd 2009

Errors in calorimetry Comparing the actual amounts of energy in food with the values found in calorimetry experiments, the experimental values are often a lot lower than the actual values. Why is this? Calorimetry experiments are not very accurate. A lot of the energy released by the burning food is not transferred into the water. It is simply lost into the atmosphere. How could the experiment be made more accurate? 16 of 34 © Boardworks Ltd 2009
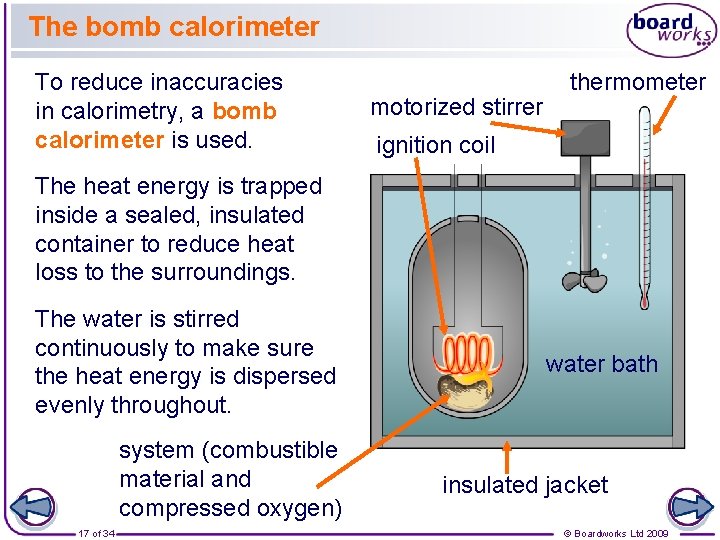
The bomb calorimeter To reduce inaccuracies in calorimetry, a bomb calorimeter is used. motorized stirrer thermometer ignition coil The heat energy is trapped inside a sealed, insulated container to reduce heat loss to the surroundings. The water is stirred continuously to make sure the heat energy is dispersed evenly throughout. system (combustible material and compressed oxygen) 17 of 34 water bath insulated jacket © Boardworks Ltd 2009

18 of 34 © Boardworks Ltd 2009

Fuels A fuel is a substance that is burned to release useful energy. The amount of energy in a fuel can be found in the same way as the amount of energy in food. Is burning a fuel endothermic or exothermic? 19 of 34 © Boardworks Ltd 2009

Energy content of fuels 20 of 34 © Boardworks Ltd 2009
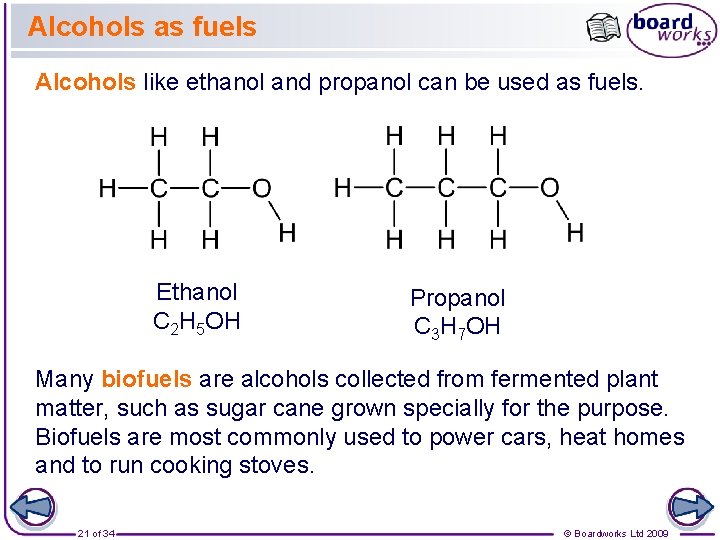
Alcohols as fuels Alcohols like ethanol and propanol can be used as fuels. Ethanol C 2 H 5 OH Propanol C 3 H 7 OH Many biofuels are alcohols collected from fermented plant matter, such as sugar cane grown specially for the purpose. Biofuels are most commonly used to power cars, heat homes and to run cooking stoves. 21 of 34 © Boardworks Ltd 2009

Alcohols as fuels Alcohols have a variety of uses as fuels: biofuels: ethanol and butanol cooking: ethanol methylated spirits burners: methanol 22 of 34 © Boardworks Ltd 2009
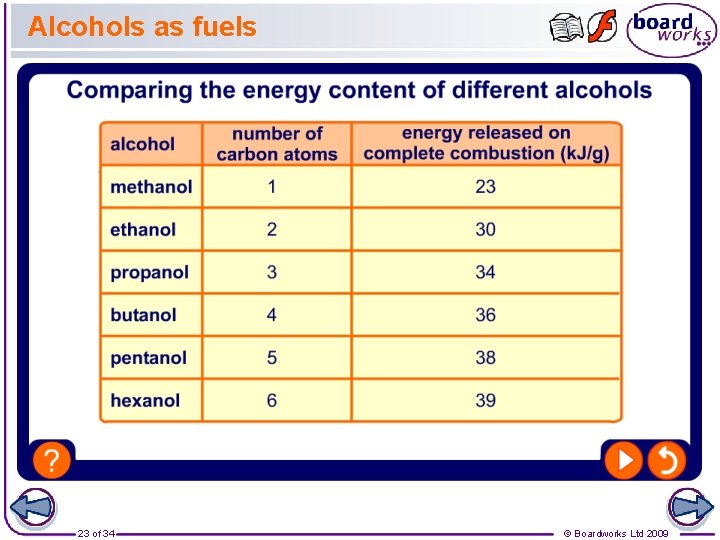
Alcohols as fuels 23 of 34 © Boardworks Ltd 2009

Hydrogen fuel cells produce electricity through the reaction of hydrogen with oxygen. The reaction which takes place in a hydrogen fuel cell is: 2 H 2 + O 2 2 H 2 O The only waste product is water vapour. Hydrogen fuel cells do not produce other pollutants like carbon dioxide, sulfur dioxide or carbon particles. However, water vapour is itself a greenhouse gas and could contribute to global warming. 24 of 34 © Boardworks Ltd 2009

How does a hydrogen fuel cell work? 25 of 34 © Boardworks Ltd 2009

Calculating the energy Bond energies can be used to calculate the amount of energy given out by the reaction in a hydrogen fuel cell. Energy is taken in to break bonds, and released when bonds are created. Bond Energy (k. J) H–H 432 O=O 498 O–H 463 energy for bond-breaking energy from bond-making = 2 H–H + O=O = 4 H–O = 2 × 432 + 498 = 4 × 463 = 1362 k. J = 1852 k. J total energy change = energy in – energy out = 1362 k. J – 1852 k. J = – 490 k. J 26 of 34 © Boardworks Ltd 2009
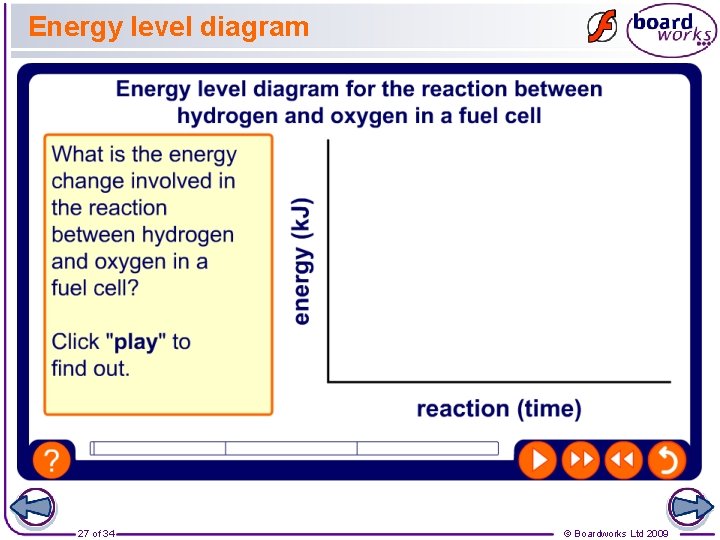
Energy level diagram 27 of 34 © Boardworks Ltd 2009

Where does the hydrogen come from? At the moment the hydrogen for fuel cells is extracted from crude oil. It may be possible to use solar power to extract hydrogen from water, but this method is still under development. What are the advantages and disadvantages of each method of making hydrogen? 28 of 34 © Boardworks Ltd 2009

Advantages of fuel cells The big advantage of hydrogen fuel cells is that the only emission they produce is water vapour. What are the other advantages? l Hydrogen has a low density, making vehicles light and efficient. l Hydrogen gives out more energy per gram than conventional fuels. What are some of the disadvantages of fuel cell technology? l Production of hydrogen from fossil fuels produces carbon dioxide – a greenhouse gas. l Hydrogen is highly flammable, making it risky to store. l Hydrogen is not cheap! 29 of 34 © Boardworks Ltd 2009

Fuel cells activity 30 of 34 © Boardworks Ltd 2009
31 of 34 © Boardworks Ltd 2009

Glossary 32 of 34 © Boardworks Ltd 2009
Anagrams 33 of 34 © Boardworks Ltd 2009
Multiple-choice quiz 34 of 34 © Boardworks Ltd 2009
- Slides: 34