1 of 33 Boardworks Ltd 2007 2 of
1 of 33 © Boardworks Ltd 2007

2 of 33 © Boardworks Ltd 2007

How many different substances? How many different substances can you think of? There are millions of different substances! What are they all made of? 3 of 33 © Boardworks Ltd 2007

All substances are made of atoms All substances are made of very tiny particles called atoms. Many substances are made up of different types of atoms. hydrogen and oxygen atoms carbon and hydrogen atoms iron, aluminium, silicon, oxygen and boron atoms 4 of 33 carbon, nitrogen, hydrogen, oxygen and sulphur atoms © Boardworks Ltd 2007

What is an element? All substances are made of very tiny particles called atoms. There about one hundred substances that are made up of just one type of atom. These are the elements. copper carbon helium The elements are the simplest substances in the universe. The elements are the building blocks of all other substances. 5 of 33 © Boardworks Ltd 2007
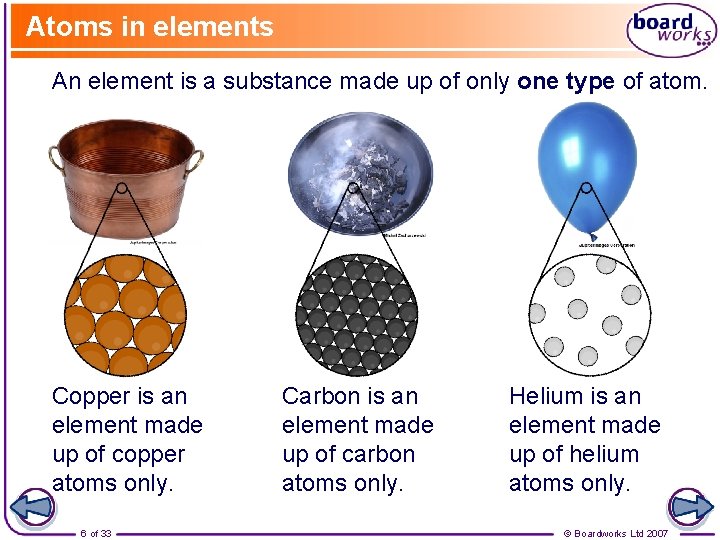
Atoms in elements An element is a substance made up of only one type of atom. Copper is an element made up of copper atoms only. 6 of 33 Carbon is an element made up of carbon atoms only. Helium is an element made up of helium atoms only. © Boardworks Ltd 2007
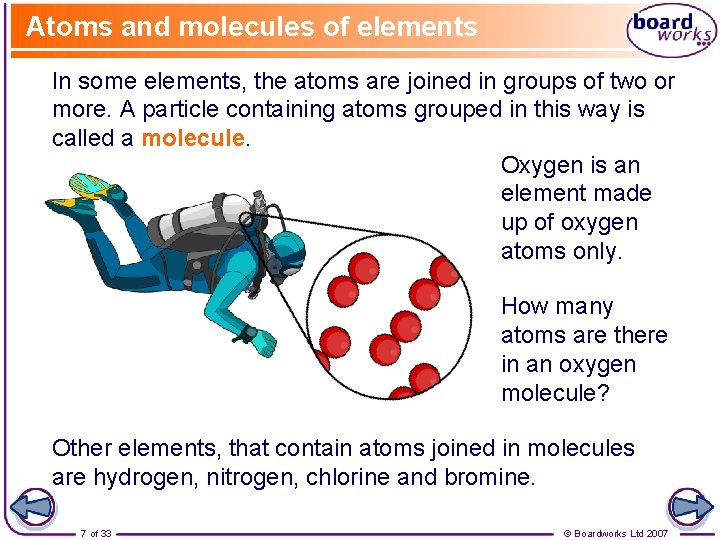
Atoms and molecules of elements In some elements, the atoms are joined in groups of two or more. A particle containing atoms grouped in this way is called a molecule. Oxygen is an element made up of oxygen atoms only. How many atoms are there in an oxygen molecule? Other elements, that contain atoms joined in molecules are hydrogen, nitrogen, chlorine and bromine. 7 of 33 © Boardworks Ltd 2007

8 of 33 © Boardworks Ltd 2007

Symbols for elements A standard set of symbols is used to represent elements. The symbol for many common elements uses just the first letter of the name, for example H = hydrogen. Other elements have the first two letters, for example Li = lithium. However, some of the symbols are not always as you might expect, for example Pb = lead. Two important rules should be followed when writing the symbols of elements so that there is no confusion. l The first letter of an element’s symbol is always a capital letter, e. g. N (not n) for nitrogen. l If there are two letters in the element’s symbol, the second letter is always a small letter, e. g. Co (not CO) for cobalt. 9 of 33 © Boardworks Ltd 2007
Chemical symbols game 10 of 33 © Boardworks Ltd 2007

Spelling with symbols activity 11 of 33 © Boardworks Ltd 2007

Spelling with symbols Write down the symbols for each element listed and use these to spell out a word that matches the clue. 1. Board game: carbon, helium, two sulphurs CHe. SS 2. Relative: sulphur, oxygen, nitrogen SON 3. Fuel: carbon, oxygen, aluminium COAl 4. Group of fish: sulphur, hydrogen, oxygen, aluminium SHOAl 5. For the rubbish: boron, iodine, nitrogen. BIN 12 of 33 © Boardworks Ltd 2007
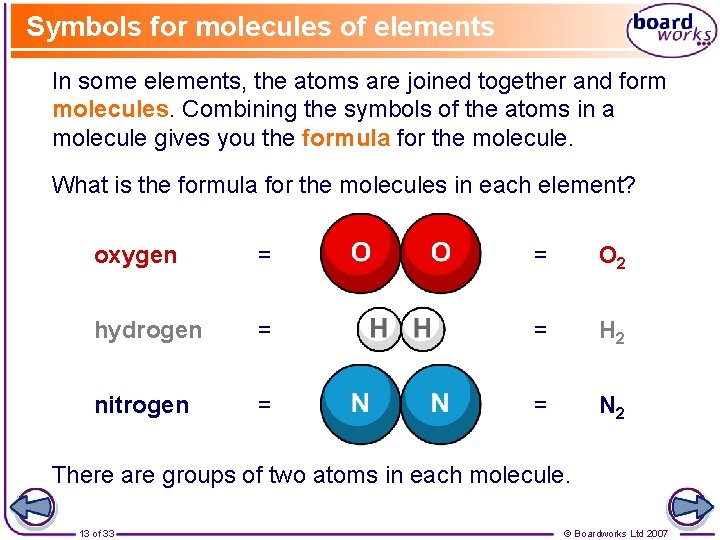
Symbols for molecules of elements In some elements, the atoms are joined together and form molecules. Combining the symbols of the atoms in a molecule gives you the formula for the molecule. What is the formula for the molecules in each element? oxygen = = O 2 hydrogen = = H 2 nitrogen = = N 2 There are groups of two atoms in each molecule. 13 of 33 © Boardworks Ltd 2007

14 of 33 © Boardworks Ltd 2007

Arranging elements 15 of 33 © Boardworks Ltd 2007

Metal or non-metal? 16 of 33 © Boardworks Ltd 2007
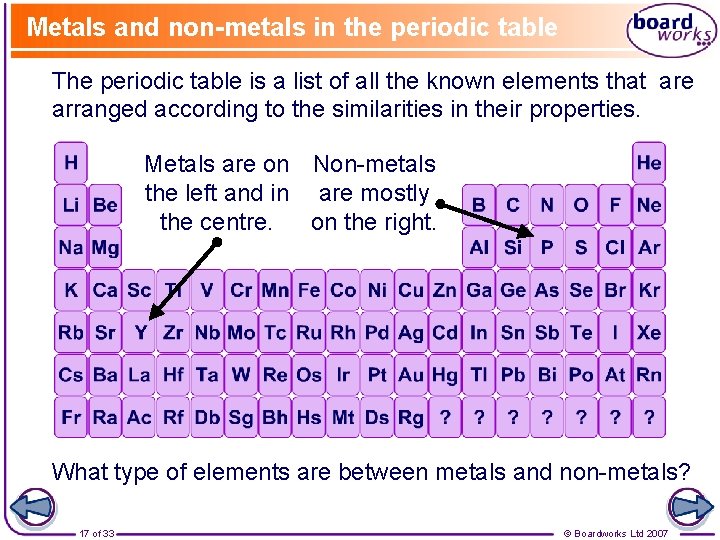
Metals and non-metals in the periodic table The periodic table is a list of all the known elements that are arranged according to the similarities in their properties. Metals are on Non-metals the left and in are mostly the centre. on the right. What type of elements are between metals and non-metals? 17 of 33 © Boardworks Ltd 2007
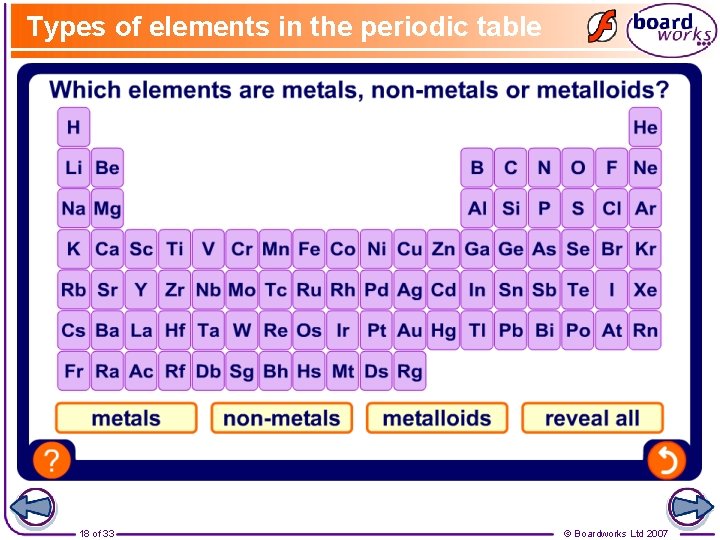
Types of elements in the periodic table 18 of 33 © Boardworks Ltd 2007

Metal, non-metal or metalloid? Metals are on the left and in the centre of the periodic table. Non-metals are located mostly on the right. Metalloids sometimes behave like metals and sometimes like non-metals. Metalloids are located between metals and non-metals in the periodic table. Silicon is an example of a metalloid. 19 of 33 © Boardworks Ltd 2007

20 of 33 © Boardworks Ltd 2007
Combining elements How is it possible to have so many different materials from a limited number of elements? 21 of 33 © Boardworks Ltd 2007

Combining elements A small number of elements can be combined in many different ways to make a huge number of different compounds. To help understand this, just think about cheese! l How are so many different types of cheese produced? l What is the key ingredient in all cheeses? Milk is combined with a small number of ingredients under different conditions to make a huge number of different cheeses. 22 of 33 © Boardworks Ltd 2007
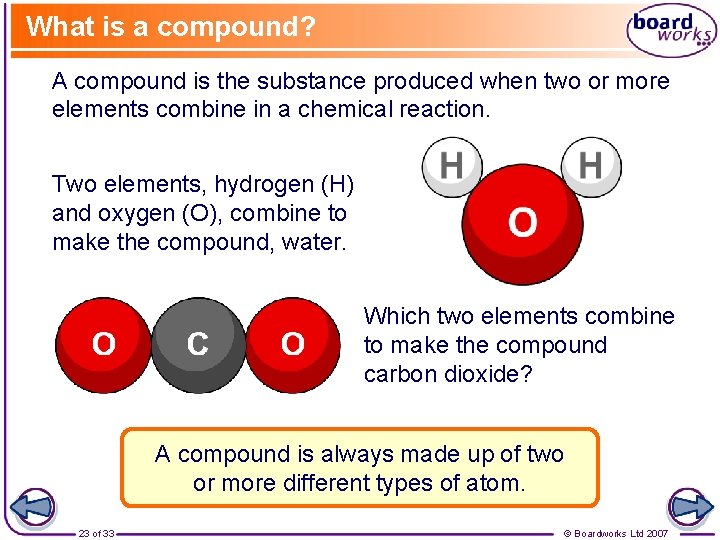
What is a compound? A compound is the substance produced when two or more elements combine in a chemical reaction. Two elements, hydrogen (H) and oxygen (O), combine to make the compound, water. Which two elements combine to make the compound carbon dioxide? A compound is always made up of two or more different types of atom. 23 of 33 © Boardworks Ltd 2007

Making a compound – carbon dioxide A compound has very different properties to the elements from which it is made. carbon (element) A black solid that can be used as a fuel. 24 of 33 + oxygen (element) A colourless gas that is essential for life. carbon dioxide (compound) A colourless gas that is used to put out fires. © Boardworks Ltd 2007
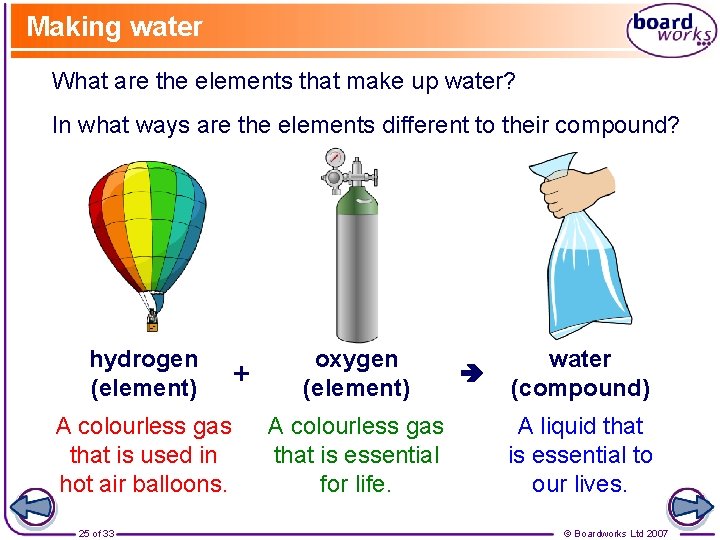
Making water What are the elements that make up water? In what ways are the elements different to their compound? hydrogen (element) A colourless gas that is used in hot air balloons. 25 of 33 + oxygen (element) A colourless gas that is essential for life. water (compound) A liquid that is essential to our lives. © Boardworks Ltd 2007
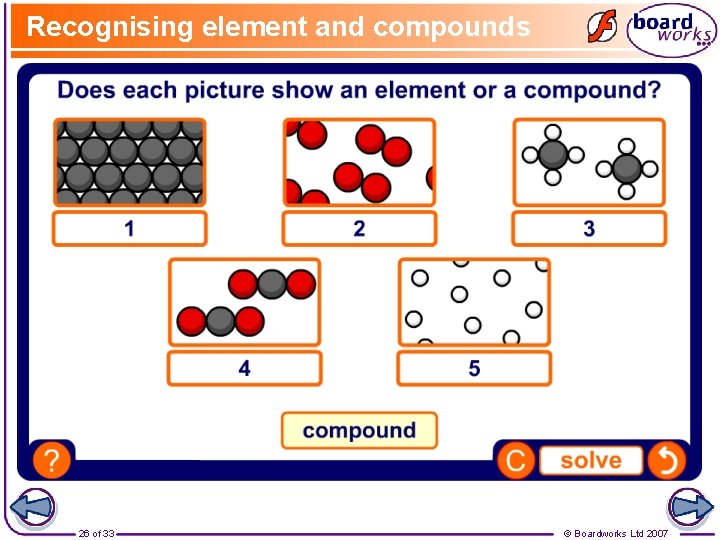
Recognising element and compounds 26 of 33 © Boardworks Ltd 2007

Element or compound? 27 of 33 © Boardworks Ltd 2007

Chemical reaction to make water In a chemical reaction new substances are formed. Elements combine with each other to make new substances called compounds. hydrogen + oxygen water The substances that combine in a reaction are the reactants. The new substances produced in a reaction are the products. The products have different properties to the reactants from which they are formed. Compounds can also combine with elements or other compounds to make new compounds. 28 of 33 © Boardworks Ltd 2007
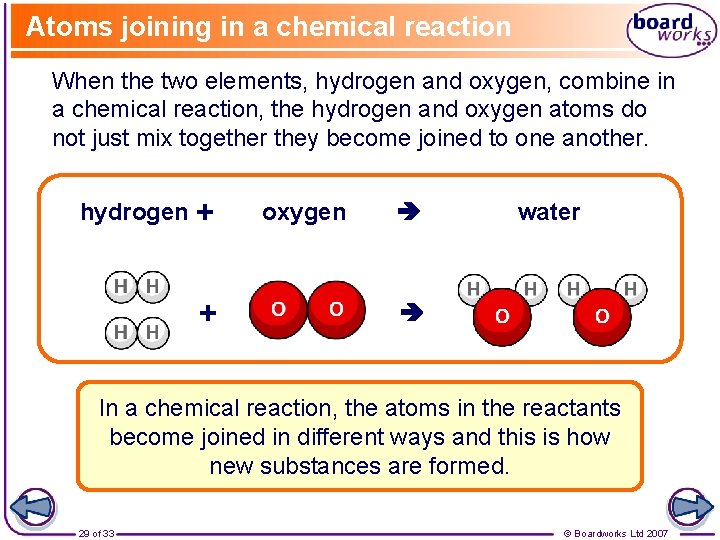
Atoms joining in a chemical reaction When the two elements, hydrogen and oxygen, combine in a chemical reaction, the hydrogen and oxygen atoms do not just mix together they become joined to one another. hydrogen + + oxygen water In a chemical reaction, the atoms in the reactants become joined in different ways and this is how new substances are formed. 29 of 33 © Boardworks Ltd 2007

30 of 33 © Boardworks Ltd 2007

Glossary atom – The smallest particle that can exist on its own. compound – Substance made up of two or more different elements that are chemically joined together. element – Substance made up of only one type of atom. formula – The symbols and numbers that represent the atoms in a substance. molecule – Two or more atoms that are chemically joined together. periodic table – Table in which all known elements are arranged based on their properties. symbol – One or two letters that represent an element. 31 of 33 © Boardworks Ltd 2007

Anagrams 32 of 33 © Boardworks Ltd 2007
Multiple-choice quiz 33 of 33 © Boardworks Ltd 2007
- Slides: 33