1 of 32 Boardworks Ltd 2007 2 of

1 of 32 © Boardworks Ltd 2007

2 of 32 © Boardworks Ltd 2007

Properties of elements and compounds Why is it safe to put sodium chloride on fish and chips… …but not safe to use sodium and chlorine? 3 of 32 © Boardworks Ltd 2007
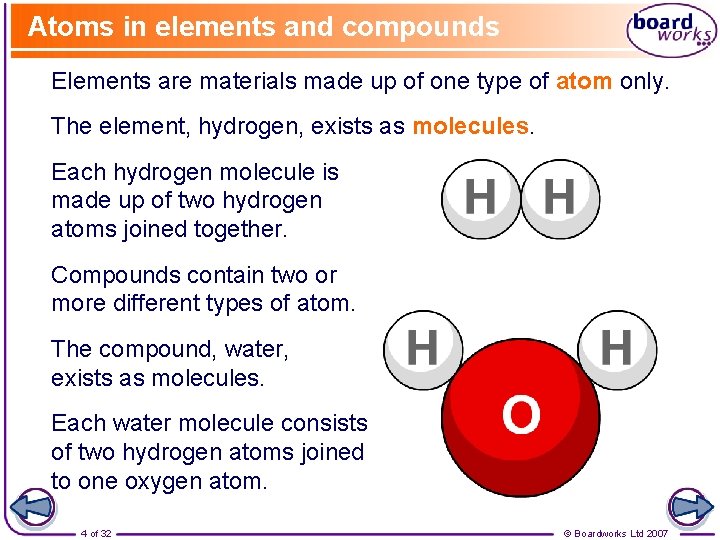
Atoms in elements and compounds Elements are materials made up of one type of atom only. The element, hydrogen, exists as molecules. Each hydrogen molecule is made up of two hydrogen atoms joined together. Compounds contain two or more different types of atom. The compound, water, exists as molecules. Each water molecule consists of two hydrogen atoms joined to one oxygen atom. 4 of 32 © Boardworks Ltd 2007

Element or compound? 5 of 32 © Boardworks Ltd 2007
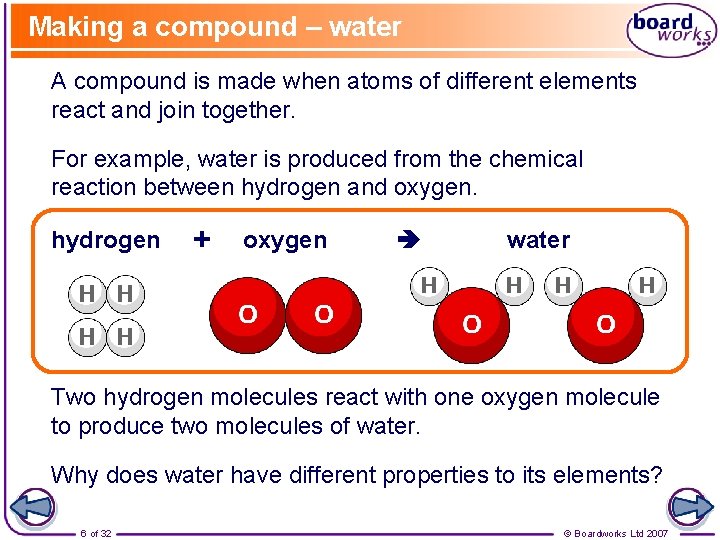
Making a compound – water A compound is made when atoms of different elements react and join together. For example, water is produced from the chemical reaction between hydrogen and oxygen. hydrogen + oxygen water Two hydrogen molecules react with one oxygen molecule to produce two molecules of water. Why does water have different properties to its elements? 6 of 32 © Boardworks Ltd 2007
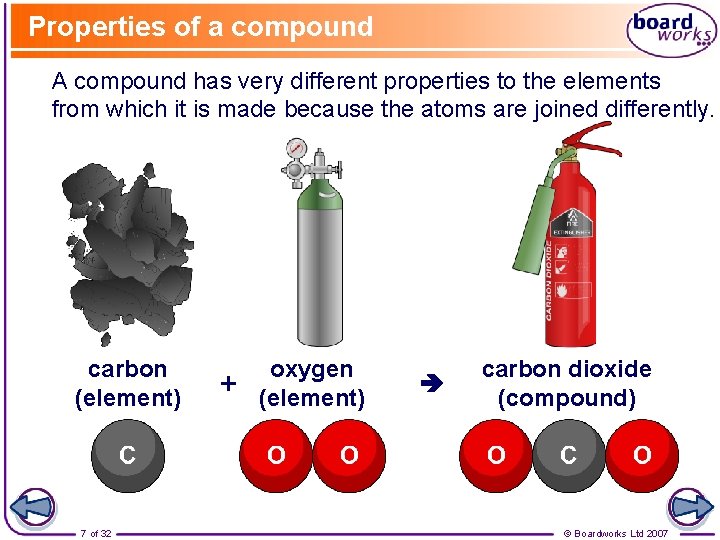
Properties of a compound A compound has very different properties to the elements from which it is made because the atoms are joined differently. carbon (element) 7 of 32 + oxygen (element) carbon dioxide (compound) © Boardworks Ltd 2007
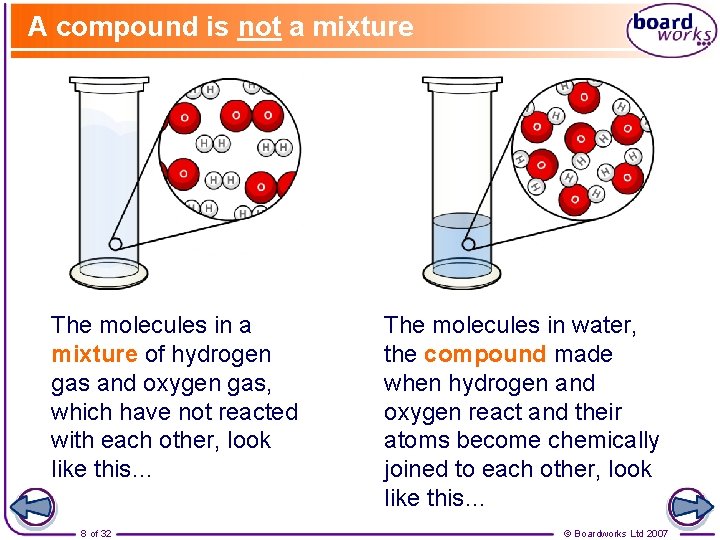
A compound is not a mixture The molecules in a mixture of hydrogen gas and oxygen gas, which have not reacted with each other, look like this… 8 of 32 The molecules in water, the compound made when hydrogen and oxygen react and their atoms become chemically joined to each other, look like this… © Boardworks Ltd 2007

9 of 32 © Boardworks Ltd 2007

Naming simple compounds To name simple compounds of metals and non-metals: 1. Write down the name of the metal. 2. Write down the name of the non-metal, changing the ending of the word to “-ide”. What is the name of the compound made when the following elements combine? l magnesium and oxygen magnesium oxide l sodium and chlorine sodium chloride l oxygen and iron oxide 10 of 32 © Boardworks Ltd 2007

Naming compounds What is the name of each compound formed by these metal and non-metal elements? Element 1 Element 2 Compound iron sulfur iron sulfide magnesium nitrogen magnesium nitride sodium chlorine sodium chloride tin oxygen tin oxide aluminium bromine nickel iodine aluminium bromide nickel iodide zinc sulfur zinc sulfide lithium nitrogen lithium nitride 11 of 32 © Boardworks Ltd 2007

Naming compounds containing oxygen Many compounds contain more than two elements. For compounds containing two elements plus oxygen, the ending of the other non-metal usually changes to “-ate”. Element 1 Element 2 Element 3 Compound nickel sulfur oxygen nickel sulfate magnesium nitrogen oxygen magnesium nitrate sodium nitrogen oxygen sodium nitrate copper sulfur oxygen copper sulfate aluminium bromine oxygen aluminium bromate 12 of 32 © Boardworks Ltd 2007

Composition of compounds A compound contains atoms from different elements that are chemically joined together. A compound always contains a particular amount of each element. It has a fixed composition. Compound names can get quite long and complicated, so the symbols of the elements are used as a shorthand. The symbols of the elements in a compound are combined to give the formula of the compound. What is the formula of carbon dioxide? 13 of 32 © Boardworks Ltd 2007

Writing a formula A formula uses the symbols of the elements in a compound. When there is more than one atom of each element, the number is always written after the symbol. carbon atoms = 1 carbon dioxide oxygen atoms = 2 hydrogen atoms = 2 water oxygen atoms =1 formula = CO 2 formula = H 2 O The formula shows the ratio of atoms in a compound. 14 of 32 © Boardworks Ltd 2007

What is the formula? What is the formula of each of these compounds? (In a formula, as when naming a compound, put the metal first. ) 1. Titanium oxide For every titanium atom there are two oxygen atoms. Formula = Ti. O 2 2. Lithium oxide For every two lithium atoms there is one oxygen atom. Formula = Li 2 O 3. Aluminium chloride For every aluminium atom there are three chlorine atoms. Formula = Al. Cl 3 15 of 32 © Boardworks Ltd 2007

What does a formula show? 16 of 32 © Boardworks Ltd 2007

What is the ratio of atoms? 17 of 32 © Boardworks Ltd 2007
18 of 32 © Boardworks Ltd 2007

Writing a word equation A word equation can be used to describe any chemical reaction, i. e. any process in which atoms become joined in different ways. The steps for writing a word equation are: 1. On the right-hand side, put the name(s) of the reactant(s). If there are two or more reactants, link them with a + sign. 2. In the middle, draw an arrow ( ). 3. On the right-hand side, put the name(s) of the product(s). If there are two or more products, link them with a + sign. reactant 1 19 of 32 + reactant 2 product 1 + product 2 © Boardworks Ltd 2007

What is the word equation? Lead reacts with oxygen to form lead oxide. What is the word equation for this reaction? lead + oxygen lead oxide Why is lead oxide so different to both lead and oxygen? The lead and oxygen don’t mix; they react to form lead oxide. This means that the lead and oxygen atoms in the product are joined differently to the atoms in the reactants. 20 of 32 © Boardworks Ltd 2007
21 of 32 © Boardworks Ltd 2007

What is a mixture? A mixture contains two or more substances that are mixed together, but have not reacted with each other. For example, sea water is a mixture of salts, water and other substances. A mixture is not the same as a compound: l The substances in a mixture are not chemically joined, and so it is usually possible to separate the ingredients (e. g. to get salt from sea water). l The proportions in a mixture are not fixed. l The properties of a mixture are often an ‘average’ of the properties of its ingredients (e. g. a mixture of black powder and white powder is grey). 22 of 32 © Boardworks Ltd 2007
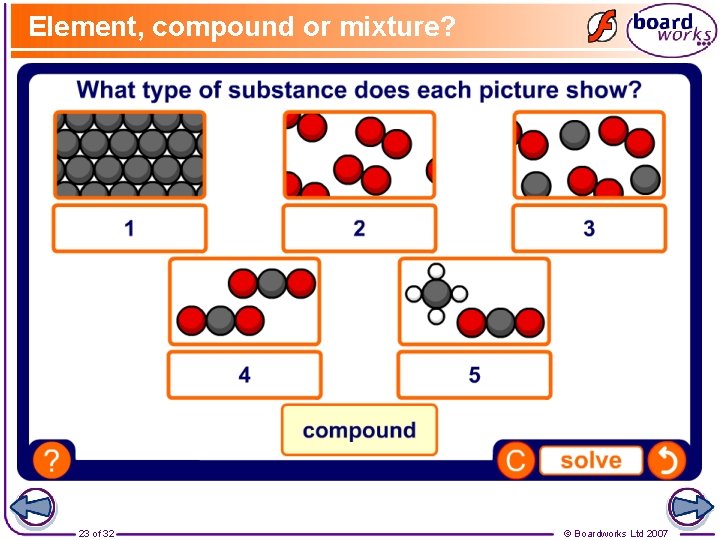
Element, compound or mixture? 23 of 32 © Boardworks Ltd 2007
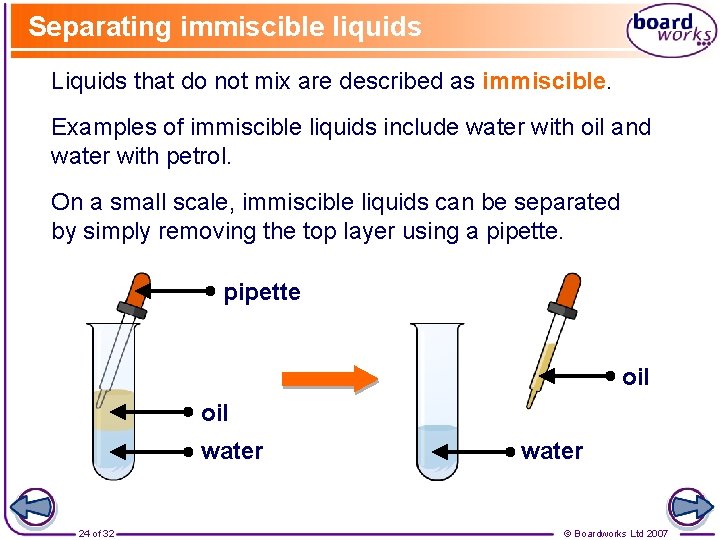
Separating immiscible liquids Liquids that do not mix are described as immiscible. Examples of immiscible liquids include water with oil and water with petrol. On a small scale, immiscible liquids can be separated by simply removing the top layer using a pipette oil water 24 of 32 water © Boardworks Ltd 2007

Separating miscible liquids Liquids that do mix are described as miscible. Examples of miscible liquids are water with alcohol and petrol with kerosine. water and Miscible liquids can be alcohol separated by heating them mixed to boiling. The components of the mixture have different together boiling points and so will boil off at different temperatures. This is called distillation. Substances with low boiling points are collected first, while those with higher boiling points are collected later. 25 of 32 © Boardworks Ltd 2007

Distillation apparatus What apparatus is needed for distillation? condenser Flask of mixed liquids water in Collection flask thermometer water out heater (bunsen) A condenser is used to recover the liquids as they boil off. This piece of apparatus is a tube that has cold water circulating around the outside. It cools down vapours and condenses them back to a liquid. 26 of 32 © Boardworks Ltd 2007

Separating mixtures 27 of 32 © Boardworks Ltd 2007

Experiment to separate a mixture Reggie has been given a mixture of salt, sand water. He needs to separate these three substances, but cannot remember how. Reggie needs your help! Write an experimental plan for Reggie to follow so that he will end up with the sand, salt and water in separate containers. Start your plan with a list of the equipment that is needed for separating this mixture. Then write a step-by-step plan that is easy for Reggie or anyone else to follow. 28 of 32 © Boardworks Ltd 2007
29 of 32 © Boardworks Ltd 2007

Glossary l atom – The smallest particle that can exist on its own. l boiling point – The temperature at which a pure liquid becomes a gas. l compound – A substance made up of two or more different types of atoms that are chemically joined together. l element – A substance made up of only one type of atom. l formula – The symbols and numbers that represent the ratio of different atoms in a substance. l immiscible – Liquids that do not mix. l miscible – Liquids that mix. l mixture – Two or more substances that are mixed but not chemically joined together. 30 of 32 © Boardworks Ltd 2007

Anagrams 31 of 32 © Boardworks Ltd 2007

Multiple-choice quiz 32 of 32 © Boardworks Ltd 2007
- Slides: 32