1 Objective Agehardening MicrostructureProperties II Age Hardening AlCu

1 Objective Agehardening Microstructure-Properties: II Age Hardening Al-Cu Al-Ag steels mechanisms 27 -302 Lecture 9 Fall, 2002 Prof. A. D. Rollett
2 Materials Tetrahedron Processing Performance Objective Agehardening Al-Cu Al-Ag steels mechanisms Microstructure Properties

3 Objective Agehardening Al-Cu Al-Ag steels mechanisms • The objective of this lecture is to describe the relationship between precipitation and hardness as an example of a key microstructure-property relationship.

4 References Objective Agehardening Al-Cu Al-Ag steels mechanisms • Phase transformations in metals and alloys, D. A. Porter, & K. E. Easterling, Chapman & Hall. • Materials Principles & Practice, Butterworth Heinemann, Edited by C. Newey & G. Weaver. • Mechanical Metallurgy, Mc. Graw. Hill, G. E. Dieter, 3 rd Ed. • Hull, D. and D. J. Bacon (1984). Introduction to Dislocations. Oxford, UK, Pergamon. • Courtney, T. H. (2000). Mechanical Behavior of Materials. Boston, Mc. Graw-Hill.

5 Notation Objective Agehardening Al-Cu Al-Ag steels mechanisms a : = lattice parameter e : = strain, misfit (or similar quantity to describe a hardening mechanism) G : = shear modulus b : = Burgers vector r : = Particle size (radius) f VV(a) : = volume fraction (of precipitates) s : = stress (macroscopic) t : = shear stress (critical value, in some cases) g : = boundary energy, e. g. anti-phase boundary <L 3> : = mean intercept length (of precipitates) l : = mean spacing (of precipitates)
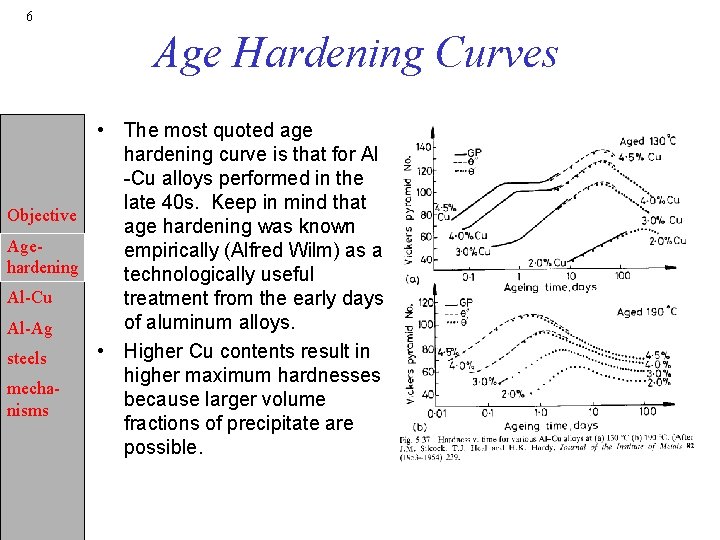
6 Age Hardening Curves • The most quoted age hardening curve is that for Al -Cu alloys performed in the late 40 s. Keep in mind that Objective age hardening was known Ageempirically (Alfred Wilm) as a hardening technologically useful Al-Cu treatment from the early days of aluminum alloys. Al-Ag • Higher Cu contents result in steels higher maximum hardnesses mechabecause larger volume nisms fractions of precipitate are possible.

7 Al-Cu precipitation sequence Objective Agehardening Al-Cu Al-Ag steels mechanisms • The sequence is: a 0 a 1 + GP-zones a 2 + q“ a 3 + q’ a 4 + q • The phase are: an - fcc aluminum; nth subscript denotes each equilibrium GP zones - mono-atomic layers of Cu on (001)Al q“ - thin discs, fully coherent with matrix q’ - disc-shaped, semi-coherent on (001)q’ bct. q - incoherent interface, ~spherical, complex bodycentered tetragonal (bct).
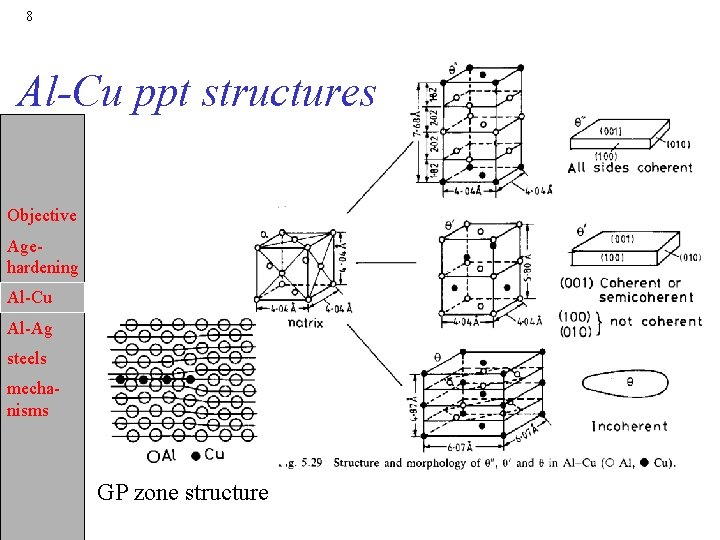
8 Al-Cu ppt structures Objective Agehardening Al-Cu Al-Ag steels mechanisms GP zone structure

9 Al-Cu microstructures • This tableau shows which of the different ppt types are associated with which part of the hardening curve. Objective Agehardening GP zones q’ Al-Cu Al-Ag steels mechanisms q” q

10 Al-Cu driving forces • Each precipitate has a different free energy curve w. r. t composition. Exception is the GP zone, which may be regarded as continuous with the alloy (leading to the possibility of spinodal decomposition, discussed later). Objective • P&E fig. 5. 27 illustrates the sequence of successively greater Agefree energy decreases and also successively greater ∆G*. hardening • P&E fig. 5. 28 illustrates the point that the nucleation barriers Al-Cu are much smaller for each individual nucleation step when the next precipitate nucleates heterogeneously on the previous Al-Ag structure. steels mechanisms
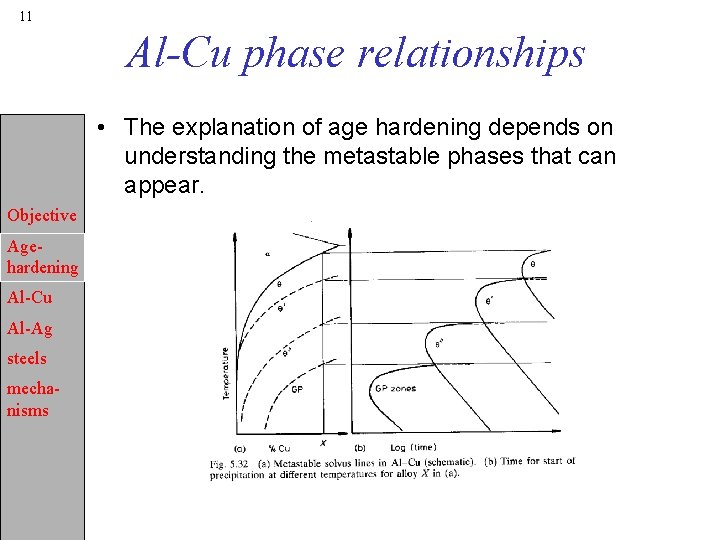
11 Al-Cu phase relationships • The explanation of age hardening depends on understanding the metastable phases that can appear. Objective Agehardening Al-Cu Al-Ag steels mechanisms

12 Nucleation sites, reversion • The nucleation sites vary depending on circumstances. • q“ most likely nucleates on GP zones by adding additional layers of Cu atoms. Objective • Similarly, q’ nucleates on q“ by in-situ transformation. • However, q’ can also nucleate on dislocations, see P&E fig. Age 5. 31 a. hardening • The full sequence is only observable for annealing Al-Cu temperatures below the GP solvus. Any of the intermediate Al-Ag precipitates can be dissolved, reverted, by increasing the steels temperature above the relevant solvus, fig. 5. 32. mechanisms
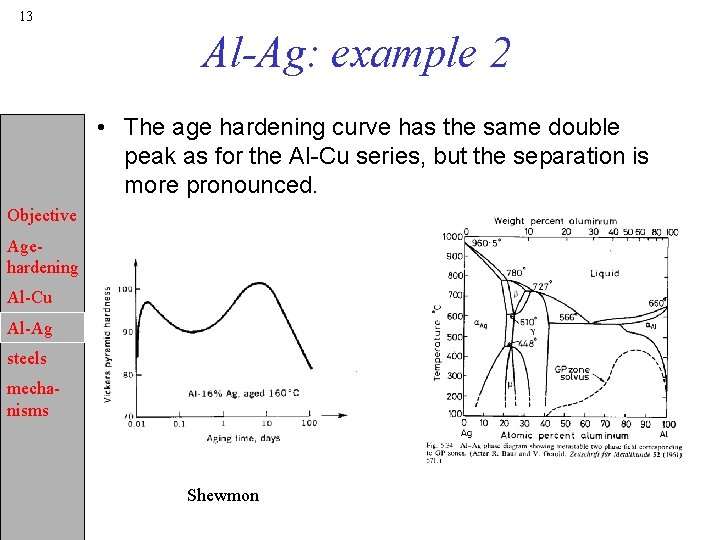
13 Al-Ag: example 2 • The age hardening curve has the same double peak as for the Al-Cu series, but the separation is more pronounced. Objective Agehardening Al-Cu Al-Ag steels mechanisms Shewmon
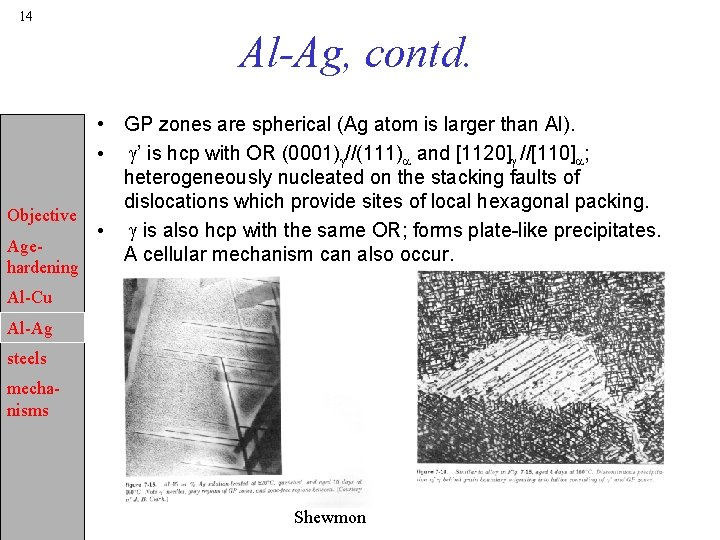
14 Al-Ag, contd. • GP zones are spherical (Ag atom is larger than Al). • g’ is hcp with OR (0001)g//(111)a and [1120]g //[110]a; heterogeneously nucleated on the stacking faults of dislocations which provide sites of local hexagonal packing. Objective • g is also hcp with the same OR; forms plate-like precipitates. Age. A cellular mechanism can also occur. hardening Al-Cu Al-Ag steels mechanisms Shewmon

15 Age hardening in steel: example 3 • It is important to understand that age hardening occurs in almost any system in which the solid solubility decreases appreciably with decreasing temperature. Ferrite has a very low solubility for carbon and therefore age hardening (also called quench hardening) occurs here too. To avoid it, the Objective soluble carbon levels must be reduced, which is a common Ageobjective of the IF or interstitial-free steel grades. These have hardening additions of carbide formers such as Ti or Nb to sequester the Al-Cu C. Al-Ag steels mechanisms Shewmon

16 Cutting versus Bowing • At small sizes, the dislocation cuts through the particle at a lower stress than the Orowan bowing stress (and so this is what is observed). Larger particles mean higher cutting stresses. Objective • At large sizes, the dislocation bows around the particle more Ageeasily than it cuts through it (so no cutting is observed). hardening Larger particles mean fewer particles (via coarsening) hence Al-Cu lower flow stresses. Al-Ag steels mechanisms Particles becoming stronger Fewer and fewer particles, further apart
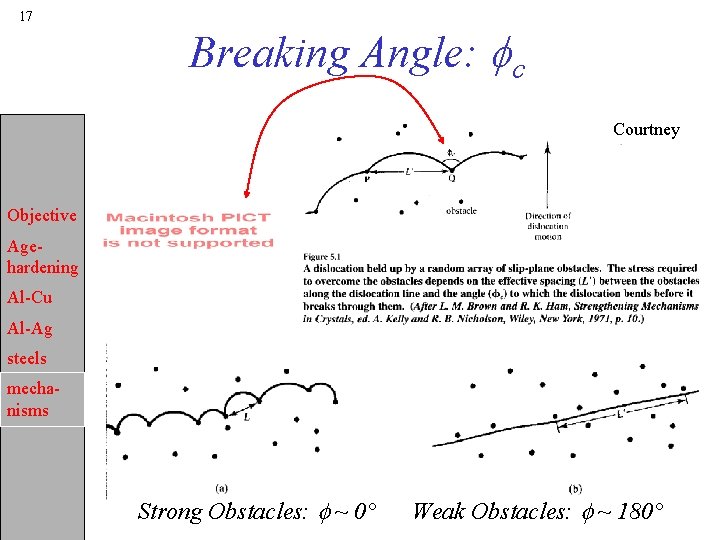
17 Breaking Angle: fc Courtney Objective Agehardening Al-Cu Al-Ag steels mechanisms Strong Obstacles: f ~ 0° Weak Obstacles: f ~ 180°

18 Hardness -microstructure relationships • In order to understand the relationship between microstructure and hardness, we need to delve into the subject of hardening mechanisms. • The central concept is that the strength of a ductile material is Objective governed by dislocation flow past obstacles. Therefore Agestrength can be designed by controlling the density and nature hardening of the obstacles to dislocation motion. Most technological Al-Cu (metallic) alloys rely on precipitation hardening in one form or another to achieve high strengths. Ceramics, on the other Al-Ag hand, are intrinsically harder and therefore the main objective steels of strengthening is to increase their fracture toughness and mechathereby increase their (reliable) load carrying capacity. The nisms objective of this discussion is therefore to bring your attention to a number of ways in which we can understand predict the contributions to strength of different types of obstacle.

19 Strengthening Methods Objective Agehardening Al-Cu Al-Ag steels mechanisms • Microstructural Feature: strength dependence. • Dislocations: strain/work hardening (discussed in 301): (dislocation spacing)1/2. • Internal Boundaries: grain boundaries can have a strong strengthening effect, i. e. the Hall-Petch effect (discussed in 301): (grain size)-1/2. • Dislocation Boundaries (low angle boundaries): (subgrain size)-1. • Second Phase Particles: particle spacing. • Solutes: (concentration)1/2.

20 Mechanisms of particle strengthening 1) Coherency Hardening: differences in density between the particle and the matrix give rise to elastic stresses in the vicinity of the particle. 2) Chemical Hardening: Objective creation of new surface when a particle is sheared increases the area of the interphase boundary, which increases the energy associated with the interface and hence an additional force must be exerted on the dislocation to force it through the particle. Age 3) Order Hardening: passage of a dislocation through an ordered particle, hardening e. g. Ni 3 Al in superalloys, results in a disordered lattice and the creation of antiphase boundaries. Al-Cu Al-Ag steels mechanisms 4) Stacking-fault Hardening: a difference in stacking fault energy between particle and matrix, e. g. Ag in Al, increases flow stress because of the different separation of partial dislocations in the two phases. 5) Modulus Hardening: a large difference in elastic modulus results in image forces when a dislocation in the matrix approaches a particle. Consider, e. g. , the difference between silver particles (nearly the same shear modulus) and iron particles (much higher shear modulus) in aluminum.

21 Dislocations • A re-statement of the governing equation for strength controlled by obstacle spacing: Objective Agehardening Al-Cu Al-Ag steels mechanisms
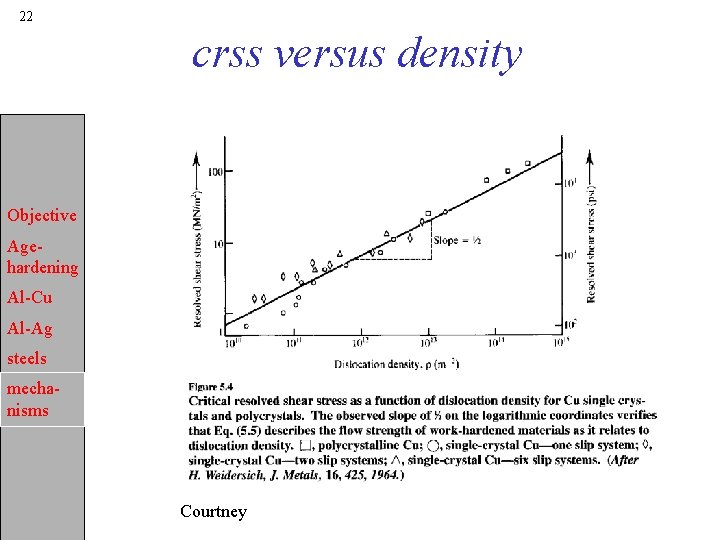
22 crss versus density Objective Agehardening Al-Cu Al-Ag steels mechanisms Courtney

23 Dislocation Boundaries Objective Agehardening Al-Cu Al-Ag steels mechanisms • At large strains and higher temperatures, low angle boundaries appear as a subgrain network forms. We distinguish this microstructural feature from the first two categories because the [lattice] misorientations are much smaller (2 -5°) than grain boundaries (15°+) and they are distinct from statistically stored dislocations. This strengthening method is most important at high temperatures where other microstructural features such as solutes are weak. • The contribution to the flow strength is typically found to be proportional to (grain size)-1; this is in contrast to the 1/√d dependence of the Hall-Patch effect.

24 Solutes Objective Agehardening Al-Cu Al-Ag steels mechanisms • Solutes in a crystal act as obstacles to dislocation motion through their elastic and/or chemical interactions with dislocations. Most solutes are weak hardeners except for the (technologically) important class of interstitial solutes that induce anisotropic distortions of the lattice, e. g. tetragonal distortions of C in Fe.

25 Substitutional solutes • Most Solutes have only a rather weak effect on strength. In other words, even if you put several per cent of a soluble atom into another element, you will not see a dramatic increase in flow stress. These remarks can be quantified by going back to the Orowan equation, i. e. the force balance between the Objective forward motion and the resisting force: Agehardening Al-Cu Al-Ag steels mechanisms tcrss = µb/l. • For substitutional solutes, the numerator in the RHS, i. e. the reaction force from the solute atoms is of order Gb 2/120, which is a small number. This is so because the small differences in size between solute and matrix atoms results in a small interaction energy with dislocations. Thus they are weak obstacles and dislocations remain nearly straight when interacting with solutes (“weak obstacles”, 7 slides before this).

26 Interstitial solutes Objective Agehardening Al-Cu Al-Ag steels mechanisms • Interstitials in bcc, however, can exert forces on the order of Gb 2/5 to Gb 2/10, which are large values. In this case, the dislocations bow out significantly between the atoms, and the breaking angle deviates significantly from 180°. In this case, the concentration dependence is easy to obtain. The spacing between interstitials is inversely proportional to the (square root of the) concentration, and so we can insert a spacing into the standard (Orowan bowing) formula to get the following, where A is a constant of order unity: t = AGb(√c/b) = AG√c.

27 Strength vs. solute content: examples Examples: Objective Agehardening Al-Cu Al-Ag steels mechanisms a) Substitutional solutes in Cu b) Interstitial solutes in Nb, Fe c) Ca in Na. Cl [Courtney]

28 Second Phase Particles • Whether introduced as insoluble particles in powder compaction, or as precipitates in a solid state reaction, second phase particles are generally the most potent strengthening agent in practical high strength engineering materials. Iron. Objective base, aluminum, nickel, titanium alloys all employ second phases to achieve high strength. Agehardening • Quantitative relationships: from previous stereological analysis Al-Cu (301 - lecture 4): Al-Ag steels mechanisms

29 Mechanisms of particle strengthening 1) Coherency Hardening: differences in density between the particle and the matrix give rise to elastic stresses in the vicinity of the particle. 2) Chemical Hardening: Objective Agehardening Al-Cu Al-Ag steels mechanisms creation of new surface when a particle is sheared increases the area of the interphase boundary, which increases the energy associated with the interface and hence an additional force must be exerted on the dislocation to force it through the particle. 3) Order Hardening: passage of a dislocation through an ordered particle, e. g. Ni 3 Al in superalloys, results in a disordered lattice and the creation of antiphase boundaries. 4) Stacking-fault Hardening: a difference in stacking fault energy between particle and matrix, e. g. Ag in Al, increases flow stress because of the different separation of partial dislocations in the two phases. 5) Modulus Hardening: a large difference in elastic modulus results in image forces when a dislocation in the matrix approaches a particle. Consider, e. g. , the difference between silver particles (nearly the same shear modulus) and iron particles (much higher shear modulus) in aluminum.

30 Coherency hardening Differences in density between the particle and the matrix give rise to elastic stresses in the vicinity of the particle. This has been analyzed on the basis of the elastic stresses that exist in the matrix adjacent to a particle that has a different lattice Objective parameter than the matrix. Ignoring differences in modulus for now, and setting a parameter, e, that approximates a strain to Agehardening characterize the magnitude of the effect. For Al-Cu Al-Ag steels mechanisms e = (aparticle – amatrix )/ amatrix t = 7|e|3/2 G(rf/b)1/2 • This mechanism applies to the early stages of precipitation, e. g. strengthening by GP zones.

31 Chemical hardening • Cutting through a particle with a dislocation displaces one half relative to the other by b, thereby creating new interfacial energy of 2πrbg, where g is the interfacial energy between the matrix and the particle. The distance over which this energy Objective has to be created occurs at the entry and exit points and so the characteristic distance is of order b. Thus the force is Agehardening d. E/dx, or, Al-Cu Al-Ag steels mechanisms F = 2πrbg/2 b = πrg

32 Chemical hardening, contd. • If the dislocations are straight, we can approximate the spacing between particles as L=2 r/f. Dividing the force by b. L to find the stress, Objective Agehardening Al-Cu Al-Ag steels mechanisms t = πfg/2 b. • A more realistic approach produces the following relationship. t = 2 G{g/Gr}3/2(fr/b)1/2 • Courtney defines a chemical hardening parameter, ech = g/Gr, related to the interfacial energy, modulus and particle size. This parameter is precisely analogous to the same parameter used, e. g. in APB hardening. Chemical hardening applies only in the early stages of precipitation.

33 Order Hardening • The hardening depends on the product of the antiphaseboundary energy (APBE) and the area swept by a dislocation in a particle. Thus the increase in flow stress is given by: Objective Agehardening Al-Cu Al-Ag steels mechanisms t = πf{APBE}/2 b • In general, low values of the APBE not only predict small increments in hardness, but also the result that the dislocations can move through the particles independently of one another. A more detailed analysis, not presented here, shows a square root dependence on volume fraction, with particle size, t = 0. 7 Ge 3/2 √(fr/b) eord= APBE/Gb • Important for Ni-based superalloys

34 Modulus hardening • The line length in the particle is 2 r and the change in tension is (Gparticle-Gmatrix)b 2/2, assuming the same Burgers vector in matrix and particle. Multiplying the two together and dividing by the distance, i. e. the radius, we get: Objective F= b 2(Gparticle-Gmatrix) = Gb 2 e, Agehardening Al-Cu Al-Ag steels mechanisms where e = (Gparticle-Gmatrix)/Gmatrix, a measure similar to that used in solution hardening. • More realistic estimates of modulus hardening lead to the following formula: t = 10 -2 G e 3/2 √{fr/b} • Think of modulus hardening as being caused by a temporary increase in dislocation line energy while it resides within a particle.

35 Summary Objective Agehardening Al-Cu Al-Ag steels mechanisms • A great variety of hardening mechanisms exist. • Their functional dependence on parameters such as particle size, spacing, volume fraction, are similar. It is difficult therefore, to distinguish experimentally between the mechanisms. • Microscopy is required in order to determine which mechanism is operative. • Particle growth results in stronger particles; in most cases, however, coarsening takes place simultaneously which increases the particle spacing. Orowan bowing takes over from particle cutting at some point in the aging process. • Particle hardening is essential to technological alloys, at least for structural applications.

36 Sample Problem • • • Objective Agehardening Al-Cu Al-Ag • • steels mechanisms • • • From Dieter, p 219 (adapted): Question: Al-4%Cu (by wt. ) has a yield stress of 600 MPa. Estimate the particle size and spacing. Solution: recognize that this stress relates to age hardening beyond the peak hardness. Therefore use the Orowan bowing stress to estimate the stress. s = <M> tcrss = <M> Gb/l G=27. 6 GPa; b=0. 25 nm; <M>=3. 1: spacing = 3. 1*27, 600*0. 25. 10 -9/ 600= 35. 7 nm Now we must estimate the volume fraction of particles for which we use the phase diagram, assuming that we are dealing with the equilibrium phase, q, which is 54 w/o Cu, and the a in equilibrium with it, 0. 5 w/o Cu. Wt. % Al = (54 -4)/(54 -0. 5) = 93. 5; wt. % q = 4 -0. 5/(54 -0. 5)=6. 5 Volume of a = 93. 5 gm/2. 7 gm/cm 3 =34. 6 cm 3 Volume of q = 6. 5/ 4. 443 gm/cm 3 = 1. 5 cm 3 Volume fraction of a = 0. 96; volume fraction of q = 0. 04. Use l=4 r(1 -f)/3 f (slide 22): r =3*0. 04*35. 7/4/(1 -0. 04) = 1. 12 nm.
- Slides: 36