1 NONRENAL INDICATIONS INTOXICATIONS INBORN ERRORS OF METABOLISM

1 NON-RENAL INDICATIONS: INTOXICATIONS & INBORN ERRORS OF METABOLISM STEFANO PICCA, MD Dialysis Unit- Dept of Nephrology and Urology “Bambino Gesù” Pediatric Research Hospital ROMA, Italy

OUTLINE • Variables in toxic agents elimination • Exogenous toxicity: Experience with toxic agents in PICU • Endogenous toxicity: Inborn Errors of Metabolism: which is the role of RRT in determining the outcome?

FACTORS POTENTIALLY AFFECTING DRUG CLEARANCE DURING RENAL REPLACEMENT THERAPY DRUG PROPERTIES DEVICE PROPERTIES • MOLECULAR WEIGHT • COMPOSITION • PLASMA PROTEIN BINDING • SURFACE AREA • VOLUME OF DISTRIBUTION • PORE SIZE • PROPORTION OF RENAL • ADSORPTION CLEARANCE What is unique to Pediatric Intoxications? • Vehicle in which the medication was delivered • Metabolism of drug • Volume of distribution • Variable size of the child Adapted from Pea F and Bunchman TE, 2010
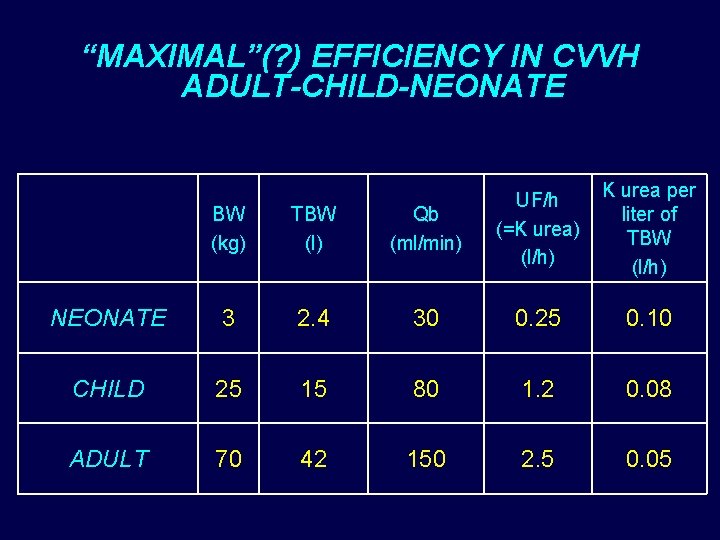
“MAXIMAL”(? ) EFFICIENCY IN CVVH ADULT-CHILD-NEONATE K urea per liter of TBW (l/h) BW (kg) TBW (l) Qb (ml/min) UF/h (=K urea) (l/h) NEONATE 3 2. 4 30 0. 25 0. 10 CHILD 25 15 80 1. 2 0. 08 ADULT 70 42 150 2. 5 0. 05
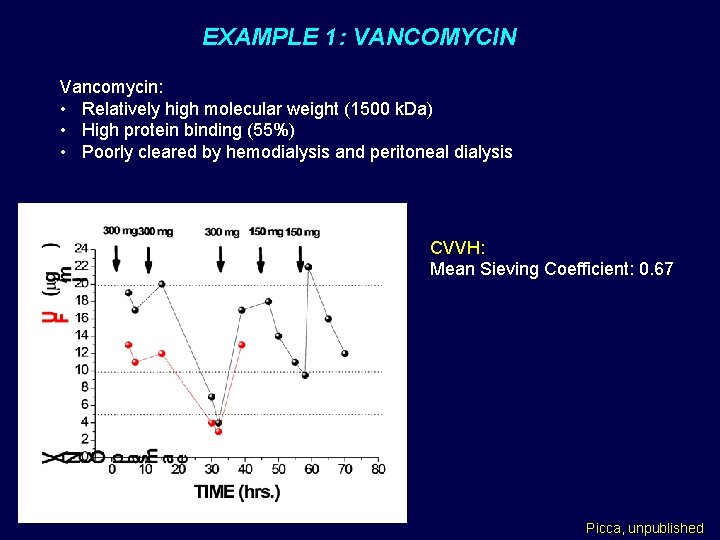
EXAMPLE 1: VANCOMYCIN Vancomycin: • Relatively high molecular weight (1500 k. Da) • High protein binding (55%) • Poorly cleared by hemodialysis and peritoneal dialysis CVVH: Mean Sieving Coefficient: 0. 67 Picca, unpublished

VANCOMYCIN OVERDOSE TREATMENT IN CHILDREN Author n membrane modality T 1/2 Bunchman (1999) 2 cellulose triacetate HD 31 to 1. 9 hrs Akil (2011) 1 polysulfone CVVH 231 to 31. 5 hrs Goebel (1999) 1 polysulfone CVVHD 41. 5 hrs during CVVHD Shah (2000) 1 polysulfone CVVH From 250 to 27 mcg/ml in 58 hrs

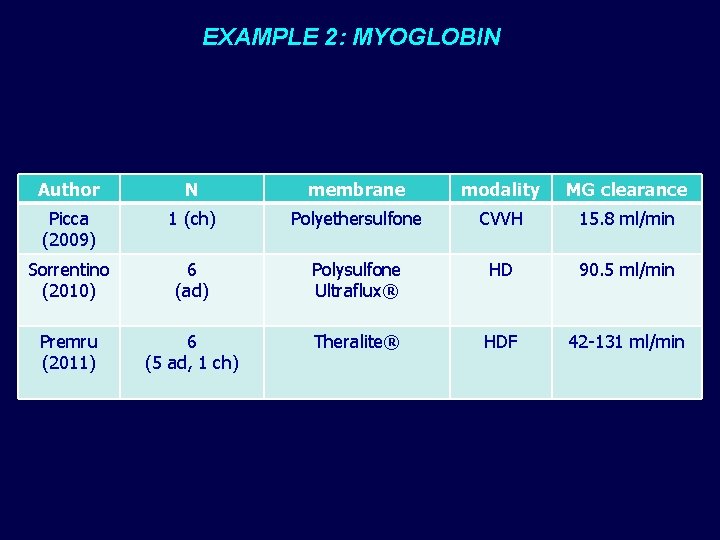
EXAMPLE 2: MYOGLOBIN Author N membrane modality MG clearance Picca (2009) 1 (ch) Polyethersulfone CVVH 15. 8 ml/min Sorrentino (2010) 6 (ad) Polysulfone Ultraflux® HD 90. 5 ml/min Premru (2011) 6 (5 ad, 1 ch) Theralite® HDF 42 -131 ml/min
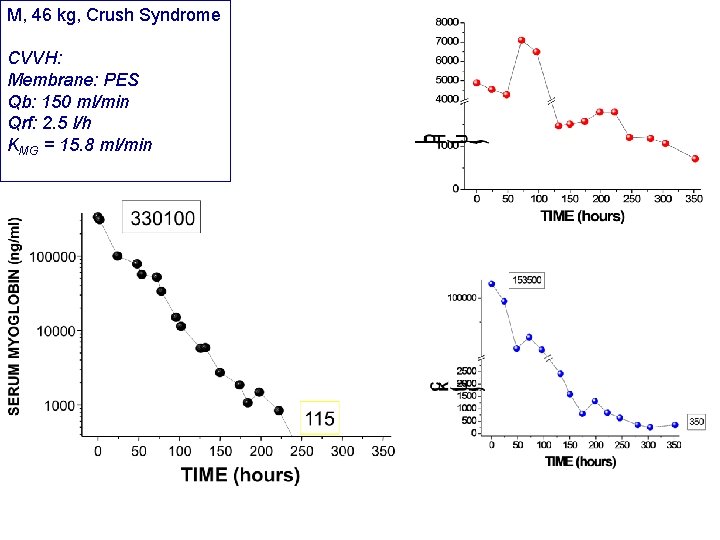
M, 46 kg, Crush Syndrome CVVH: Membrane: PES Qb: 150 ml/min Qrf: 2. 5 l/h KMG = 15. 8 ml/min

EXAMPLE 3: BORON • Boron (boric acid): component of topical disinfectants • Acute boron intoxication: erythematous rash (“boiled lobster”), AKI, vomiting, diarrhea, restlessness, headache, irritability, delirium, seizure, and coma • 65% boron acute intoxications in pediatric age (2009 Annual Report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 27 th Annual Report, 2009) • Although severe toxicity is reported only with very high boron serum levels (>300 µg/ml), lethal dose in infants is considered to be 3 -6 g • Dialysis is known to be effective in adults. No data in children with extracorporeal dialysis. • Case: 5. 5 kg, three-month infant, accidental ingestion of 160 ml of milk and water saturated solution of boric acid (3, 6 g). At admittance: no symptoms, normal hepatic and kidney function. Metabolic acidosis.
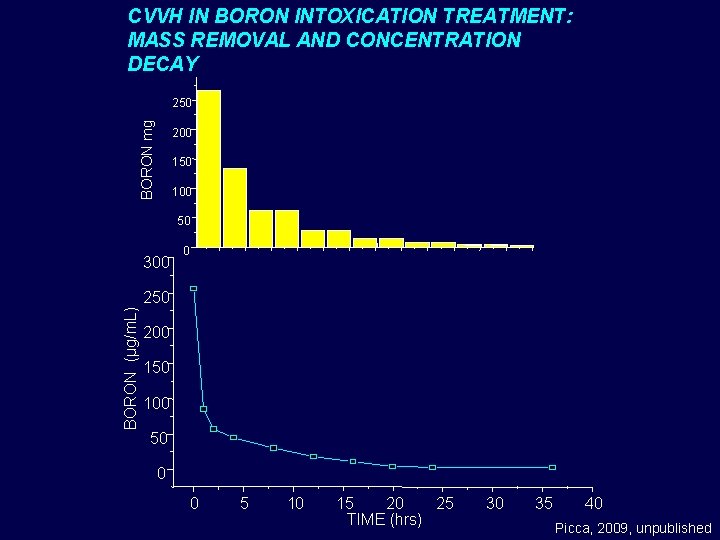
CVVH IN BORON INTOXICATION TREATMENT: MASS REMOVAL AND CONCENTRATION DECAY BORON mg 250 200 150 100 50 300 0 BORON (µg/m. L) 250 200 150 100 50 0 0 5 10 15 20 25 TIME (hrs) 30 35 40 Picca, 2009, unpublished

KEY POINTS OF NEONATAL HYPERAMMONEMIA • Neonatal hyperammonemia is mainly due to urea cycle defects and organic acidurias • Hyperammonemia is extremely toxic (per se and through intracellular excess glutamine formation) to the brain causing astrocyte swelling, brain edema, coma, death or severe disability • When hyperammonemia does not respond to medical and dietetic treatment, dialysis has to be established in order to achieve rapid ammonium removal before neurological impairment or death occur • Ammonium easily diffuses through membranes. Extracorporeal dialysis provides higher and faster ammonium removal than peritoneal dialysis.

PROGNOSTIC INDICATORS IN DIALYZED NEONATES ASSOCIATED WITH SURVIVAL Schaefer, 1999 Picca, 2001 Mc. Bryde, 2006 Pela, 2008 • 50% p. NH 4 decay time < 7 hrs • (catheter > 5 F) • pre-treatment coma duration < 33 hrs (no influence of post-treatment duration) • responsiveness to pharmacological therapy • p. NH 4 at admission<180 mol/L • Time to RRT<24 hrs • Medical treatment<24 hrs • BP> 5%ile at RRT initiation • HD initial RRT (trend) • pre-treatment coma duration < 10 hrs Arbeiter, 2009 • Citrullinemia Westrope, 2010 • • Favorable PRISM score Lesser cardioactive drug requirement

SINP ITALIAN SOCIETY OF PEDIATRIC NEPHROLOGY Italian Study Group “Dialysis Treatment of Neonatal hyperammonemia” (Coord. : S. Picca, MD)
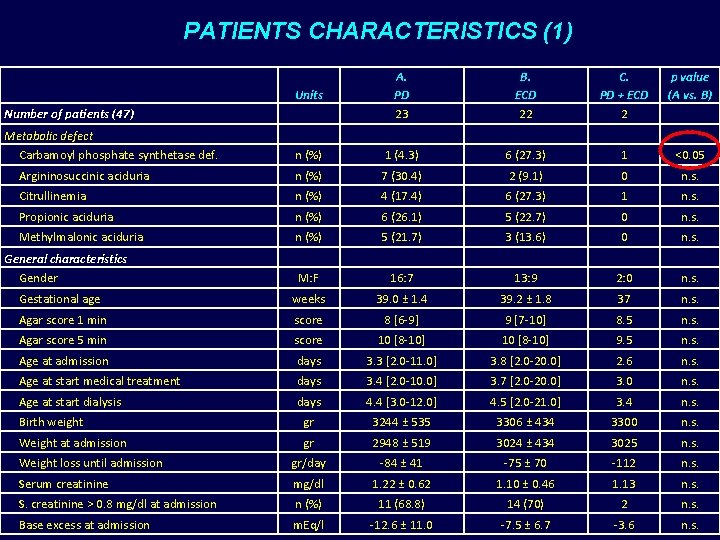
PATIENTS CHARACTERISTICS (1) Units A. PD 23 B. ECD 22 C. PD + ECD 2 p value (A vs. B) n (%) 1 (4. 3) 6 (27. 3) 1 <0. 05 Argininosuccinic aciduria n (%) 7 (30. 4) 2 (9. 1) 0 n. s. Citrullinemia n (%) 4 (17. 4) 6 (27. 3) 1 n. s. Propionic aciduria n (%) 6 (26. 1) 5 (22. 7) 0 n. s. Methylmalonic aciduria n (%) 5 (21. 7) 3 (13. 6) 0 n. s. M: F 16: 7 13: 9 2: 0 n. s. Gestational age weeks 39. 0 ± 1. 4 39. 2 ± 1. 8 37 n. s. Agar score 1 min score 8 [6 -9] 9 [7 -10] 8. 5 n. s. Agar score 5 min score 10 [8 -10] 9. 5 n. s. Age at admission days 3. 3 [2. 0 -11. 0] 3. 8 [2. 0 -20. 0] 2. 6 n. s. Age at start medical treatment days 3. 4 [2. 0 -10. 0] 3. 7 [2. 0 -20. 0] 3. 0 n. s. Age at start dialysis days 4. 4 [3. 0 -12. 0] 4. 5 [2. 0 -21. 0] 3. 4 n. s. Birth weight gr 3244 ± 535 3306 ± 434 3300 n. s. Weight at admission gr 2948 ± 519 3024 ± 434 3025 n. s. Weight loss until admission gr/day -84 ± 41 -75 ± 70 -112 n. s. Serum creatinine mg/dl 1. 22 ± 0. 62 1. 10 ± 0. 46 1. 13 n. s. S. creatinine > 0. 8 mg/dl at admission n (%) 11 (68. 8) 14 (70) 2 n. s. Base excess at admission m. Eq/l -12. 6 ± 11. 0 -7. 5 ± 6. 7 -3. 6 n. s. Number of patients (47) Metabolic defect Carbamoyl phosphate synthetase def. General characteristics Gender
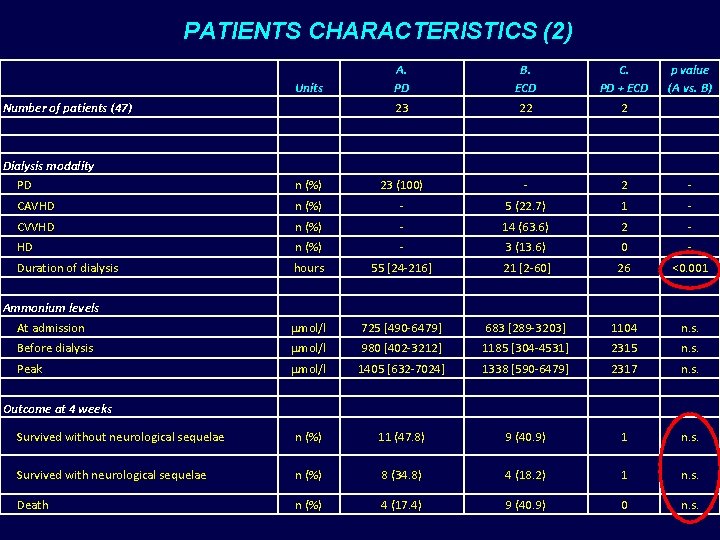
PATIENTS CHARACTERISTICS (2) Units A. PD 23 B. ECD 22 C. PD + ECD 2 p value (A vs. B) n (%) 23 (100) - 2 - CAVHD n (%) - 5 (22. 7) 1 - CVVHD n (%) - 14 (63. 6) 2 - HD n (%) - 3 (13. 6) 0 - Duration of dialysis hours 55 [24 -216] 21 [2 -60] 26 <0. 001 mol/l 725 [490 -6479] 683 [289 -3203] 1104 n. s. Before dialysis mol/l 980 [402 -3212] 1185 [304 -4531] 2315 n. s. Peak mol/l 1405 [632 -7024] 1338 [590 -6479] 2317 n. s. Survived without neurological sequelae n (%) 11 (47. 8) 9 (40. 9) 1 n. s. Survived with neurological sequelae n (%) 8 (34. 8) 4 (18. 2) 1 n. s. Death n (%) 4 (17. 4) 9 (40. 9) 0 n. s. Number of patients (47) Dialysis modality PD Ammonium levels At admission Outcome at 4 weeks
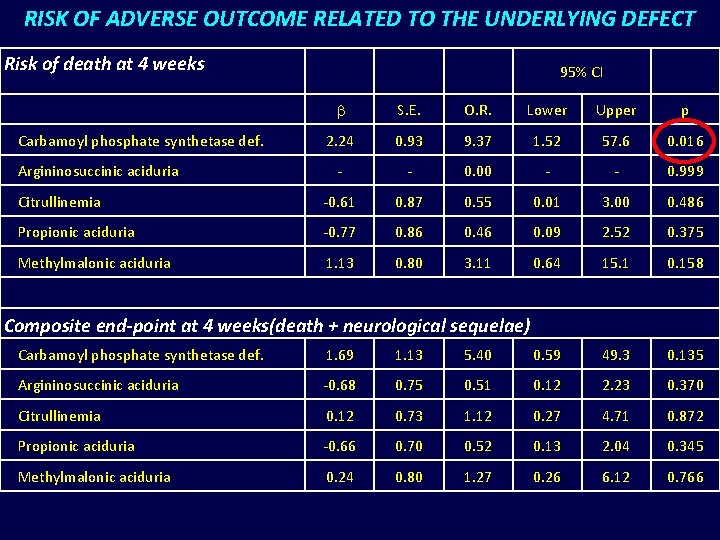
RISK OF ADVERSE OUTCOME RELATED TO THE UNDERLYING DEFECT Risk of death at 4 weeks 95% CI b S. E. O. R. Lower Upper p 2. 24 0. 93 9. 37 1. 52 57. 6 0. 016 - - 0. 00 - - 0. 999 Citrullinemia -0. 61 0. 87 0. 55 0. 01 3. 00 0. 486 Propionic aciduria -0. 77 0. 86 0. 46 0. 09 2. 52 0. 375 Methylmalonic aciduria 1. 13 0. 80 3. 11 0. 64 15. 1 0. 158 Carbamoyl phosphate synthetase def. Argininosuccinic aciduria Composite end-point at 4 weeks(death + neurological sequelae) Carbamoyl phosphate synthetase def. 1. 69 1. 13 5. 40 0. 59 49. 3 0. 135 Argininosuccinic aciduria -0. 68 0. 75 0. 51 0. 12 2. 23 0. 370 Citrullinemia 0. 12 0. 73 1. 12 0. 27 4. 71 0. 872 Propionic aciduria -0. 66 0. 70 0. 52 0. 13 2. 04 0. 345 Methylmalonic aciduria 0. 24 0. 80 1. 27 0. 26 6. 12 0. 766
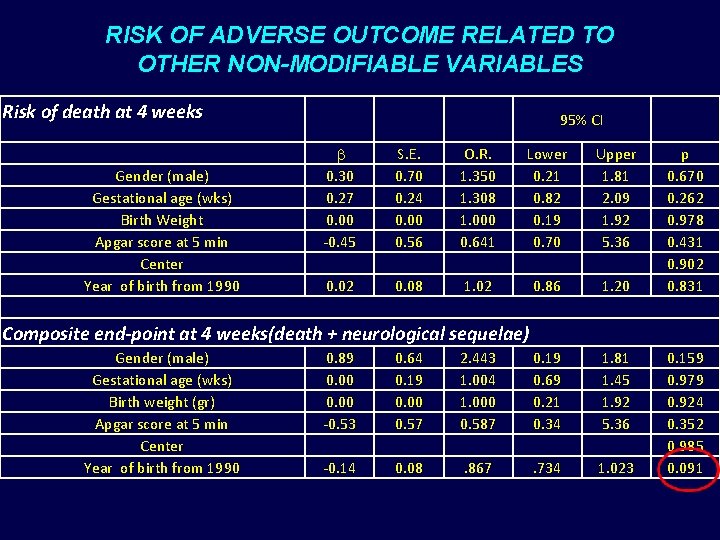
RISK OF ADVERSE OUTCOME RELATED TO OTHER NON-MODIFIABLE VARIABLES Risk of death at 4 weeks Gender (male) Gestational age (wks) Birth Weight Apgar score at 5 min Center Year of birth from 1990 95% CI b 0. 30 0. 27 0. 00 -0. 45 S. E. 0. 70 0. 24 0. 00 0. 56 O. R. 1. 350 1. 308 1. 000 0. 641 Lower 0. 21 0. 82 0. 19 0. 70 Upper 1. 81 2. 09 1. 92 5. 36 0. 02 0. 08 1. 02 0. 86 1. 20 p 0. 670 0. 262 0. 978 0. 431 0. 902 0. 831 Composite end-point at 4 weeks(death + neurological sequelae) Gender (male) Gestational age (wks) Birth weight (gr) Apgar score at 5 min Center Year of birth from 1990 0. 89 0. 00 -0. 53 0. 64 0. 19 0. 00 0. 57 2. 443 1. 004 1. 000 0. 587 0. 19 0. 69 0. 21 0. 34 1. 81 1. 45 1. 92 5. 36 -0. 14 0. 08 . 867 . 734 1. 023 0. 159 0. 979 0. 924 0. 352 0. 985 0. 091
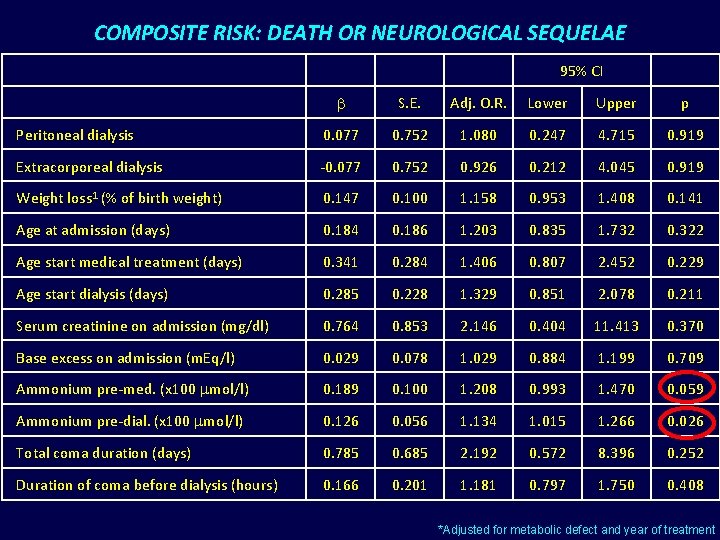
COMPOSITE RISK: DEATH OR NEUROLOGICAL SEQUELAE 95% CI b S. E. Adj. O. R. Lower Upper p Peritoneal dialysis 0. 077 0. 752 1. 080 0. 247 4. 715 0. 919 Extracorporeal dialysis -0. 077 0. 752 0. 926 0. 212 4. 045 0. 919 Weight loss 1 (% of birth weight) 0. 147 0. 100 1. 158 0. 953 1. 408 0. 141 Age at admission (days) 0. 184 0. 186 1. 203 0. 835 1. 732 0. 322 Age start medical treatment (days) 0. 341 0. 284 1. 406 0. 807 2. 452 0. 229 Age start dialysis (days) 0. 285 0. 228 1. 329 0. 851 2. 078 0. 211 Serum creatinine on admission (mg/dl) 0. 764 0. 853 2. 146 0. 404 11. 413 0. 370 Base excess on admission (m. Eq/l) 0. 029 0. 078 1. 029 0. 884 1. 199 0. 709 Ammonium pre-med. (x 100 mol/l) 0. 189 0. 100 1. 208 0. 993 1. 470 0. 059 Ammonium pre-dial. (x 100 mol/l) 0. 126 0. 056 1. 134 1. 015 1. 266 0. 026 Total coma duration (days) 0. 785 0. 685 2. 192 0. 572 8. 396 0. 252 Duration of coma before dialysis (hours) 0. 166 0. 201 1. 181 0. 797 1. 750 0. 408 *Adjusted for metabolic defect and year of treatment
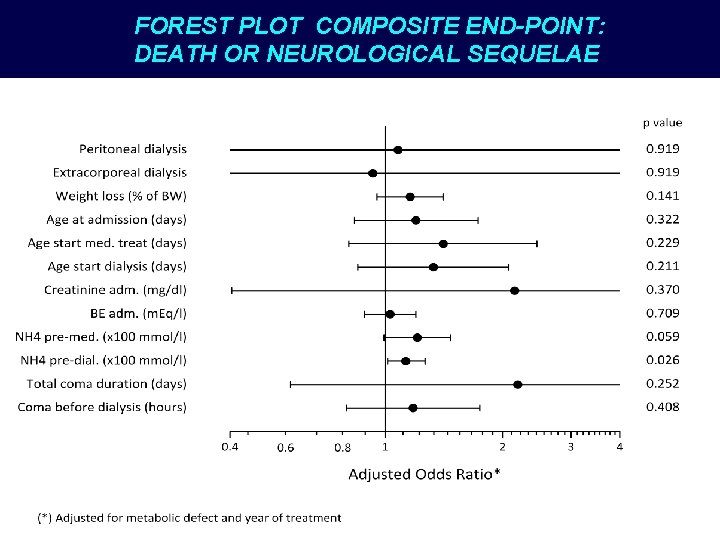
FOREST PLOT COMPOSITE END-POINT: DEATH OR NEUROLOGICAL SEQUELAE
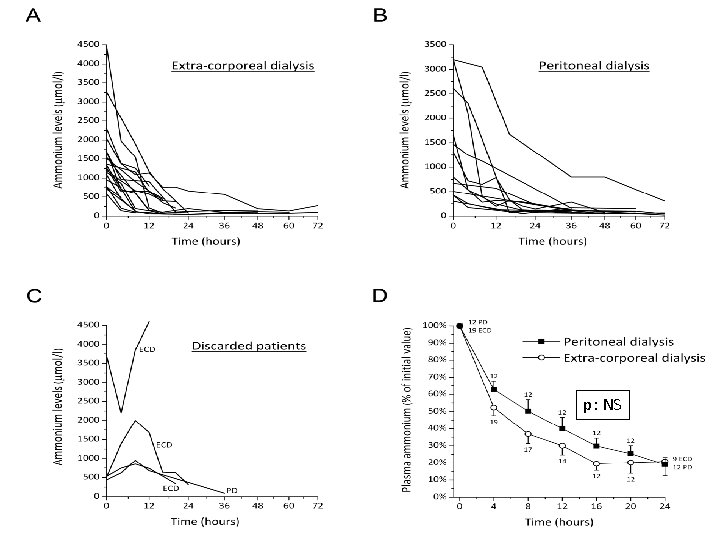
p: NS

THE EVOLUTION OF UCD LONG TERM SURVIVAL Uchino, 1998 • 216 pts with UCD (1978 -1995) • 92 with neonatal onset • 1 -yr survival: 43% (90% with severe neuro-deficit) Kido, 2012 • 254 pts with UCD (1999 -2009) • 77 with neonatal onset • 1 -yr survival: 83% (neuro-deficit NA)



Outcome Neonatal Onset pts (n=29) Short-term <2 nd year of life Long-term >2 nd year of life (median 1. 3 yrs, range 0 -2) (median 12. 5 yrs, range 3 -21) Mortality 48% 27. 5% Cognitive development Normal 71% Mild MR 4. 7% Severe MR 23% 28. 5% 9. 5% 57% No significative difference between UCDs and OAs

CONCLUSIONS • RRT represent a key step in the treatment of endogenous and exogenous intoxications unresponsive to medical treatment • Compared with adults, the depuration of toxic compounds in children is facilitated by the small patient volume • In general, extracorporeal dialysis provides higher and faster detoxification if compared with peritoneal dialysis • In neonatal hyperammonemia, extracorporeal dialysis provides fastest ammonium removal • However, surprisingly, in our cohort extracorporeal and peritoneal dialysis induced a similar ammonium decay (higher glucose uptake with PD? Lesser degree of severity in PD patients? ) • Early initiation of medical treatment may be more important in decreasing ammonium generation rate than using more efficient dialysis techniques (i. e. : extracorporeal dialysis) • Last but most important, dialysis modality did not affect the short term outcome In light of these findings and waiting for validation of these results in other cohorts of patients, peritoneal dialysis in the treatment of neonatal hyperammonemia must be considered as a valid alternative to extracorporeal dialysis.

ACKNOWLEDGEMENTS Bambino Gesù Children Hospital: • Metabolic Unit: Carlo Dionisi-Vici, MD; Andrea Bartuli, MD; Gaetano Sabetta, MD • Clinical Biochemistry Lab: Cristiano Rizzo BSc, Ph. D; Anna Pastore BSc, Ph. D • NICU: all doctors and nurses • Dialysis Unit: Francesco Emma, MD, all doctors and nurses (thanks!) In Italy: • SINP (Italian Society of Pediatric Nephrology) • All doctors from Pediatric Nephrology and NICUs of Genova, Milan, Turin, Padua, Florence, Naples, Bari. In USA • Tim Bunchman, Stuart Goldstein for this opportunity. • Thanks guys.
- Slides: 27