1 Natural History of Type 2 Diabetes Years

1

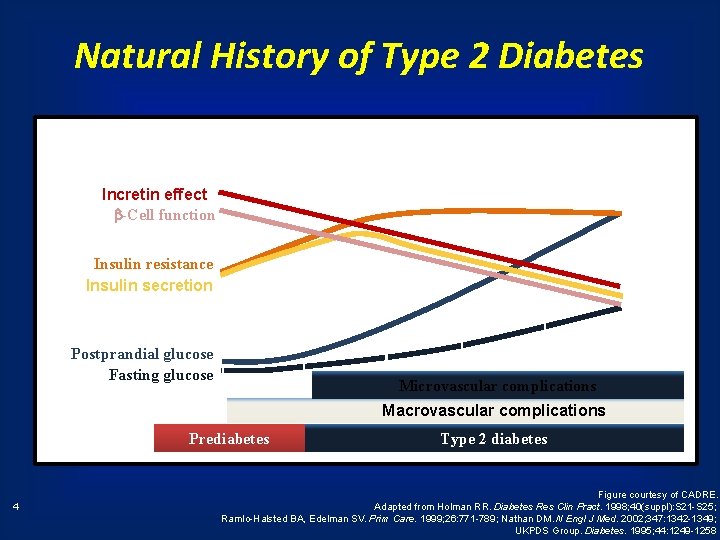
Natural History of Type 2 Diabetes Years from diagnosis – 10 – 5 Onset 5 0 10 15 Diagnosis Incretin effect -Cell function Insulin resistance Insulin secretion Postprandial glucose Fasting glucose Microvascular complications Macrovascular complications Prediabetes 4 Type 2 diabetes Figure courtesy of CADRE. Adapted from Holman RR. Diabetes Res Clin Pract. 1998; 40(suppl): S 21 -S 25; Ramlo-Halsted BA, Edelman SV. Prim Care. 1999; 26: 771 -789; Nathan DM. N Engl J Med. 2002; 347: 1342 -1349; UKPDS Group. Diabetes. 1995; 44: 1249 -1258

DCCT: Results Summary
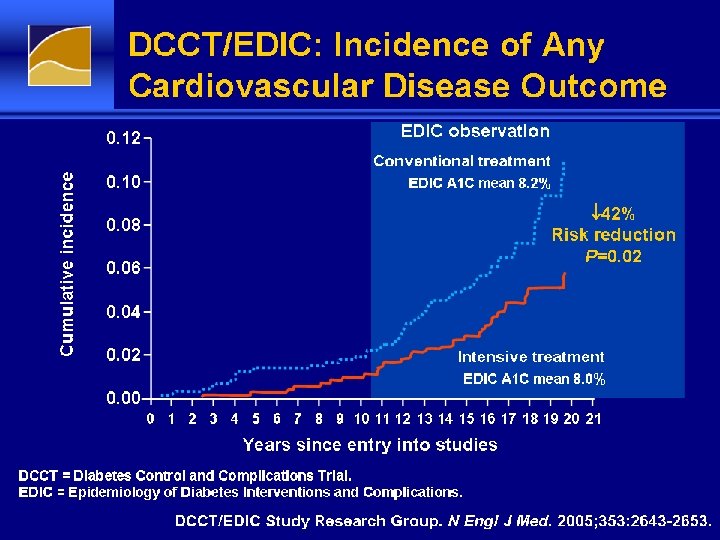
DCCT/EDIC: Incidence of Any Cardiovascular Disease Outcome

DCCT and EDIC Findings • Intensive treatment reduced the risks of retinopathy, nephropathy, and neuropathy by 35% to 90% compared with conventional treatment • Absolute risks of retinopathy and nephropathy were proportional to the A 1 C • Intensive treatment was most effective when begun early, before complications were detectable • Risk reductions achieved at a median A 1 C 7. 3% for intensive treatment (vs 9. 1% for conventional) • Benefits of 6. 5 years of intensive treatment extended well beyond the period of most intensive implementation (“metabolic memory”) Intensive treatment should be started as soon as is safely possible after the onset of T 1 DM and maintained thereafter DCCT/EDIC Research Group. JAMA. 2002; 15; 287: 2563 -2569.

Macrovascular Risk Reduction in T 2 DM • • • Individualized glucose control Hypertension control Dyslipidemia control Smoking cessation Aspirin therapy Diagnosis and management of: – Autonomic cardiac neuropathy – Kidney disease 9 Handelsman Y, et al. Endocr Pract. 2011; 17(suppl 2): 1 -53.

Glycemic targets Diabetes Care 2015; 38(Suppl. 1): S 33–S 40 | DOI: 10. 2337/dc 15 -S 009 10

More Stringent Hb. A 1 c Targets (6. 0 – 6. 5%) • Short disease duration • Long life expectancy • No significant CVD if this can be achieved without significant hypoglycemia or other adverse effects of treatment 11

Less Stringent Hb. A 1 c Targets (7. 5 – 8. 0% or even slightly higher) • History of severe hypoglycemia • Limited life expectancy • Advanced complications • Extensive comorbid conditions • In whom the target is difficult to attain despite intensive self -management education, repeated counseling, and effective doses of multiple glucose-lowering agents, including insulin 12

Lifestyle Interventions At diagnosis, highly motivated patients with Hb. A 1 c already near target (<7. 5%) could be given the opportunity to engage in lifestyle change for a period of 3– 6 months before embarking on pharmacotherapy (usually metformin) 13

Oral Agents and Non-insulin Injectables 14

Metformin • • • Excretion is via the kidney 1 Accumulation is associated with a risk of lactic acidosis 1 Metformin is contraindicated in patients with e. GFR <301 • • However, some national recommendations allow use at lower e. GFR (e. g. NICE) 2 2012 KDOQI guidelines state that metformin may be used in patients with an e. GFR >45 3 e. GFR (m. L/min/1. 73 m 2) Agent Metformin 4 Metabolites Unchanged Elimination ~100% urine >60 60– 30 <15 � � *? � � � indicated; ? indication variable; contraindicated; *consider dose reduction, frequent monitoring and relevant health status e. GFR, estimated glomerular filtration rate; NICE, National Institute for Health and Care Excellence; KDOQI, Kidney Disease Outcomes Quality Initiative 1. Bailey CJ, Day C. Br J Diabetes Vasc Dis 2012; 12: 167– 171; 2. National Institute for Health and Clinical Excellence. Clinical guideline 87. Available at: http: //www. nice. org. uk/guidance/cg 87/resources/guidance-type-2 -diabetes-pdf. Accessed October 2014; 3. National Kidney Foundation. Am J Kidney Dis 2012; 60: 850– 886; 4. Glucophage Summary of Product Characteristics October 2010.
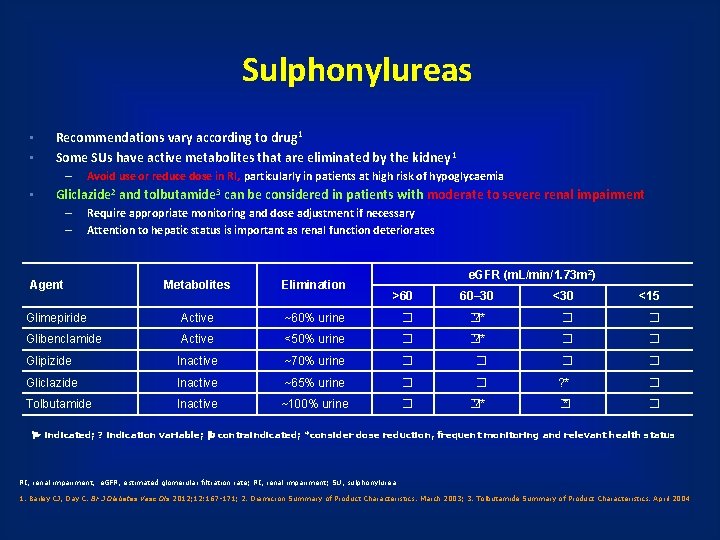
Sulphonylureas • • Recommendations vary according to drug 1 Some SUs have active metabolites that are eliminated by the kidney 1 – • Avoid use or reduce dose in RI, particularly in patients at high risk of hypoglycaemia Gliclazide 2 and tolbutamide 3 can be considered in patients with moderate to severe renal impairment – – Require appropriate monitoring and dose adjustment if necessary Attention to hepatic status is important as renal function deteriorates Agent e. GFR (m. L/min/1. 73 m 2) Metabolites Elimination Glimepiride Active ~60% urine � Glibenclamide Active <50% urine Glipizide Inactive Gliclazide Tolbutamide >60 60– 30 <15 � ? * � � ~70% urine � � Inactive ~65% urine � � ? * � Inactive ~100% urine � � ? * � indicated; ? indication variable; contraindicated; *consider dose reduction, frequent monitoring and relevant health status RI, renal impairment, e. GFR, estimated glomerular filtration rate; RI, renal impairment; SU, sulphonylurea 1. Bailey CJ, Day C. Br J Diabetes Vasc Dis 2012; 12: 167 -171; 2. Diamicron Summary of Product Characteristics. March 2003; 3. Tolbutamide Summary of Product Characteristics. April 2004
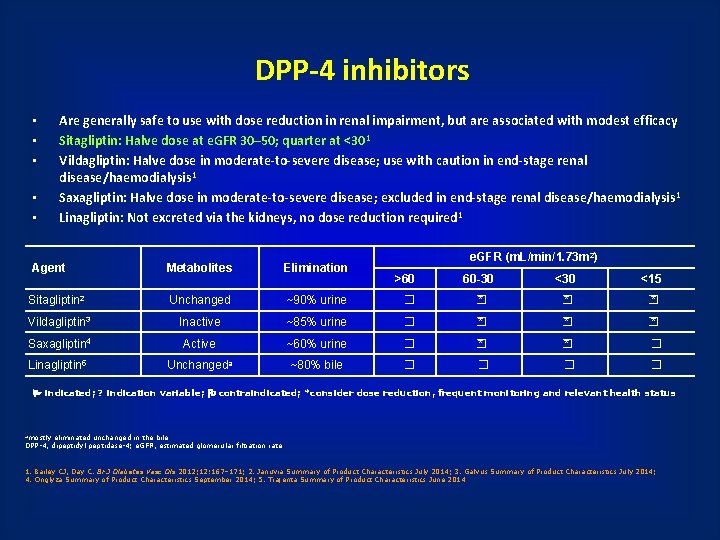
DPP-4 inhibitors • • • Are generally safe to use with dose reduction in renal impairment, but are associated with modest efficacy Sitagliptin: Halve dose at e. GFR 30– 50; quarter at <301 Vildagliptin: Halve dose in moderate-to-severe disease; use with caution in end-stage renal disease/haemodialysis 1 Saxagliptin: Halve dose in moderate-to-severe disease; excluded in end-stage renal disease/haemodialysis 1 Linagliptin: Not excreted via the kidneys, no dose reduction required 1 Agent Metabolites Elimination Sitagliptin 2 Unchanged ~90% urine Vildagliptin 3 Inactive Saxagliptin 4 Linagliptin 5 e. GFR (m. L/min/1. 73 m 2) >60 60 -30 <15 � � * � * ~85% urine � � * � * Active ~60% urine � � * � Unchangeda ~80% bile � � indicated; ? indication variable; contraindicated; *consider dose reduction, frequent monitoring and relevant health status amostly eliminated unchanged in the bile DPP-4, dipeptidyl peptidase-4; e. GFR, estimated glomerular filtration rate 1. Bailey CJ, Day C. Br J Diabetes Vasc Dis 2012; 12: 167– 171; 2. Januvia Summary of Product Characteristics July 2014; 3. Galvus Summary of Product Characteristics July 2014; 4. Onglyza Summary of Product Characteristics September 2014; 5. Trajenta Summary of Product Characteristics June 2014
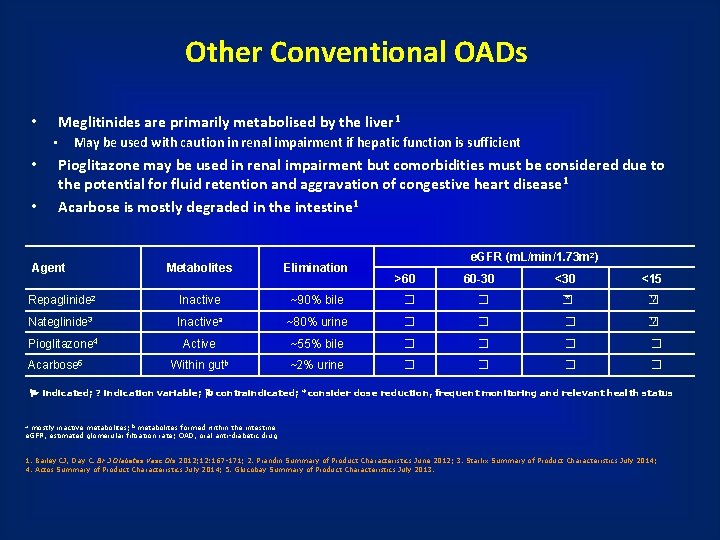
Other Conventional OADs Meglitinides are primarily metabolised by the liver 1 • May be used with caution in renal impairment if hepatic function is sufficient • • • Pioglitazone may be used in renal impairment but comorbidities must be considered due to the potential for fluid retention and aggravation of congestive heart disease 1 Acarbose is mostly degraded in the intestine 1 Agent e. GFR (m. L/min/1. 73 m 2) Metabolites Elimination Repaglinide 2 Inactive ~90% bile � Nateglinide 3 Inactivea ~80% urine Pioglitazone 4 Active Within gutb Acarbose 5 >60 60 -30 <15 � � * � ? � � ? ~55% bile � � ~2% urine � � indicated; ? indication variable; contraindicated; *consider dose reduction, frequent monitoring and relevant health status mostly inactive metabolites; b metabolites formed within the intestine e. GFR, estimated glomerular filtration rate; OAD, oral anti-diabetic drug a 1. Bailey CJ, Day C. Br J Diabetes Vasc Dis 2012; 12: 167 -171; 2. Prandin Summary of Product Characteristics June 2012; 3. Starlix Summary of Product Characteristics July 2014; 4. Actos Summary of Product Characteristics July 2014; 5. Glucobay Summary of Product Characteristics July 2013.
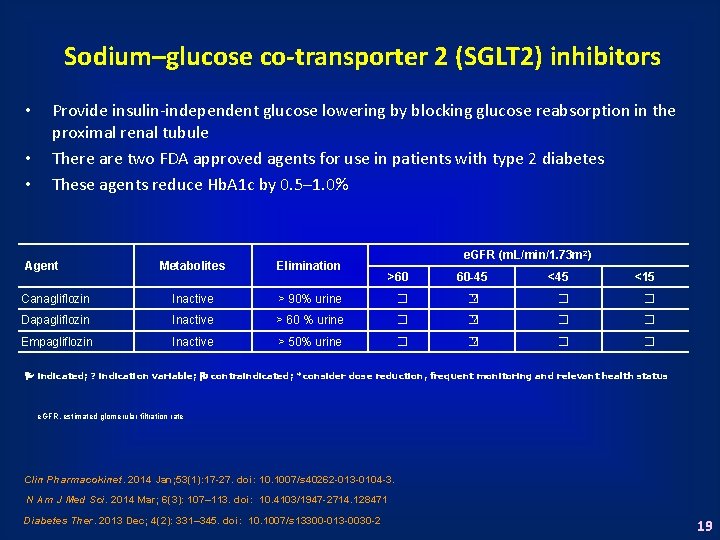
Sodium–glucose co-transporter 2 (SGLT 2) inhibitors • • • Provide insulin-independent glucose lowering by blocking glucose reabsorption in the proximal renal tubule There are two FDA approved agents for use in patients with type 2 diabetes These agents reduce Hb. A 1 c by 0. 5– 1. 0% Agent e. GFR (m. L/min/1. 73 m 2) Metabolites Elimination Canagliflozin Inactive > 90% urine � Dapagliflozin Inactive > 60 % urine Empagliflozin Inactive > 50% urine >60 60 -45 <15 � ? � � � � ? � � indicated; ? indication variable; contraindicated; *consider dose reduction, frequent monitoring and relevant health status e. GFR, estimated glomerular filtration rate Clin Pharmacokinet. 2014 Jan; 53(1): 17 -27. doi: 10. 1007/s 40262 -013 -0104 -3. N Am J Med Sci. 2014 Mar; 6(3): 107– 113. doi: 10. 4103/1947 -2714. 128471 Diabetes Ther. 2013 Dec; 4(2): 331– 345. doi: 10. 1007/s 13300 -013 -0030 -2 19

Which SU Should Be Used? § Among SUs, § based on evidence from observational studies showing lower cardiovascular risks of Gliclazide over other Sus , o A lower incidence of hypoglycemic events, o o Less weight gain.

Gliclazide Pharmacokinetics § § § Well absorption Peak plasma concentration occurring within 4 -6 hrs Extensively (94%) bound to plasma proteins Half-life: Approximately 10– 12 hrs Metabolized in the liver to inactive metabolites: o. The metabolites have no significant hypoglycaemic effect. o. Metabolites are primarily eliminated via the kidneys (60 -70%), or the faeces (10 -20%). o<5% of the dose is excreted unchanged in the urine. § Use in pregnancy & lactation: o Category C Gliclazide 80 mg: Datapharm Communications Ltd ; Summary of product characteristics 2013. Gliclazide 80 mg: Arrow Pharmaceuticals NZ Limited Fact Sheet 2006.

Gliclazide: Posology & Method of Administration § The total daily dose may vary from 40 to 320 mg taken orally. § The dose should be adjusted according to the individual patient’s response, commencing with 40– 80 mg daily (½ - 1 tablet) and increasing until adequate control is achieved. § A single dose should not exceed 160 mg (2 tablets). § If > 160 mg/d are required, tablets should be taken twice daily according to the main meals of the day. Starting Dose Maximum Recommended Dose 40 -80 mg/day (1/2 -1 of 80 mg tablet) 320 mg/day Summary of product characteristics; Gliclazide 80 mg

Gliclazide MR: Dosage and administration § The daily dose of GLICLAZIDE MR may vary from 30 -120 mg (1 -4 tablets) once daily. § The recommended starting dose of GLICLAZIDE MR is 1 tablet per day (30 mg), even in elderly patients (> 65 years old). § A single daily dose provides effective blood glucose control. § Dose adjustment should be carried out in steps of 30 mg • Each step should last for at least two weeks. Administration § It is recommended that the medication be taken at breakfast time. The tablets should be swallowed whole and must not be chewed or crushed. § Previously untreated patients should commence with a dose of 30 mg. § GLICLAZIDE MR can replace gliclazide 80 mg immediate release tablets. § GLICLAZIDE MR can replace an antidiabetic treatment without any transitional period. PRODUCT MONOGRAPH; Gliclazide Modified Release Tablets 30 mg, AA PHARMA INC. , June 25, 2010

Diamicron MR once daily is effective and well tolerated in T 2 DM: A double-blind, randomized, multinational study Drouin P. J Diabetes Complications. 2000; 14(4): 185 -91.

Jørgensen et al. Cardiovascular Diabetology 2010; 9: 54.

Zeller et al. , J Clin Endocrinol Metab. 2010; 95(11): 4993– 5002

Review of Gliclazide MR effects § Controlling blood glucose, maintaining this control over the long term, and preventing the development of microvascular and macrovascular complications are main challenges in type 2 diabetes management. § Gliclazide MR has an unmatched level of clinical evidence demonstrating powerful glycemic efficacy maintained over the long term, unique ESKD prevention, cardiovascular safety, and an optimal safety profile in terms of hypoglycemia and weight gain. § It is postulated that oxidative stress, which is abnormally high in T 2 DM, has a negative impact on progression of diabetes by reducing β-cell function, and the development of diabetic nephropathy and cardiovascular disease, particularly by increasing atherosclerosis. § Gliclazide MR, thanks to its unique chemical structure, reduces oxidative stress, increasing the resistance of LDL to oxidation and slowing the progression of atherosclerosis in T 2 DM. Ruiz M. Diamicron MR: the secretagogue with clinical benefits beyond insulin secretion. MEDICOGRAPHIA, 2013; 35(1): 81 -9.
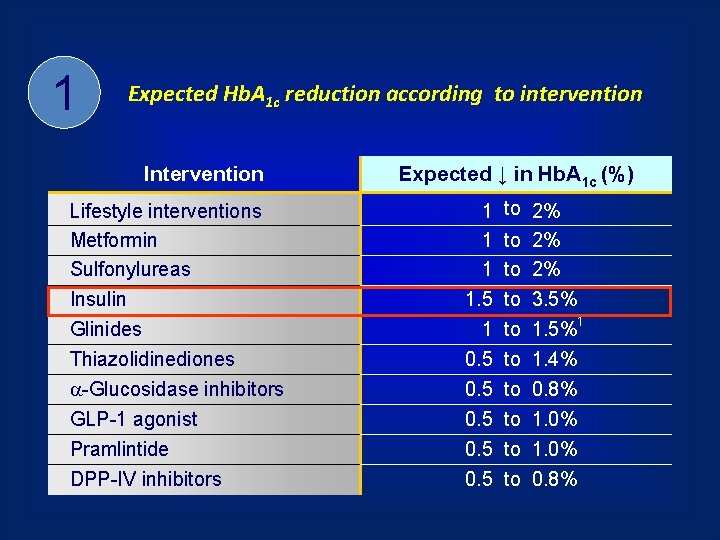
1 Expected Hb. A 1 c reduction according to intervention Intervention Lifestyle interventions Metformin Sulfonylureas Insulin Glinides Thiazolidinediones -Glucosidase inhibitors GLP-1 agonist Pramlintide DPP-IV inhibitors Expected ↓ in Hb. A 1 c (%) 1 1. 5 1 0. 5 0. 5 to 2% to 3. 5% 1 to 1. 5% to 1. 4% to 0. 8% to 1. 0% to 0. 8%
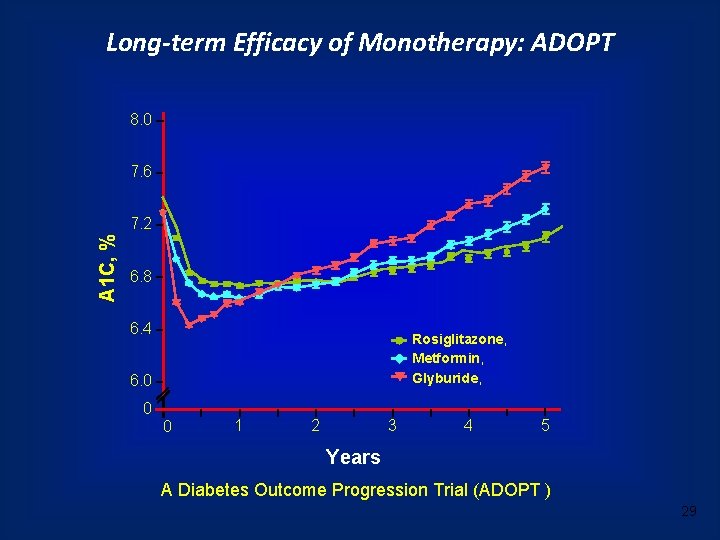
Long-term Efficacy of Monotherapy: ADOPT 8. 0 7. 6 A 1 C, % 7. 2 6. 8 6. 4 Rosiglitazone, Metformin, Glyburide, 6. 0 0 0 1 2 3 4 5 Years A Diabetes Outcome Progression Trial (ADOPT ) 29
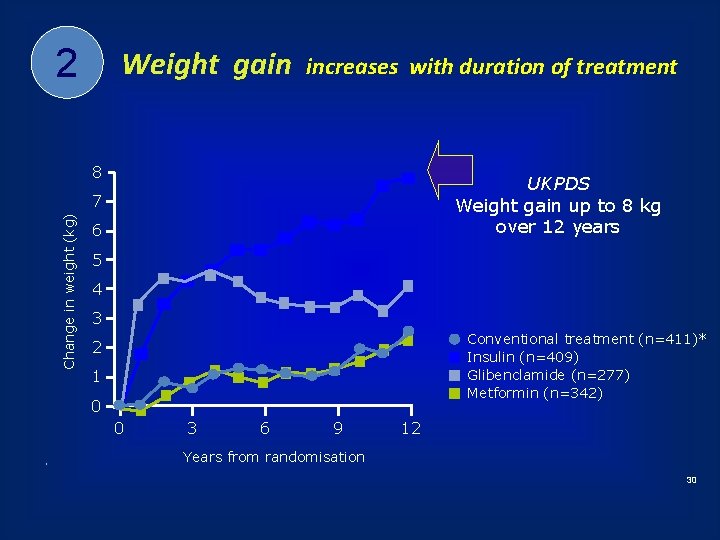
2 Weight gain increases with duration of treatment 8 UKPDS Weight gain up to 8 kg over 12 years Change in weight (kg) 7 6 5 4 3 Conventional treatment (n=411)* Insulin (n=409) Glibenclamide (n=277) Metformin (n=342) 2 1 0 0 3 6 9 12 Years from randomisation 30
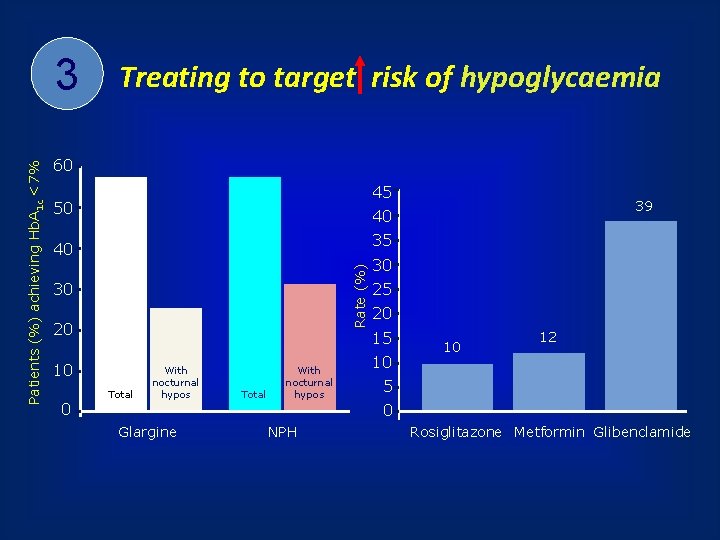
Treating to target risk of hypoglycaemia 60 45 50 35 40 30 20 10 0 39 40 Rate (%) Patients (%) achieving Hb. A 1 c <7% 3 Total With nocturnal hypos 30 25 20 15 10 10 12 5 0 Glargine NPH Rosiglitazone Metformin Glibenclamide

• A 52 y/o male comes in with HBA 1 C 8, FBS 180, 2 hrpp 250, Cr. 1. 8, Wt : 56 Kg. • • • What are your recommendations? Repaglinide Glutazone Metformin Insulin Combination therapy 32
What are the Incretins ? Peptides produced by the intestines and released in response to meals: 1 - Glucose-dependent insulinotropic peptide ( GIP ) 42 AA , K-cells of proximal duodenum Brown & Pederson : 1970 Brown , Ross & Watson : 1973
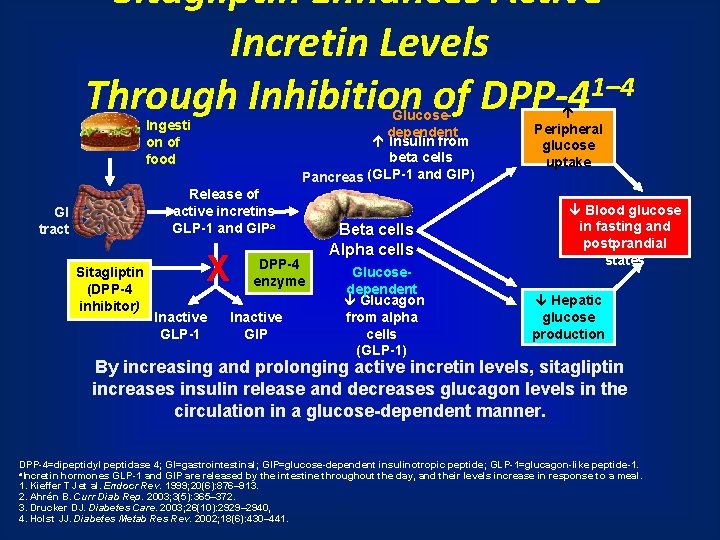
Sitagliptin Enhances Active Incretin Levels 1– 4 Through Inhibition of DPP-4 Glucosedependent Insulin from beta cells Pancreas (GLP-1 and GIP) Ingesti on of food Release of active incretins GLP-1 and GIPa GI tract Sitagliptin (DPP-4 inhibitor) X Inactive GLP-1 DPP-4 enzyme Inactive GIP Beta cells Alpha cells Glucosedependent Glucagon from alpha cells (GLP-1) Peripheral glucose uptake Blood glucose in fasting and postprandial states Hepatic glucose production By increasing and prolonging active incretin levels, sitagliptin increases insulin release and decreases glucagon levels in the circulation in a glucose-dependent manner. DPP-4=dipeptidyl peptidase 4; GI=gastrointestinal; GIP=glucose-dependent insulinotropic peptide; GLP-1=glucagon-like peptide-1. a. Incretin hormones GLP-1 and GIP are released by the intestine throughout the day, and their levels increase in response to a meal. 1. Kieffer TJ et al. Endocr Rev. 1999; 20(6): 876– 913. 2. Ahrén B. Curr Diab Rep. 2003; 3(5): 365– 372. 3. Drucker DJ. Diabetes Care. 2003; 26(10): 2929– 2940, 4. Holst JJ. Diabetes Metab Res Rev. 2002; 18(6): 430– 441.
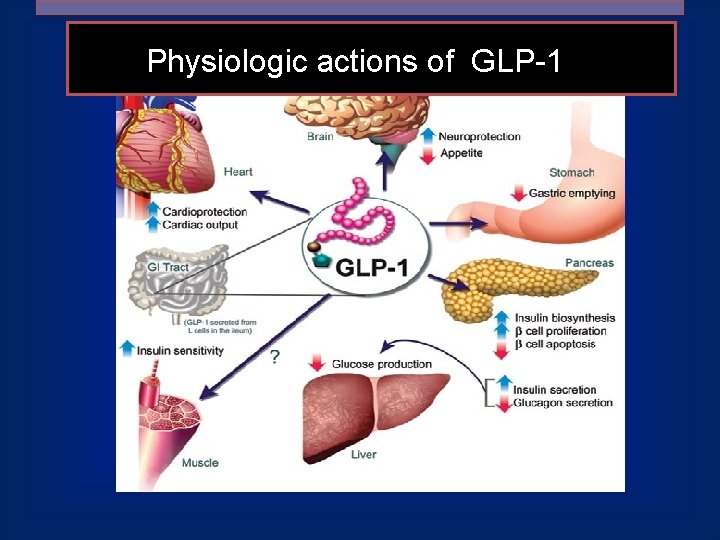
Physiologic actions of GLP-1
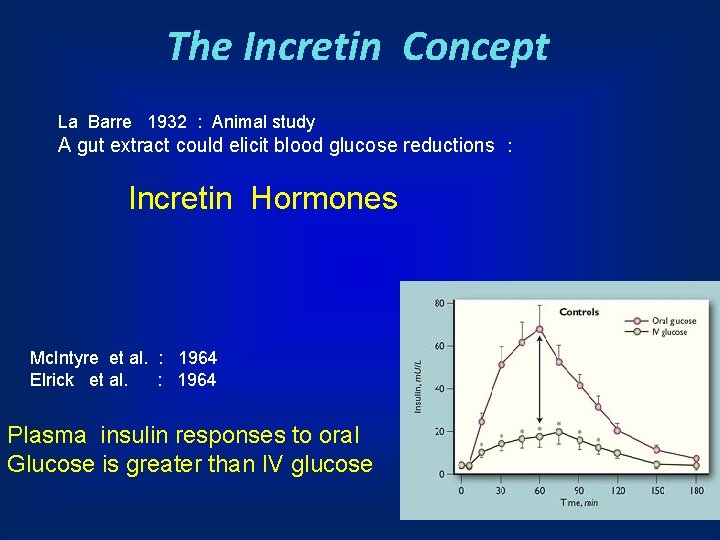
The Incretin Concept La Barre 1932 : Animal study A gut extract could elicit blood glucose reductions : Incretin Hormones Mc. Intyre et al. : 1964 Elrick et al. : 1964 Plasma insulin responses to oral Glucose is greater than IV glucose
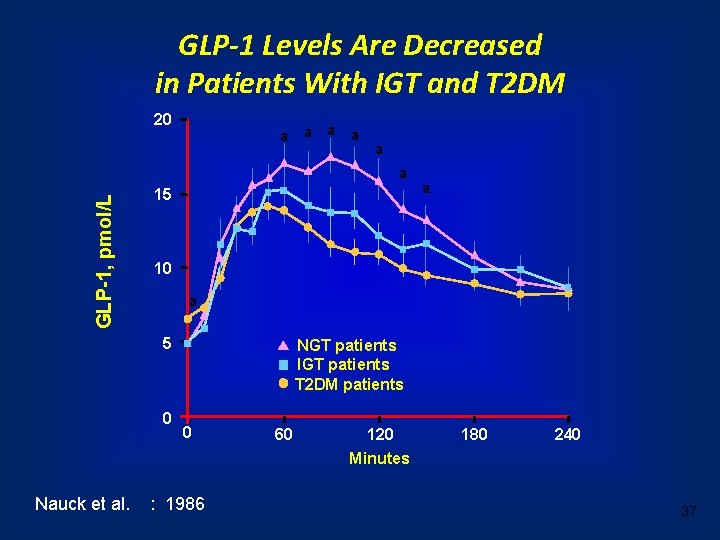
GLP-1 Levels Are Decreased in Patients With IGT and T 2 DM 20 a a a GLP-1, pmol/L a 15 10 a 5 0 Nauck et al. a NGT patients IGT patients T 2 DM patients 0 : 1986 60 120 Minutes 180 240 37
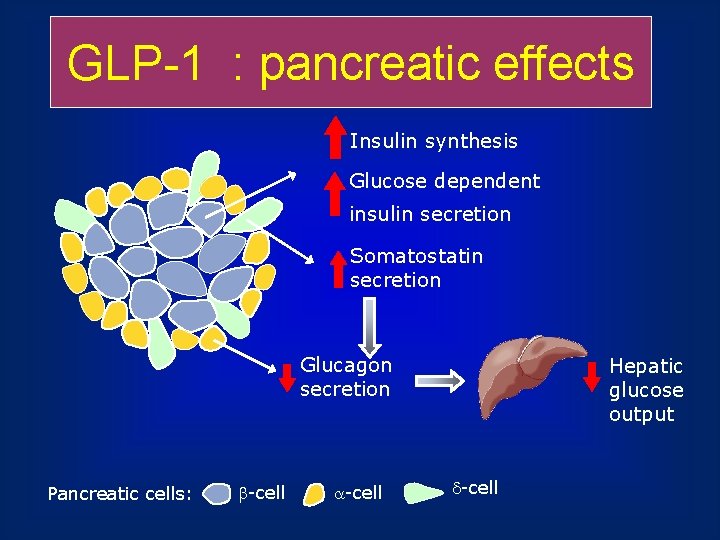
GLP-1 : pancreatic effects Insulin synthesis Glucose dependent insulin secretion Somatostatin secretion Glucagon secretion Pancreatic cells: -cell Hepatic glucose output -cell
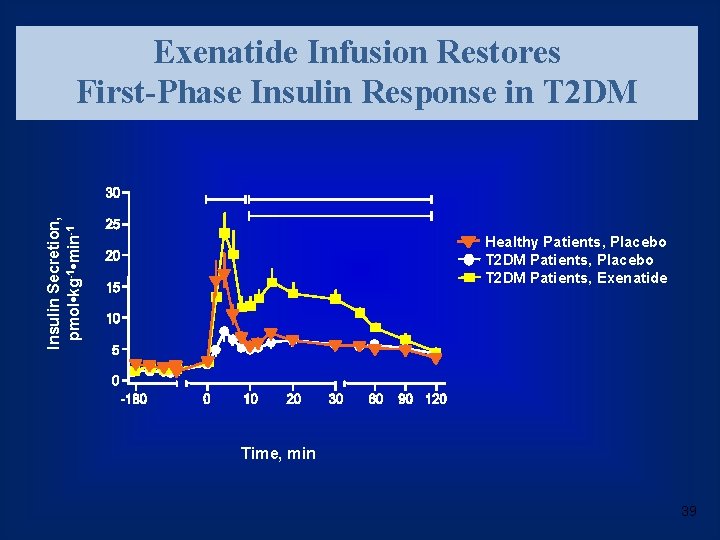
Insulin Secretion, pmol • kg-1 • min-1 Exenatide Infusion Restores First-Phase Insulin Response in T 2 DM Healthy Patients, Placebo T 2 DM Patients, Exenatide Time, min 39

DPP-4 Inhibitors Sitagliptin (Januvia ) Saxagliptin ( Onglysa ) Vildagliptin (Galvus ) Alogliptin

• Sitagliptin and vildagliptin are the first agents in this class to have received FDA approval. • Incidence of adverse reactions was reported to be very low in a pooled safety data from 5141 patients. ADA meeting, Chicago, June 2007. • They are indicated as monotherapy and in combination with metformin, thiazolidinediones and insulin. • They look to be at least weight neutral

Conclusions: There was a reduction in all-cause mortality for patients treated with metformin combined with DPP-4 i versus metformin plus SU, and a similar trend for MACE. 42 6/4/20 21

What are the Recommendations? 43
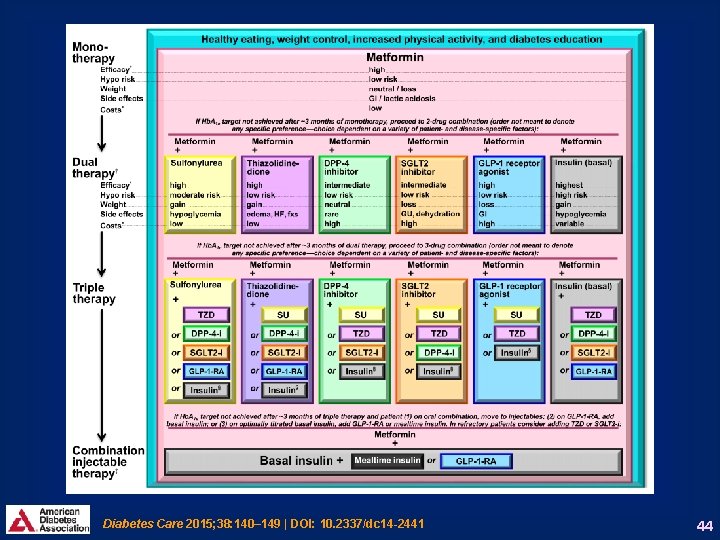
Diabetes Care 2015; 38: 140– 149 | DOI: 10. 2337/dc 14 -2441 44
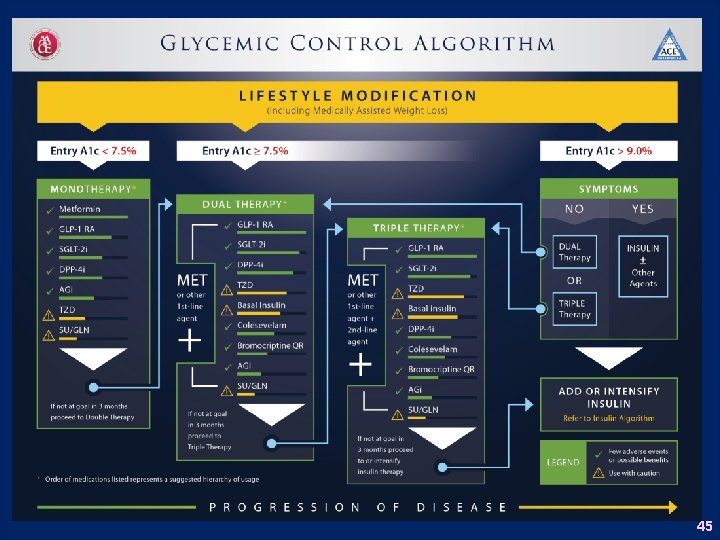
45

What are the Recommendations? 46

Initial Drug Therapy Metformin • If not contraindicated and if tolerated, is the preferred initial pharmacological agent for type 2 diabetes. A Diabetes Care 2015; 38(Suppl. 1): S 42 | DOI: 10. 2337/dc 15 -S 009 47

Initial Drug Therapy Patients with a high baseline Hb. A 1 c (≥ 9. 0%) • Low probability of achieving a near normal target with monotherapy • It is justified to start directly with a combination of two noninsulin agents or with insulin itself in this circumstance Diabetes Care 2015; 38: 145 | DOI: 10. 2337/dc 14 -2441 48

Initial Drug Therapy If BS> 300– 350 mg/d. L or Hb. A 1 c≥ 10. 0 – 12. 0% consider combination injectable therapy • especially if patient is symptomatic or if catabolic features (weight loss, ketosis) are present, in which case basal insulin + mealtime insulin is the preferred initial regimen. Diabetes Care 2015; 38: 145 | DOI: 10. 2337/dc 14 -2441 49

Comparative Effectiveness Meta-analyses • Each new class of noninsulin agents added to initial therapy lowers A 1 C around 0. 9 – 1. 1% Bennett WL, Maruthur NM, Singh S, et al. Comparative effectiveness and safety of medications for type 2 diabetes: an update including new drugs and 2 - drug combinations. Ann Intern Med 2011; 154: 602– 613. 50

Oral Anti-hyperglycemic agents for use in T 2 DM † A 1 C percentage/relative reduction expected when agent from this class is added to metformin therapy S. A. Imran et al. Canadian Diabetes Association. Can J Diabetes 2013; 37: S 62 51

Oral Anti-hyperglycemic agents for use in T 2 DM † A 1 C percentage/relative reduction expected when agent from this class is added to metformin therapy S. A. Imran et al. Canadian Diabetes Association. Can J Diabetes 2013; 37: S 63 52

Oral Anti-hyperglycemic agents for use in T 2 DM † A 1 C percentage/relative reduction expected when agent from this class is added to metformin therapy S. A. Imran et al. Canadian Diabetes Association. Can J Diabetes 2013; 37: S 63 53
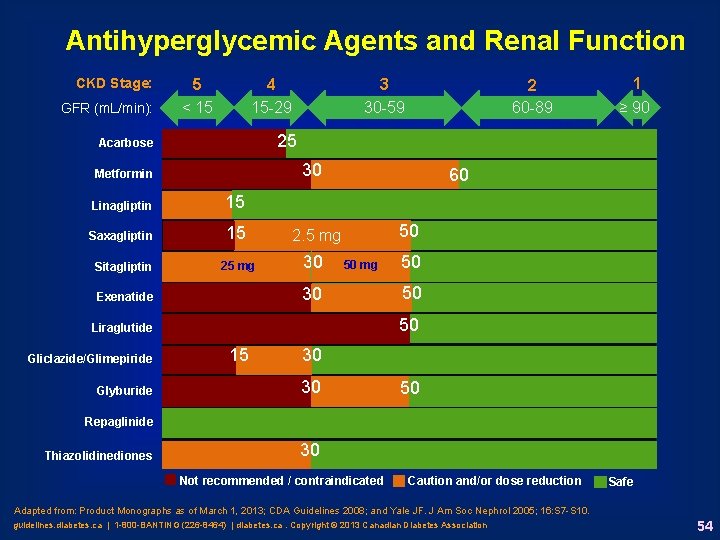
Antihyperglycemic Agents and Renal Function CKD Stage: GFR (m. L/min): 5 < 15 4 15 -29 3 30 -59 30 Metformin Linagliptin 15 Saxagliptin 15 2. 5 mg Sitagliptin 25 mg 30 ≥ 90 60 50 50 mg 30 Exenatide 50 50 50 Liraglutide Glyburide 1 25 Acarbose Gliclazide/Glimepiride 2 60 -89 15 30 30 50 Repaglinide Thiazolidinediones 30 Not recommended / contraindicated Caution and/or dose reduction Adapted from: Product Monographs as of March 1, 2013; CDA Guidelines 2008; and Yale JF. J Am Soc Nephrol 2005; 16: S 7 -S 10. guidelines. diabetes. ca | 1 -800 -BANTING (226 -8464) | diabetes. ca. Copyright © 2013 Canadian Diabetes Association Safe 54
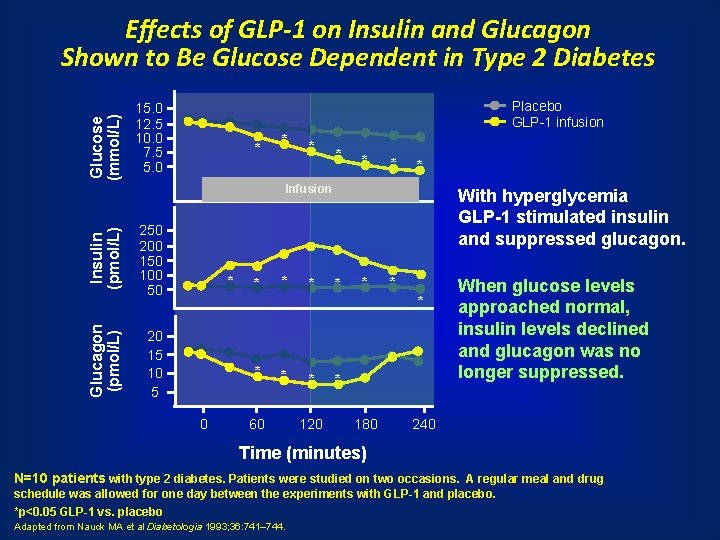
Glucose (mmol/L) Effects of GLP-1 on Insulin and Glucagon Shown to Be Glucose Dependent in Type 2 Diabetes Placebo GLP-1 infusion 15. 0 12. 5 10. 0 7. 5 5. 0 * * * * Glucagon (pmol/L) Insulin (pmol/L) Infusion 250 200 150 100 50 * * With hyperglycemia GLP-1 stimulated insulin and suppressed glucagon. * * 20 15 10 5 * 0 * 60 * 120 * 180 When glucose levels approached normal, insulin levels declined and glucagon was no longer suppressed. 240 Time (minutes) N=10 patients with type 2 diabetes. Patients were studied on two occasions. A regular meal and drug schedule was allowed for one day between the experiments with GLP-1 and placebo. *p<0. 05 GLP-1 vs. placebo Adapted from Nauck MA et al Diabetologia 1993; 36: 741– 744.

the Core Metabolic Defects of Type 2 Beta-Cell Insulin Diabetes Dysfunction Resistanc Sitagliptin e improves betacell function and increases insulin synthesis and release. 1 Sitagliptin reduces HGO through suppression of glucagon from alpha cells. 2 Hepatic Glucose Overproduction (HGO) 1. Aschner P et al. Diabetes Care. 2006; 29(12): 2632– 2637. 2. Data on file. 3. Abbasi F et al. Diabetes Care. 1998; 21(8): 1301– 1305. 4. Kirpichnikov D et al. Ann Intern Med. 2002; 137(1): 25– 33. 5. Zhou G et al. J Clin Invest. 2001; 108(8): 1167– 1174. Metformin has insulin - sensitizing properties. 3– 5 (Liver > Muscle, fat) Metformin decreases HGO by targeting the liver to decrease gluconeogenesis and glycogenolysis. 4

Mechanisms of Action of Major Oral Monotherapies Are Unable to Address the 3 Core Defects in Type 2 Diabetes Mechanisms of Action Oral Monotherapies Improves insulin secretion SUs Meglitinides TZDs Metformin α-Glucosidase Inhibitors DPP-4 Inhibitors Improves insulin resistance Lowers hepatic glucose production SUs=sulfonylureas; TZD=thiazolidinediones; DPP-4=dipeptidyl peptidase 4. Inzucchi SE. JAMA 2002; 287: 360– 372; Gallwitz B. Minerva Endocrinol. 2006; 31: 133– 147; Nathan DM et al. Diabetologia. 2006; 49: 1711– 1721.
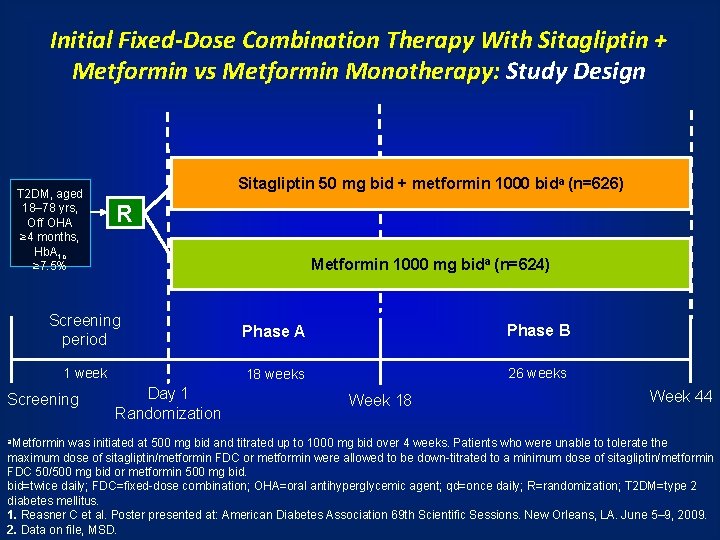
Initial Fixed-Dose Combination Therapy With Sitagliptin + Metformin vs Metformin Monotherapy: Study Design T 2 DM, aged 18– 78 yrs, Off OHA ≥ 4 months, Hb. A 1 c ≥ 7. 5% Sitagliptin 50 mg bid + metformin 1000 bida (n=626) R Metformin 1000 mg bida (n=624) Screening period Phase A Phase B 1 week 18 weeks 26 weeks Screening a. Metformin Day 1 Randomization Week 18 Week 44 was initiated at 500 mg bid and titrated up to 1000 mg bid over 4 weeks. Patients who were unable to tolerate the maximum dose of sitagliptin/metformin FDC or metformin were allowed to be down-titrated to a minimum dose of sitagliptin/metformin FDC 50/500 mg bid or metformin 500 mg bid=twice daily; FDC=fixed-dose combination; OHA=oral antihyperglycemic agent; qd=once daily; R=randomization; T 2 DM=type 2 diabetes mellitus. 1. Reasner C et al. Poster presented at: American Diabetes Association 69 th Scientific Sessions. New Orleans, LA. June 5– 9, 2009. 2. Data on file, MSD.
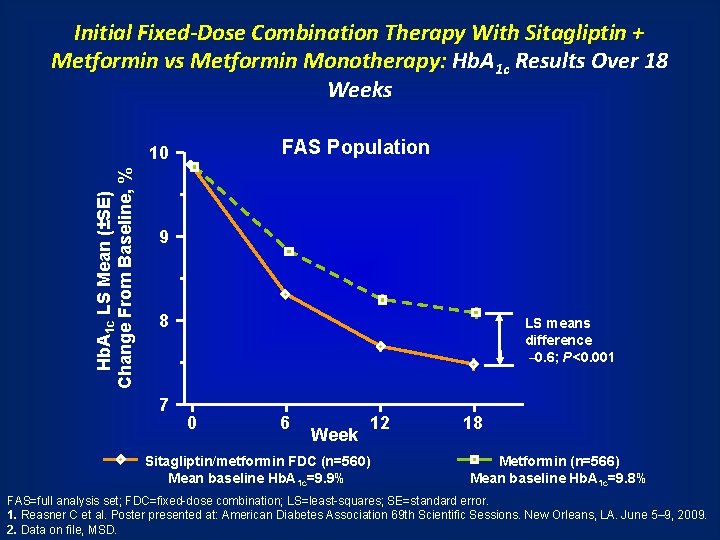
Initial Fixed-Dose Combination Therapy With Sitagliptin + Metformin vs Metformin Monotherapy: Hb. A 1 c Results Over 18 Weeks FAS Population Hb. A 1 c LS Mean (±SE) Change From Baseline, % 10 9 8 7 LS means difference – 0. 6; P<0. 001 0 6 Week 12 Sitagliptin/metformin FDC (n=560) Mean baseline Hb. A 1 c=9. 9% 18 Metformin (n=566) Mean baseline Hb. A 1 c=9. 8% FAS=full analysis set; FDC=fixed-dose combination; LS=least-squares; SE=standard error. 1. Reasner C et al. Poster presented at: American Diabetes Association 69 th Scientific Sessions. New Orleans, LA. June 5– 9, 2009. 2. Data on file, MSD.
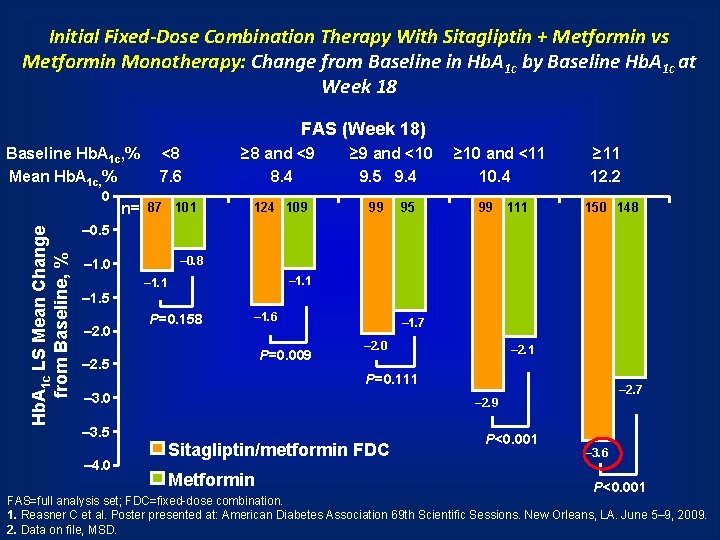
Initial Fixed-Dose Combination Therapy With Sitagliptin + Metformin vs Metformin Monotherapy: Change from Baseline in Hb. A 1 c by Baseline Hb. A 1 c at Week 18 FAS (Week 18) Baseline Hb. A 1 c, % Mean Hb. A 1 c, % Hb. A 1 c LS Mean Change from Baseline, % 0 <8 7. 6 n= 87 101 ≥ 8 and <9 8. 4 124 109 ≥ 9 and <10 9. 5 9. 4 99 95 ≥ 10 and <11 10. 4 99 111 ≥ 11 12. 2 150 148 – 0. 5 – 0. 8 – 1. 0 – 1. 1 – 1. 5 – 2. 0 P=0. 158 – 1. 6 P=0. 009 – 2. 5 – 1. 7 – 2. 0 P=0. 111 – 3. 0 – 3. 5 – 4. 0 – 2. 1 – 2. 7 – 2. 9 Sitagliptin/metformin FDC Metformin P<0. 001 – 3. 6 P<0. 001 FAS=full analysis set; FDC=fixed-dose combination. 1. Reasner C et al. Poster presented at: American Diabetes Association 69 th Scientific Sessions. New Orleans, LA. June 5– 9, 2009. 2. Data on file, MSD.

Sitagliptin Side effects • Rare: Acute pancreatitis was in 0. 1 per 100 patient-years. • Nasopharyngitis 1 -10 percent. • Hypersensitivity. . Rarely angioedema, Steven Johnson. • Hepatic enzyme elevation. • Worsening renal function. • HTN. • Headache 61
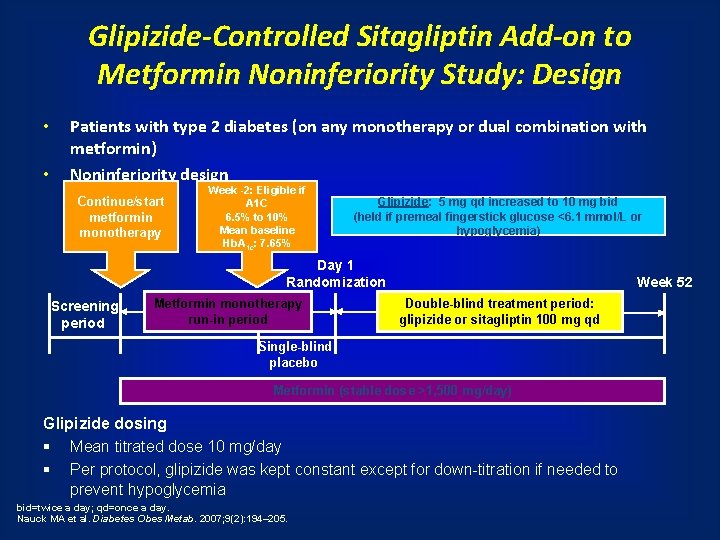
Glipizide-Controlled Sitagliptin Add-on to Metformin Noninferiority Study: Design • • Patients with type 2 diabetes (on any monotherapy or dual combination with metformin) Noninferiority design Continue/start metformin monotherapy Week -2: Eligible if A 1 C 6. 5% to 10% Mean baseline Hb. A 1 c: 7. 65% Glipizide: 5 mg qd increased to 10 mg bid (held if premeal fingerstick glucose <6. 1 mmol/L or hypoglycemia) Day 1 Randomization Screening period Metformin monotherapy run-in period Week 52 Double-blind treatment period: glipizide or sitagliptin 100 mg qd Single-blind placebo Metformin (stable dose >1, 500 mg/day) Glipizide dosing § Mean titrated dose 10 mg/day § Per protocol, glipizide was kept constant except for down-titration if needed to prevent hypoglycemia bid=twice a day; qd=once a day. Nauck MA et al. Diabetes Obes Metab. 2007; 9(2): 194– 205.
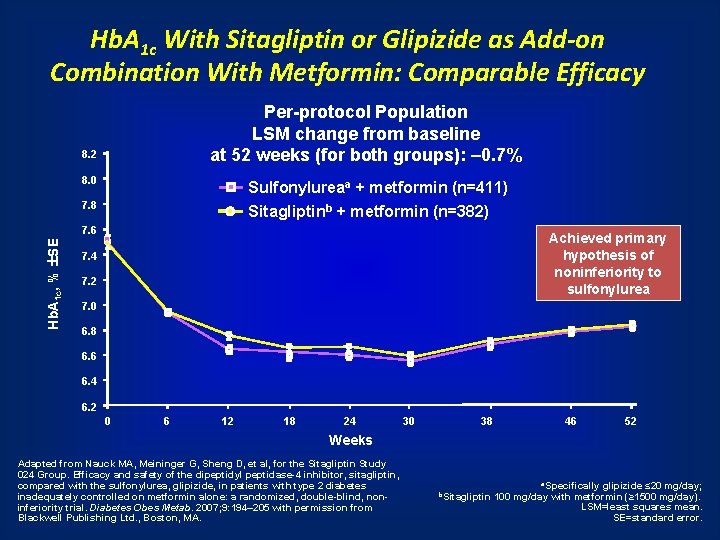
Hb. A 1 c With Sitagliptin or Glipizide as Add-on Combination With Metformin: Comparable Efficacy Per-protocol Population LSM change from baseline at 52 weeks (for both groups): – 0. 7% 8. 2 8. 0 Sulfonylureaa + metformin (n=411) Sitagliptinb + metformin (n=382) 7. 8 Hb. A 1 c, % ±SE 7. 6 Achieved primary hypothesis of noninferiority to sulfonylurea 7. 4 7. 2 7. 0 6. 8 6. 6 6. 4 6. 2 0 6 12 18 24 30 38 46 52 Weeks Adapted from Nauck MA, Meininger G, Sheng D, et al, for the Sitagliptin Study 024 Group. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor, sitagliptin, compared with the sulfonylurea, glipizide, in patients with type 2 diabetes inadequately controlled on metformin alone: a randomized, double-blind, noninferiority trial. Diabetes Obes Metab. 2007; 9: 194– 205 with permission from Blackwell Publishing Ltd. , Boston, MA. b. Sitagliptin a. Specifically glipizide ≤ 20 mg/day; 100 mg/day with metformin (≥ 1500 mg/day). LSM=least squares mean. SE=standard error.
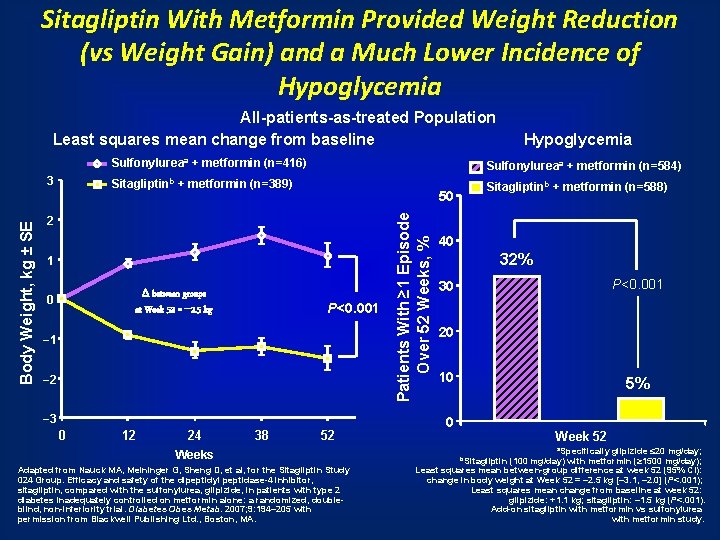
Sitagliptin With Metformin Provided Weight Reduction (vs Weight Gain) and a Much Lower Incidence of Hypoglycemia All-patients-as-treated Population Least squares mean change from baseline Sulfonylureaa + metformin (n=416) Sulfonylureaa + metformin (n=584) Sitagliptinb + metformin (n=389) Sitagliptinb + metformin (n=588) 50 2 1 between groups at Week 52 = – 2. 5 kg 0 P<0. 001 − 2 − 3 0 12 24 38 52 Weeks Adapted from Nauck MA, Meininger G, Sheng D, et al, for the Sitagliptin Study 024 Group. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor, sitagliptin, compared with the sulfonylurea, glipizide, in patients with type 2 diabetes inadequately controlled on metformin alone: a randomized, doubleblind, non-inferiority trial. Diabetes Obes Metab. 2007; 9: 194– 205 with permission from Blackwell Publishing Ltd. , Boston, MA. Patients With ≥ 1 Episode Over 52 Weeks, % Body Weight, kg ± SE 3 Hypoglycemia 40 32% P<0. 001 30 20 10 5% 0 Week 52 a. Specifically glipizide ≤ 20 mg/day; (100 mg/day) with metformin (≥ 1500 mg/day); Least squares mean between-group difference at week 52 (95% CI): change in body weight at Week 52 = – 2. 5 kg [– 3. 1, – 2. 0] (P<. 001); Least squares mean change from baseline at week 52: glipizide: +1. 1 kg; sitagliptin: – 1. 5 kg (P<. 001). Add-on sitagliptin with metformin vs sulfonylurea with metformin study. b. Sitagliptin

Sitagliptin or Glipizide as Add-on Combination With Metformin Post Hoc Analysis Patients Achieving Composite EP of The composite endpoint of A 1 C reduction (>0. 5%), no body weight gain and no hypoglycemia Over 52 Weeks Pre AD sente d A 2 010 Adapted from Thomas L. Seck et al. poster presented American Diabetes Association 70 th Scientific Sessions. Orlando, USA. June 25– 29, 2010.

Addition of Sitagliptin or Glimepiride in Patients Inadequately Controlled on Metformin: Study Design 1 Patients ≥ 18 years of age with T 2 DM on stable dose of metformin (≥ 1500 mg/day) for ≥ 12 weeks and Hb. A 1 c 6. 5%– 9. 0% Sitagliptin 100 mg qd R Glimepiride (started at 1 mg qd and up-titrated until week 18 as needed up to maximum dose of 6 mg qd) Continue stable dose of metformin Screening Period Week – 4 Single-blind Placebo Run-in Week – 2 Day 1 qd=once daily; R=randomization; T 2 DM=type 2 diabetes mellitus. 1. Arechavaleta R et al. Diabetes Obes Metab. 2011; 13(2): 160– 168. Double-blind Treatment Period Week 30
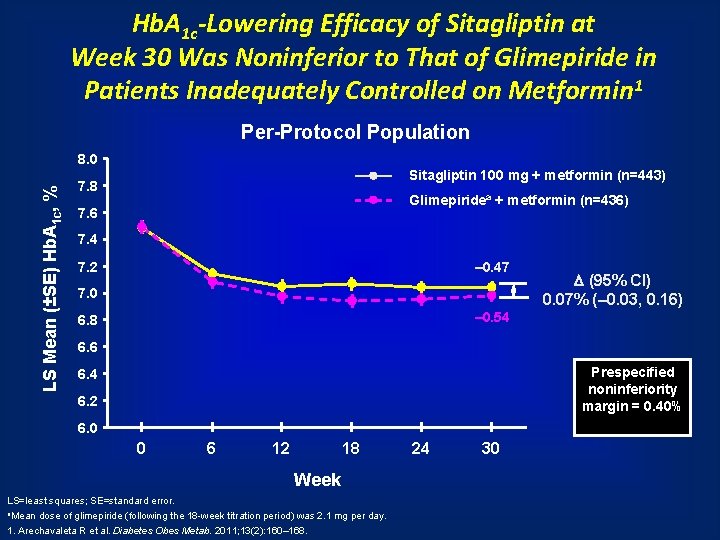
Hb. A 1 c-Lowering Efficacy of Sitagliptin at Week 30 Was Noninferior to That of Glimepiride in Patients Inadequately Controlled on Metformin 1 Per-Protocol Population LS Mean (±SE) Hb. A 1 c, % 8. 0 Sitagliptin 100 mg + metformin (n=443) 7. 8 Glimepiridea + metformin (n=436) 7. 6 7. 4 7. 2 – 0. 47 7. 0 (95% CI) 0. 07% (– 0. 03, 0. 16) – 0. 54 6. 8 6. 6 Prespecified noninferiority margin = 0. 40% 6. 4 6. 2 6. 0 0 6 12 18 Week LS=least squares; SE=standard error. a. Mean dose of glimepiride (following the 18 -week titration period) was 2. 1 mg per day. 1. Arechavaleta R et al. Diabetes Obes Metab. 2011; 13(2): 160– 168. 24 30
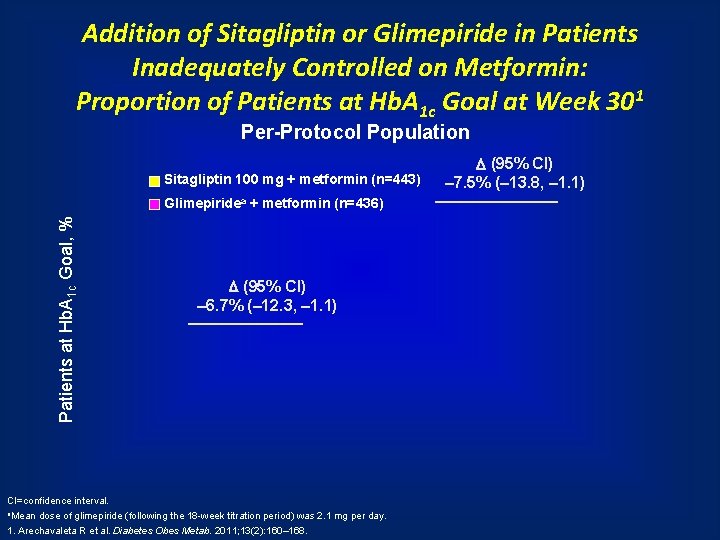
Addition of Sitagliptin or Glimepiride in Patients Inadequately Controlled on Metformin: Proportion of Patients at Hb. A 1 c Goal at Week 301 Per-Protocol Population Sitagliptin 100 mg + metformin (n=443) Patients at Hb. A 1 c Goal, % Glimepiridea + metformin (n=436) (95% CI) – 6. 7% (– 12. 3, – 1. 1) CI=confidence interval. a. Mean dose of glimepiride (following the 18 -week titration period) was 2. 1 mg per day. 1. Arechavaleta R et al. Diabetes Obes Metab. 2011; 13(2): 160– 168. (95% CI) – 7. 5% (– 13. 8, – 1. 1)
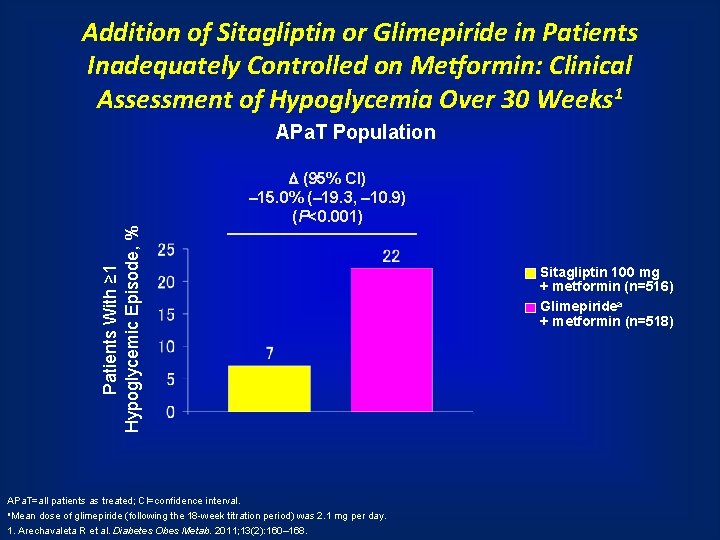
Addition of Sitagliptin or Glimepiride in Patients Inadequately Controlled on Metformin: Clinical Assessment of Hypoglycemia Over 30 Weeks 1 Patients With ≥ 1 Hypoglycemic Episode, % APa. T Population (95% CI) – 15. 0% (– 19. 3, – 10. 9) (P<0. 001) APa. T=all patients as treated; CI=confidence interval. a. Mean dose of glimepiride (following the 18 -week titration period) was 2. 1 mg per day. 1. Arechavaleta R et al. Diabetes Obes Metab. 2011; 13(2): 160– 168. Sitagliptin 100 mg + metformin (n=516) Glimepiridea + metformin (n=518)
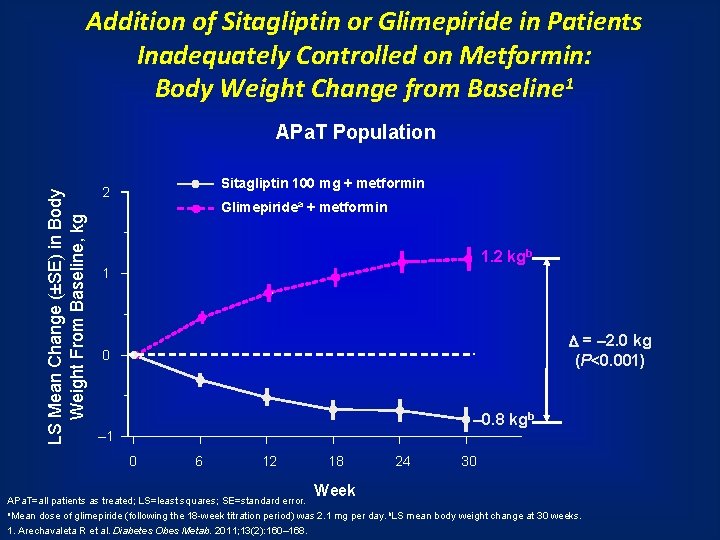
Addition of Sitagliptin or Glimepiride in Patients Inadequately Controlled on Metformin: Body Weight Change from Baseline 1 LS Mean Change (±SE) in Body Weight From Baseline, kg APa. T Population Sitagliptin 100 mg + metformin 2 Glimepiridea + metformin 1. 2 kgb 1 = – 2. 0 kg (P<0. 001) 0 – 0. 8 kgb – 1 0 6 12 18 Week 24 30 APa. T=all patients as treated; LS=least squares; SE=standard error. a. Mean dose of glimepiride (following the 18 -week titration period) was 2. 1 mg per day. b. LS mean body weight change at 30 weeks. 1. Arechavaleta R et al. Diabetes Obes Metab. 2011; 13(2): 160– 168.

CASE - 1 : A 48 -year-old woman, presents with polyuria and polydipsia during the past 3 months. She is overweight and complains of feeling tired all the time. FBS = 245 mg/dl HDL = 30 mg/d. L LDL = 170 mg/d. L. A 1 c = 9. 4%. which of the following is the most appropriate intervention that should be initiated as the first step in treating new-onset type 2 diabetes mellitus (T 2 DM)? 1 - Basal insulin 2 - Lifestyle and metformin 3 - Lifestyle and metformin and sulfonylurea 4 - Lifestyle and metformin and basal insulin

CASE - 2 : A 35 -y old woman with T 2 DM has been on the maximum tolerated dose of Metformin since 8 months ago. Over the past 7 months, her postdinner blood glucose level rose to 200 mg/d. L, and her A 1 c level is now at 7. 7%. She has been compliant with her medications and has continued to follow a diet that has resulted in the loss of almost 6 kg since diagnosis. She states that she wants to lose more weight because it has helped her self-esteem. Which of the following is the next most appropriate intervention for her to help achieve glycemic control? 1 - Add a glucagon-like peptide (GLP)-1 receptor agonist 2 - Add a sulfonylurea 3 - Add a thiazolidinedione (TZD) 4 - Add a dipeptidyl peptidase (DPP)-4 inhibitor 5 - Add basal insulin
- Slides: 72