1 MIGRATORY INSERTION DEINSERTION AND BETA HYDRIDE ELEMINATION

1 MIGRATORY INSERTION, DEINSERTION AND BETA HYDRIDE ELEMINATION DR SAADIA RASHID TARIQ TENURED ASSOCIATE PROFESSOR , DEPTT. CHEMISTRY, LCWU

2 MIGRATORY INSERTION, DEINSERTION AND BETA HYDRIDE ELEMINATION An insertion reaction involves the formal insertion of one ligand (usually unsaturated) into another metal-ligand bond on the same complex. The generic reaction is shown below, where U = an unsaturated ligand: U = CO, C 2 H 4, C 2 R 2, NO, CR 2, CNR, RCN, O 2, CO 2 X = H, alkyl, aryl, OR, NR 2 The net result is a decrease in the coordination number by one, along with the formation of a new U-X bond. The reverse of this reaction is generally called a deinsertion. When the deinserting group is an olefin, this has the special name of beta-hydride elimination.
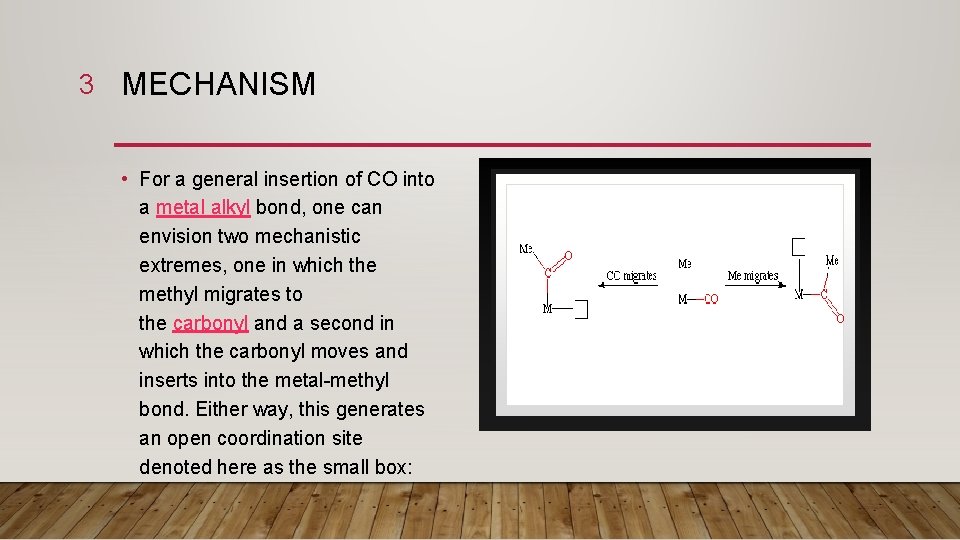
3 MECHANISM • For a general insertion of CO into a metal alkyl bond, one can envision two mechanistic extremes, one in which the methyl migrates to the carbonyl and a second in which the carbonyl moves and inserts into the metal-methyl bond. Either way, this generates an open coordination site denoted here as the small box:

4 • This subtle but important difference was studied by Calderazzo who examined the reverse reaction, deinsertion of CO from a metal acyl complex. By the Principle of Microscopic Reversibility, the insertion and deinsertion must follow the same mechanistic route, only in different directions. • By labeling one of the carbonyls cis to the acetyl group with 13 CO, one can differentiate the two possible mechanisms. If the CO moves during the deinsertion, then it can only move to a cis position, displacing another CO in the process. As there are four cis CO's and only one of them is labeled with 13 CO, then we would expect to remove the labeled carbonyl 25% of the time.
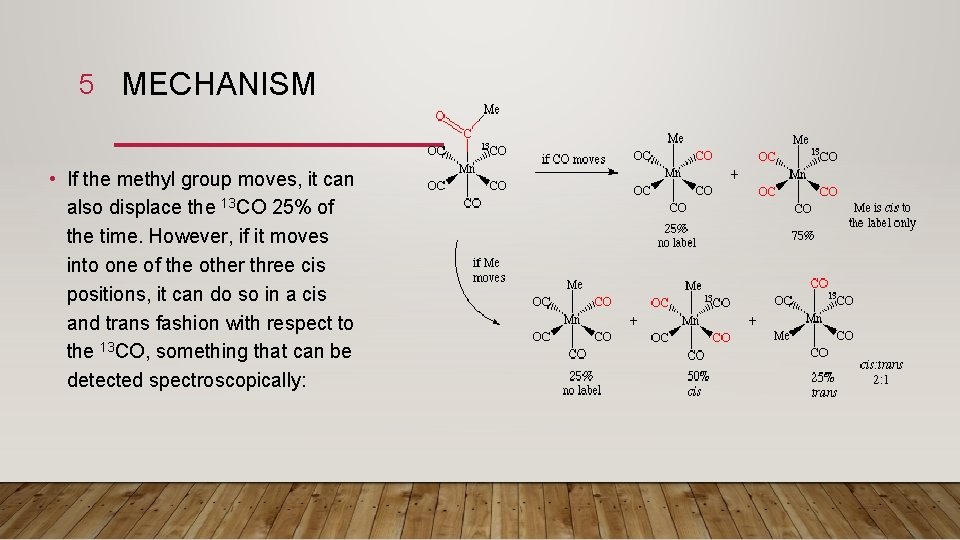
5 MECHANISM • If the methyl group moves, it can also displace the 13 CO 25% of the time. However, if it moves into one of the other three cis positions, it can do so in a cis and trans fashion with respect to the 13 CO, something that can be detected spectroscopically:

6 MECHANISM • The experimental result, a 2: 1 cis: trans ratio, is that the methyl group moves. For this reason, insertion reactions are sometimes called migratory insertion reactions. Of course, not all insertions have to take place by this mechanism. • For example, here's a case by Flood in which both mechanisms appear to operate! This terrific example uses an optically resolved transition metal complex to determine which group moved. The mechanism differs depending on the solvent used for the reaction:
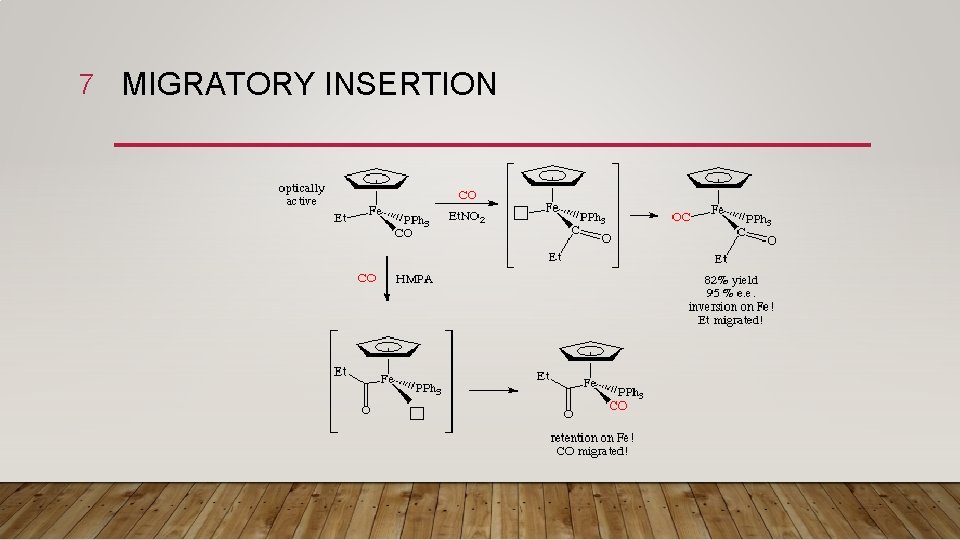
7 MIGRATORY INSERTION
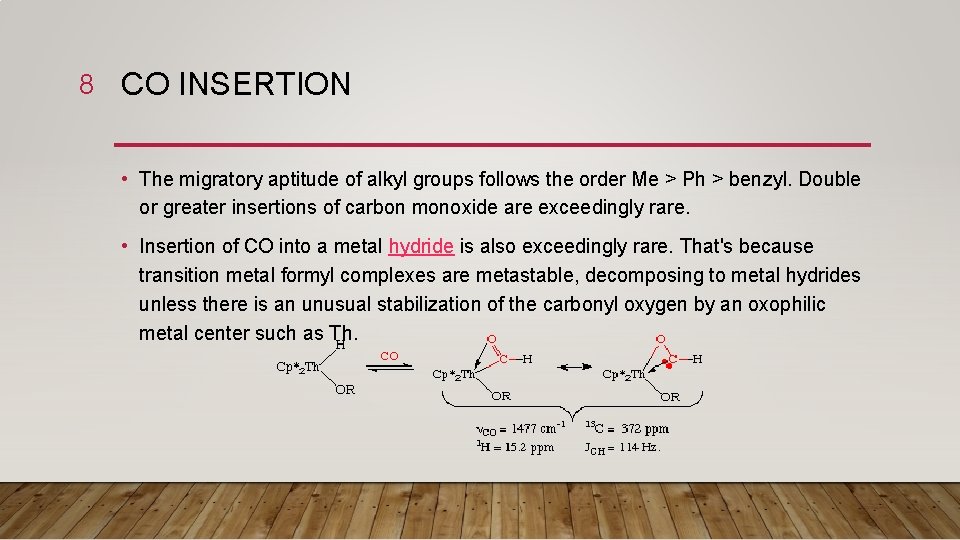
8 CO INSERTION • The migratory aptitude of alkyl groups follows the order Me > Ph > benzyl. Double or greater insertions of carbon monoxide are exceedingly rare. • Insertion of CO into a metal hydride is also exceedingly rare. That's because transition metal formyl complexes are metastable, decomposing to metal hydrides unless there is an unusual stabilization of the carbonyl oxygen by an oxophilic metal center such as Th.

9 CO INSERTION • Solvent can play a role in migratory insertions. This was studied first by Basolo who suggested that direct attack of solvent at the metal center could induce insertion. This was later confirmed by Bergman, who studied the steric effect of solvent on insertion in a Cp-Mo system.
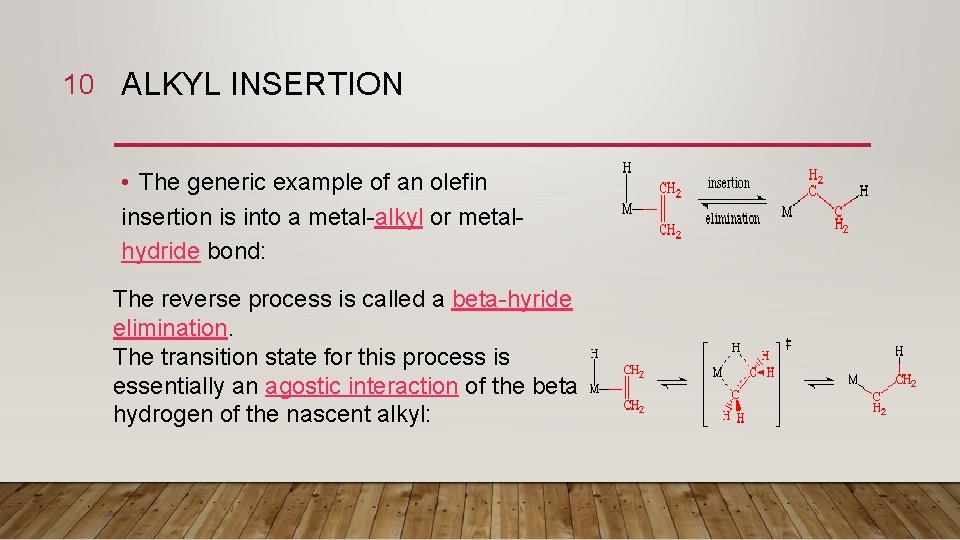
10 ALKYL INSERTION • The generic example of an olefin insertion is into a metal-alkyl or metalhydride bond: The reverse process is called a beta-hyride elimination. The transition state for this process is essentially an agostic interaction of the beta hydrogen of the nascent alkyl:
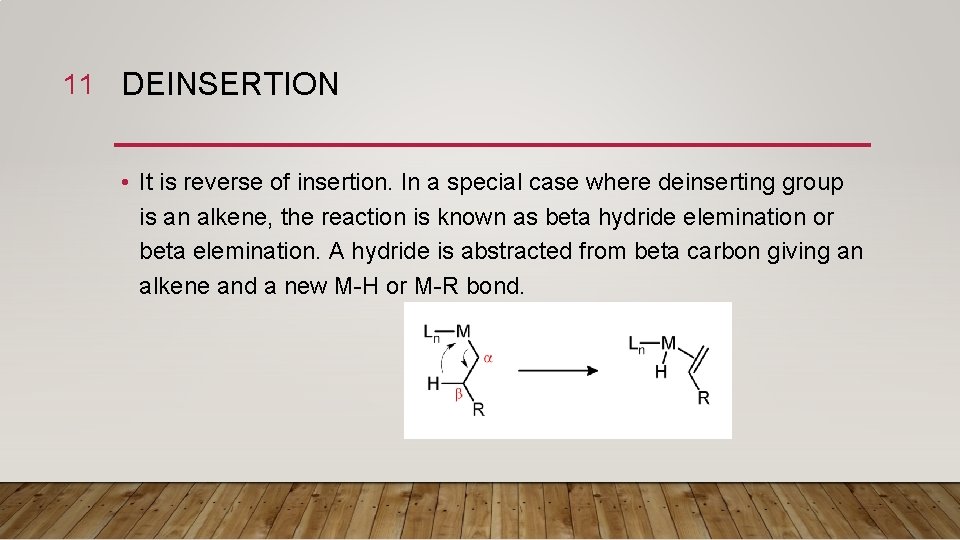
11 DEINSERTION • It is reverse of insertion. In a special case where deinserting group is an alkene, the reaction is known as beta hydride elemination or beta elemination. A hydride is abstracted from beta carbon giving an alkene and a new M-H or M-R bond.

12 BETA ELEMINATION • The metal gains a new ligand the electron count of complex is increased by two units. • Insertion creates a vacant site on the metal while beta elemination requires one and it must be cis to the eleminating group.

13 Thanks Questions?
- Slides: 13