1 Microwave Spectra and Structures of H 2



![Comparison of Nuclear Quadrupole Coupling Constants [ (63 Cu)] [ / MHz (35 Cl)] Comparison of Nuclear Quadrupole Coupling Constants [ (63 Cu)] [ / MHz (35 Cl)]](https://slidetodoc.com/presentation_image_h2/011e3db4d8b01610f16fbcab5f763b80/image-12.jpg)

- Slides: 13

1 Microwave Spectra and Structures of H 2 S-Cu. Cl and H 2 S-Ag. Cl by Nicholas R. Walker, David Wheatley, Anthony C. Legon 64 th OSU International Symposium on Molecular Spectroscopy 25 th June, 2009.

Introduction • Bonding between sulfur and copper is recurring motif of inorganic chemistry. Explore and extend the range of LMX species that can be generated using a laser ablation source. • Ziurys and co-workers studied Cu. SH and many other metal hydrosulfides. [Janczyk, A. ; Walter, S. K. ; Ziurys, L. M. ; Chem. Phys. Lett. 401 211 -216 (2005)] • Previous studies by laser ablation have explored OC-MX, Ar-MX, Kr-MX, Xe-MX; (Gerry and co-workers), N 2 -Cu. F (Bristol). • Provides opportunity to compare molecular geometry of H 2 S-MCl with H 2 S-HCl, H 2 S-ICl, H 2 S-Br. Cl studied by Legon and co-workers.

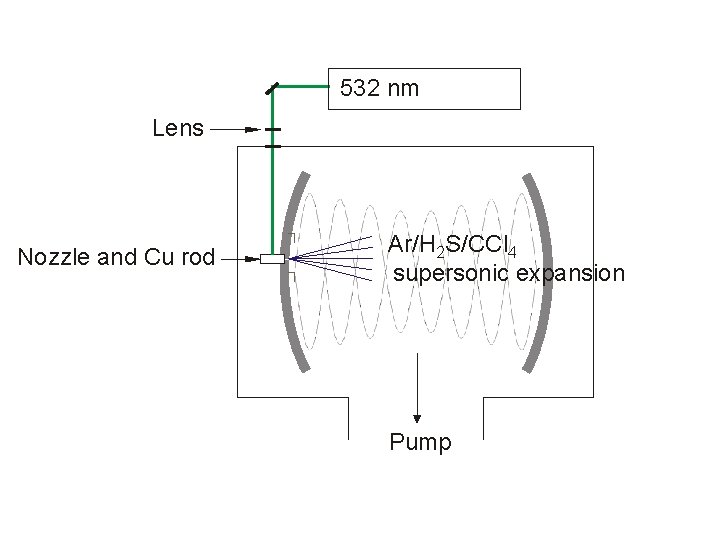
Ar/H 2 S/CCl 4

532 nm Lens Nozzle and Cu rod Ar/H 2 S/CCl 4 supersonic expansion Pump

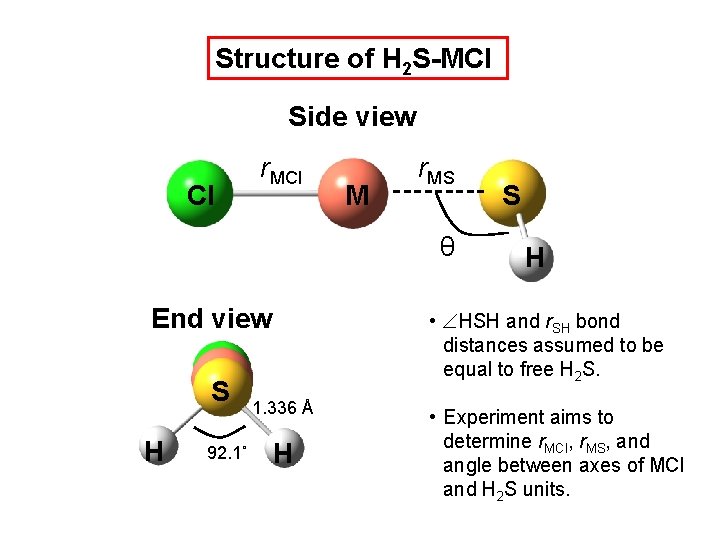
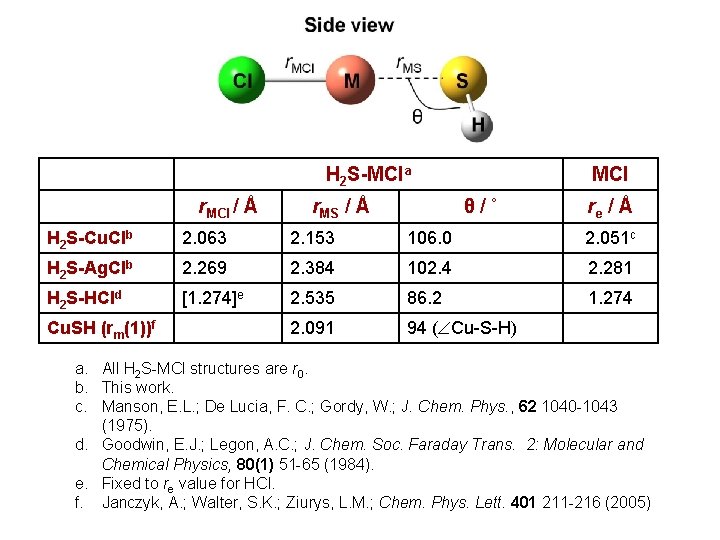
Structure of H 2 S-MCl Side view Cl r. MCl M r. MS θ End view S H 92. 1˚ S H • HSH and r. SH bond distances assumed to be equal to free H 2 S. 1. 336 Å H • Experiment aims to determine r. MCl, r. MS, and angle between axes of MCl and H 2 S units.

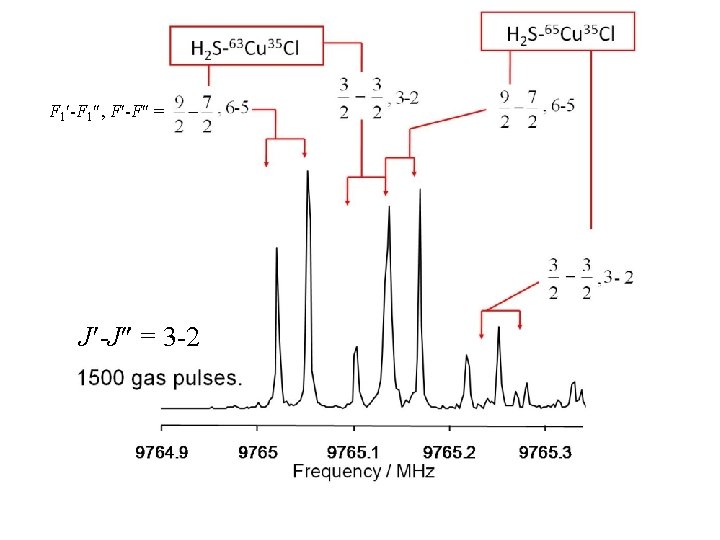
F 1 -F 1 , F -F = J -J = 3 -2

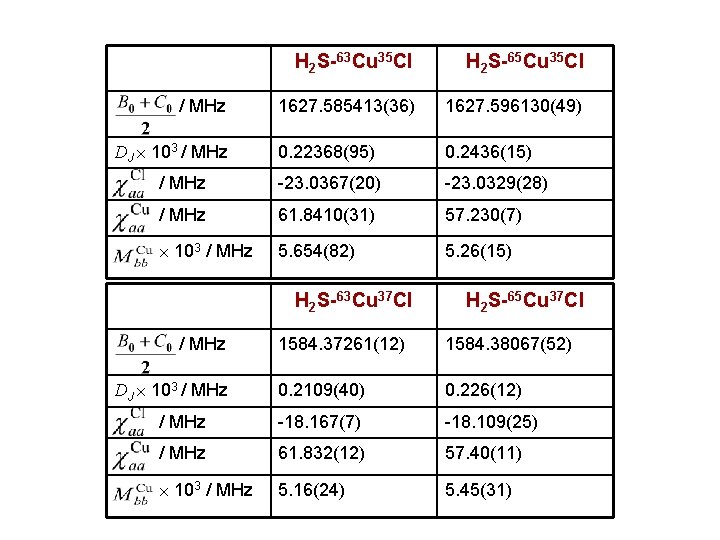
H 2 S-63 Cu 35 Cl H 2 S-65 Cu 35 Cl 1627. 585413(36) 1627. 596130(49) 0. 22368(95) 0. 2436(15) / MHz -23. 0367(20) -23. 0329(28) / MHz 61. 8410(31) 57. 230(7) 103 / MHz 5. 654(82) 5. 26(15) / MHz DJ 103 / MHz H 2 S-63 Cu 37 Cl / MHz H 2 S-65 Cu 37 Cl 1584. 37261(12) 1584. 38067(52) 0. 2109(40) 0. 226(12) / MHz -18. 167(7) -18. 109(25) / MHz 61. 832(12) 57. 40(11) 103 / MHz 5. 16(24) 5. 45(31) DJ 103 / MHz

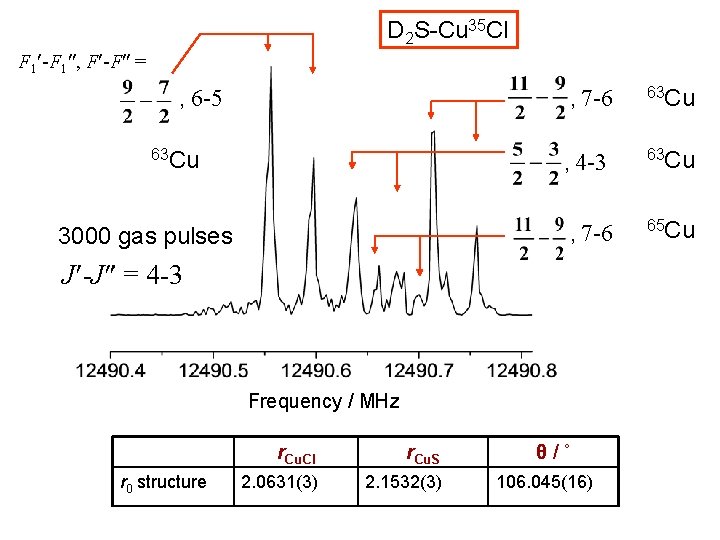
D 2 S-Cu 35 Cl F 1 -F 1 , F -F = , 6 -5 , 7 -6 63 Cu , 4 -3 , 7 -6 3000 gas pulses J -J = 4 -3 Frequency / MHz r 0 structure r. Cu. Cl r. Cu. S 2. 0631(3) 2. 1532(3) θ/˚ 106. 045(16) 63 Cu 65 Cu

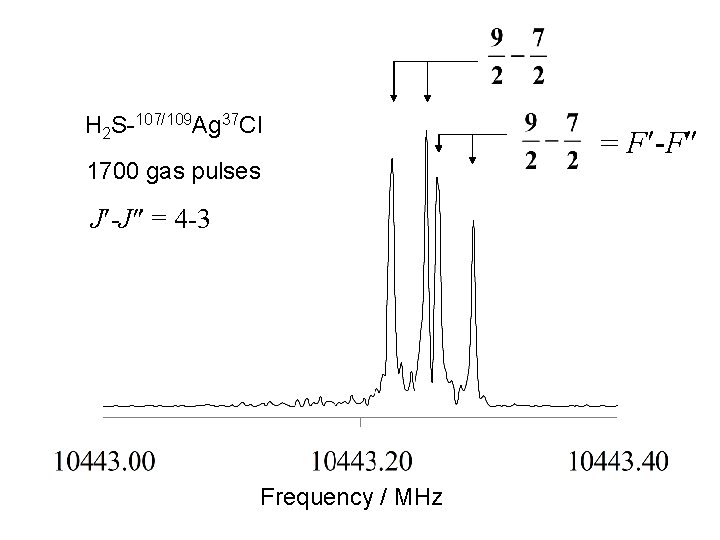
H 2 S-107/109 Ag 37 Cl 1700 gas pulses J -J = 4 -3 Frequency / MHz = F -F

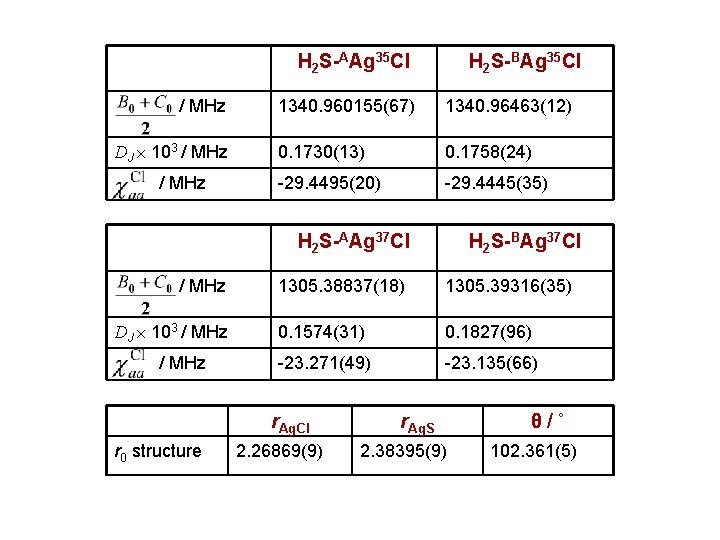
H 2 S-AAg 35 Cl / MHz DJ 103 / MHz H 2 S-BAg 35 Cl 1340. 960155(67) 1340. 96463(12) 0. 1730(13) 0. 1758(24) -29. 4495(20) -29. 4445(35) H 2 S-AAg 37 Cl / MHz DJ 103 / MHz 1305. 38837(18) 1305. 39316(35) 0. 1574(31) 0. 1827(96) -23. 271(49) -23. 135(66) r. Ag. Cl r 0 structure H 2 S-BAg 37 Cl 2. 26869(9) r. Ag. S 2. 38395(9) θ/˚ 102. 361(5)

H 2 S-MCla r. MCl / Å r. MS / Å MCl θ/˚ re / Å H 2 S-Cu. Clb 2. 063 2. 153 106. 0 2. 051 c H 2 S-Ag. Clb 2. 269 2. 384 102. 4 2. 281 H 2 S-HCld [1. 274]e 2. 535 86. 2 1. 274 2. 091 94 ( Cu-S-H) Cu. SH (rm(1))f a. All H 2 S-MCl structures are r 0. b. This work. c. Manson, E. L. ; De Lucia, F. C. ; Gordy, W. ; J. Chem. Phys. , 62 1040 -1043 (1975). d. Goodwin, E. J. ; Legon, A. C. ; J. Chem. Soc. Faraday Trans. 2: Molecular and Chemical Physics, 80(1) 51 -65 (1984). e. Fixed to re value for HCl. f. Janczyk, A. ; Walter, S. K. ; Ziurys, L. M. ; Chem. Phys. Lett. 401 211 -216 (2005)
![Comparison of Nuclear Quadrupole Coupling Constants 63 Cu MHz 35 Cl Comparison of Nuclear Quadrupole Coupling Constants [ (63 Cu)] [ / MHz (35 Cl)]](https://slidetodoc.com/presentation_image_h2/011e3db4d8b01610f16fbcab5f763b80/image-12.jpg)
Comparison of Nuclear Quadrupole Coupling Constants [ (63 Cu)] [ / MHz (35 Cl)] / MHz (a) Cu. Cl 16. 17 -32. 13 (a) Ar-Cu. Cl 33. 18 -28. 03 (b) Kr-Cu. Cl 36. 52 -27. 30 H 2 S-Cu. Cl 61. 84 -23. 04 OC-Cu. Cl 70. 83 -21. 47 (c) a. C. J. Evans and M. C. L. Gerry, J. Chem. Phys. 112, 1321 (2000) b. J. M. Michaud, S. A. Cooke and M. C. L. Gerry, Inorg. Chem. 43, 3871, (2004) c. N. R. Walker and M. C. L. Gerry, Inorg. Chem. 40, 6158 (2001)

Conclusions • H 2 S-MCl generated and studied in the gas phase for the first time where M=Cu or Ag. • MCl distance only slightly longer than in free MCl in both cases. • Angle between axes of H 2 S and MCl units measured. Angle is larger than in H 2 S-HCl in both cases. MS distances in both H 2 S-Cu. Cl and H 2 S-Ag. Cl are significantly shorter than in H 2 S-HCl. Acknowledgements The Royal Society Engineering and Physical Sciences Research Council (U. K. ) Vacancies (available to EU or UK residents) EPSRC-funded Ph. D studentship available. 1 year postdoctoral assistantship available.