1 Index About SYNCORP 3 Service Offering landscape

1

Index • • • About SYNCORP ----------- 3 Service Offering landscape ----------- 4 -9 SMO/CRO services ----------- 10 Project Management ----------- 11 -13 Trial Management Experience ----------- 14 Quality ----------- 15 Medical Writing Services ----------- 16 Consulting/Clinical Solutions ----------- 17 -19 Contact Us ----------- 20 2

About SYNCORP Clincare technologies (P) Ltd. (www. syncorp. in) was established in the year 2010 and we serve Pharma/Biotech companies, CROs, KPO/ BPOs with wide range of best clinical consulting and service solutions. Syncorp is fastest growing organization has it’s presence at Bangalore as a Head Quarter and have other clinical business units in Hyderabad and Bhubaneswar in India. SYNCORP educational trust (www. syncorp. org) is associated with the University of Mysore offers Master Degree in Clinical Research, other Diploma Course, PG Graduate Programs and Professional skill development programs Our Vision A Leading Consulting & Contract Research Organization driven by Knowledge, Integrity and Performance committed to our clients success Our Mission Our purpose is to enable our customer businesses to manage their services tailored to meet the specific and ever-changing business challenges. Build shareholder value by delivering pharmaceutical and healthcare products services and solutions in innovative and cost effective ways by consistently enabling businesses to produce superior operating results. 3

Syncorp Clincare Service Offerings Snapshot Syncorp Clincare Technologies (P) Ltd. , emerged as one of the fastest growing service provider in life sciences and healthcare sector. It provides services in Manpower Consulting, CRO/SMO Services, Education/ Training & Clinical Solutions. Our Goal is to serve our clients and emerge as one of the leaders in life sciences and healthcare sector Manpower Consulting CRO/SMO Services Education/ Training • Fresher • Lateral Hiring • Leadership Hiring • Clinical trial site management • Clinical Trial Monitoring • M. Sc. Programs (Affiliated to university of Mysore) • Diploma course for Pharma/Life Science/Medical science/ graduate/ Post Graduate • Staff Trainings • Corporate trainings- GCP/GLP/ISO Quality Training Consulting/Clinical solution • Scientific Writing • Medical Writing • Marketing Materials preparation • Diagnostic Lab set up and assisting in the accreditation procedure • Data Services 4

SYNCORP Core Team A leading CRO with comprehensive Phase I-IV Clinical Development Services We have industry experts in solving customer key problem/challenges and manage clinical trials project efficiencies. We are serving more than 10 clients Medical Director Core Team § Director: Mr. Subham Dutta, M. Sc. Biochemistry, Total Years of experience: 8+ Chief Advisor: Mr. Deepak, MBA. Total Years of experience: 22 § Consultants/SMEs § § 17 Academic experts 5 TA consultants 6 Medical writing experts 2 Biostatisticians § Dr. Pravina Koteshwar, MD Pharmacology. Total Years of experience: 12 Project Managers /CRCs § § Ms. Sathvayathi L. M. , M. Sc Clinical Research. Total Years of experience: 8+ Mr. Sudeep Patra. MPhrm Pharmacology. Total Years of experience: 7 5 CRCs 5 CRAs (part time) Supporting Team HR: Nethra Finance: Mr. Venkat IT Support: Mr. Pradeep Garg Training: Arvind 5

SYNCORP advantages Team’s expertise in clinical research, discovery and pre- clinical research, Diagnostics & other health care services Adaptability and scalability based on unique needs Large pool of Industry experts in our teaching academy Our team and consultants have decade plus years of experience in Clinical Research, consulting and services sector Strong Clinical Trial sites management capability to support global trials in India, including eastern part of India Strong team with International and Indian Regulatory experience in writing regulatory documents for USFDA, EMA and DCGI submissions Our learning and development academy affiliated to University of Mysore Expertise in academic scientific writing Manpower consulting/ Resource management system in place to assist our customer in their talent acquisition strategy – we have large diverse profiles database 6
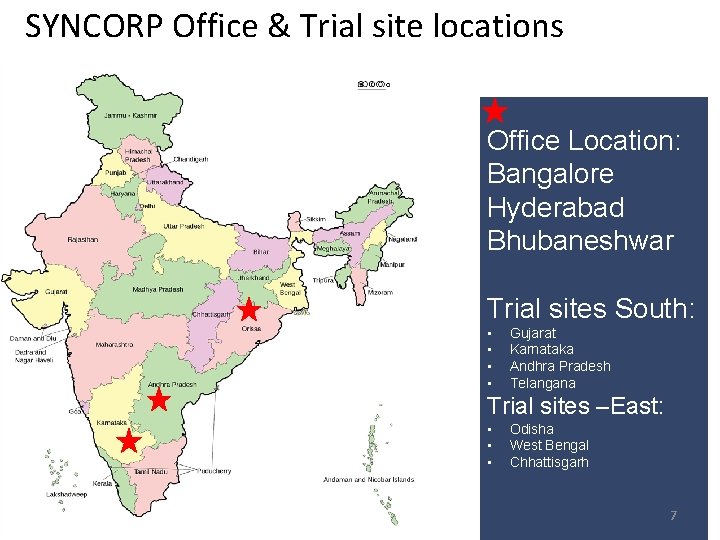
SYNCORP Office & Trial site locations Office Location: Bangalore Hyderabad Bhubaneshwar Trial sites South: • • Gujarat Karnataka Andhra Pradesh Telangana Trial sites –East: • • • Odisha West Bengal Chhattisgarh 7
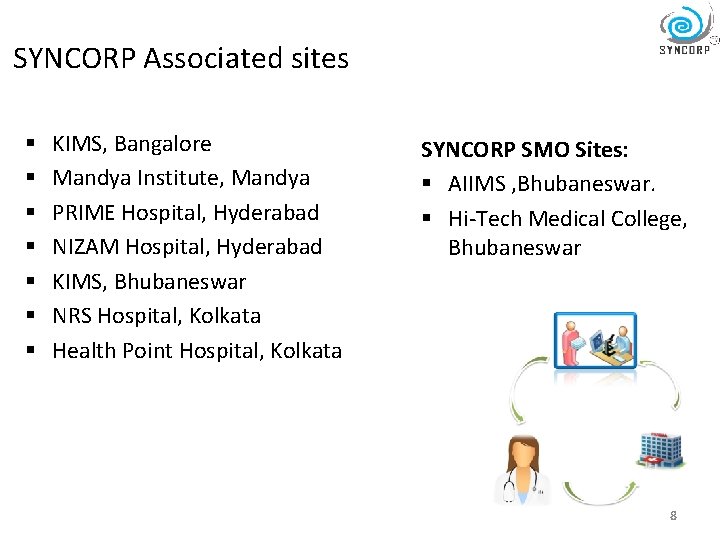
SYNCORP Associated sites § § § § KIMS, Bangalore Mandya Institute, Mandya PRIME Hospital, Hyderabad NIZAM Hospital, Hyderabad KIMS, Bhubaneswar NRS Hospital, Kolkata Health Point Hospital, Kolkata SYNCORP SMO Sites: § AIIMS , Bhubaneswar. § Hi-Tech Medical College, Bhubaneswar 8

PI network Access to Key Decision Makers & Investigators in most of the Therapeutic area’s & they represent top hospitals and research institutes and they are GCP trained, experienced in Clinical trial services Bone / Osteoporosis Immunology Orthopaedics Cardiovascular Infectious diseases Pulmonology CNS Inflammation Psychiatry Dermatology Internal diseases Respiratory Diabetes Metabolic diseases Rheumatology Endocrinology Nephrology Surgery ENT Neurology Urology Gastroenterology Oncology Vaccines Haematology Ophthalmology Thrombosis 9

CRO/SMO Services Key Services § Study Feasibility Support site feasibility for CROs/Sponsors. § Site and Investigator Selection Provide accurate, responsible, experience sites. § Clinical Trial Monitoring Provide therapeutic expertise GCP trained monitor. § Site Management PI experienced in local and global trial and dedicated site CRC. § Project Management Experienced, skilled and trained project managers and support of latest IT technology. § Clinical Trial Supply Management Excellent logistic network and support services. § Ethics Committee Services Support for SOP writing, GCP training and registration of IRB. § Retail chain Pharmacy & Clinic Management Retail pharmacy counter and pediatric clinic for Tier II 10 and Tier III cities.
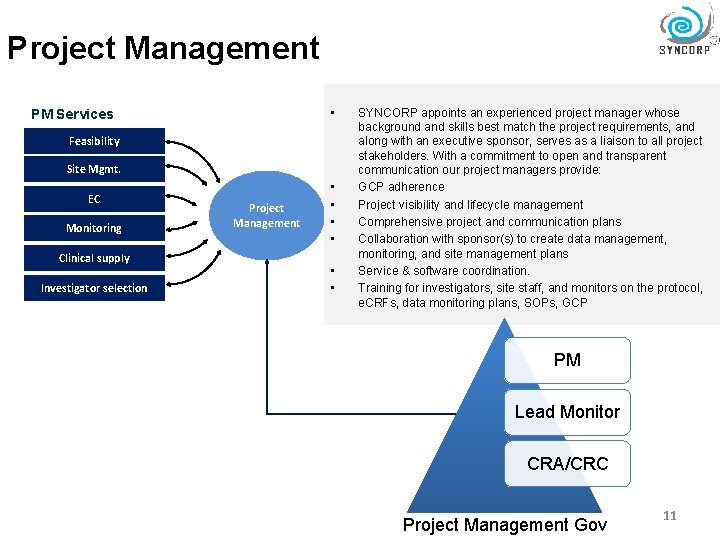
Project Management • PM Services Feasibility Site Mgmt. EC Monitoring Clinical supply Investigator selection Project Management • • • SYNCORP appoints an experienced project manager whose background and skills best match the project requirements, and along with an executive sponsor, serves as a liaison to all project stakeholders. With a commitment to open and transparent communication our project managers provide: GCP adherence Project visibility and lifecycle management Comprehensive project and communication plans Collaboration with sponsor(s) to create data management, monitoring, and site management plans Service & software coordination. Training for investigators, site staff, and monitors on the protocol, e. CRFs, data monitoring plans, SOPs, GCP PM Lead Monitor CRA/CRC Project Management Gov 11
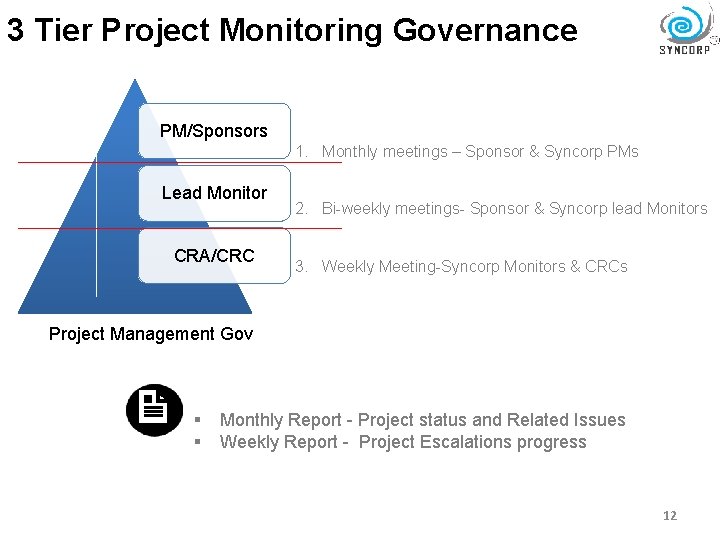
3 Tier Project Monitoring Governance PM/Sponsors 1. Monthly meetings – Sponsor & Syncorp PMs Lead Monitor CRA/CRC 2. Bi-weekly meetings- Sponsor & Syncorp lead Monitors 3. Weekly Meeting-Syncorp Monitors & CRCs Project Management Gov § § Monthly Report - Project status and Related Issues Weekly Report - Project Escalations progress 12
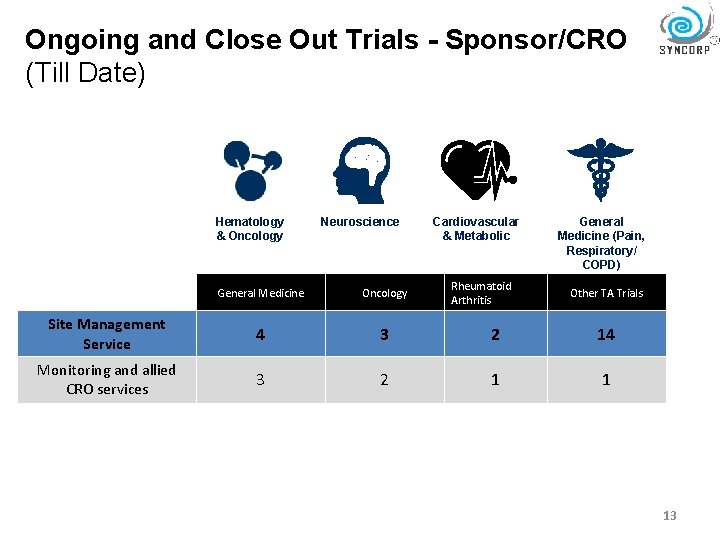
Ongoing and Close Out Trials - Sponsor/CRO (Till Date) Hematology & Oncology Neuroscience Cardiovascular & Metabolic Rheumatoid Arthritis General Medicine (Pain, Respiratory/ COPD) General Medicine Oncology Other TA Trials Site Management Service 4 3 2 14 Monitoring and allied CRO services 3 2 1 1 13
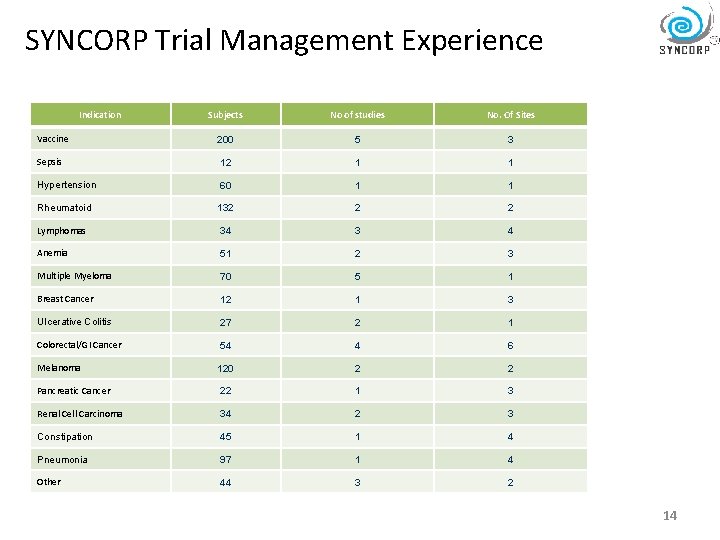
SYNCORP Trial Management Experience Indication Subjects No of studies No. Of Sites Vaccine 200 5 3 Sepsis 12 1 1 Hypertension 60 1 1 Rheumatoid 132 2 2 Lymphomas 34 3 4 Anemia 51 2 3 Multiple Myeloma 70 5 1 Breast Cancer 12 1 3 Ulcerative Colitis 27 2 1 Colorectal/GI Cancer 54 4 6 Melanoma 120 2 2 Pancreatic Cancer 22 1 3 Renal Cell Carcinoma 34 2 3 Constipation 45 1 4 Pneumonia 97 1 4 Other 44 3 2 14
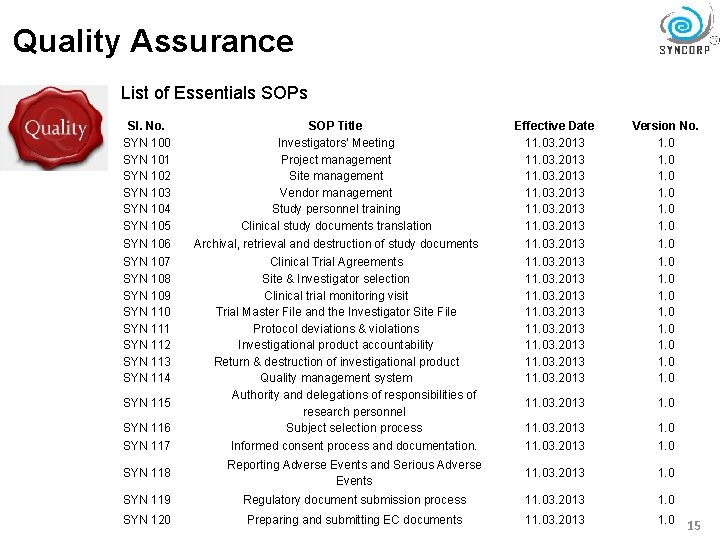
Quality Assurance List of Essentials SOPs Sl. No. SYN 100 SYN 101 SYN 102 SYN 103 SYN 104 SYN 105 SOP Title Investigators' Meeting Project management Site management Vendor management Study personnel training Clinical study documents translation Effective Date 11. 03. 2013 Version No. 1. 0 1. 0 SYN 106 Archival, retrieval and destruction of study documents 11. 03. 2013 1. 0 SYN 107 SYN 108 SYN 109 SYN 110 SYN 111 SYN 112 SYN 113 SYN 114 11. 03. 2013 11. 03. 2013 1. 0 11. 03. 2013 1. 0 SYN 116 Clinical Trial Agreements Site & Investigator selection Clinical trial monitoring visit Trial Master File and the Investigator Site File Protocol deviations & violations Investigational product accountability Return & destruction of investigational product Quality management system Authority and delegations of responsibilities of research personnel Subject selection process 11. 03. 2013 1. 0 SYN 117 Informed consent process and documentation. 11. 03. 2013 1. 0 SYN 118 Reporting Adverse Events and Serious Adverse Events 11. 03. 2013 1. 0 SYN 119 Regulatory document submission process 11. 03. 2013 1. 0 SYN 120 Preparing and submitting EC documents 11. 03. 2013 1. 0 SYN 115 15

Medical Writing Services (Consulting and Clinical Services) Under Medical writing services we offer (1) Academic scientific Writing (2) Medico Marketing Writing & (3) Regulatory writings for all our esteemed clients. Academic Scientific Writing (For Doctors/Scientist/Research Scholars) § § § Manuscript Writing Article Writing Case Studies writing Thesis Writing Support Statistical Analysis Support Regulatory Writing (CROs/Pharma organization) Medico Marketing Writing (For Pharma/Biotech Organizations) Content and developing design: § Visual Aid for Pharmaceuticals/Medical Devices § Pharmaceutical Product Monograph § Continuous Medical Education Slides for medical professionals § Training manuals- for sales force trainings § Clinical Trial - Regulatory Documents preparation We ensure documents comply with regulatory, journal, or other guidelines in terms of content, format, and structure 16

Consulting Services (Business Solutions) SYNCORP business solutions that partners with its clients to simplify, strengthen and transform their business needs and ensure the highest level of Quality and satisfaction through our commitment and comprehensive industry expertise. Pharma/CRO’s clinical consulting services Diagnostic & Health care consulting services § § § § § Data Services Statistical Services Quality Services/Audit Preparation ISO certification preparation. Business Process Re-engineering NABL Accreditation services ISO certification preparation Laboratory design and setup services Validation services Business Process Re-engineering 17
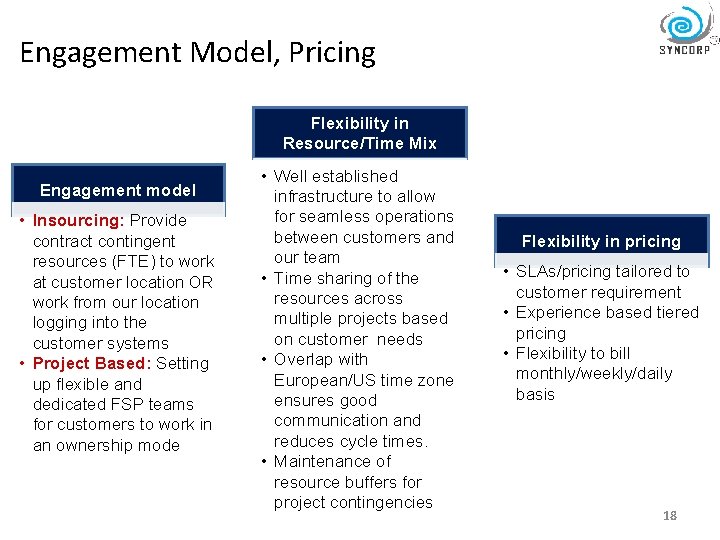
Engagement Model, Pricing Flexibility in Resource/Time Mix Engagement model • Insourcing: Provide contract contingent resources (FTE) to work at customer location OR work from our location logging into the customer systems • Project Based: Setting up flexible and dedicated FSP teams for customers to work in an ownership mode • Well established infrastructure to allow for seamless operations between customers and our team • Time sharing of the resources across multiple projects based on customer needs • Overlap with European/US time zone ensures good communication and reduces cycle times. • Maintenance of resource buffers for project contingencies Flexibility in pricing • SLAs/pricing tailored to customer requirement • Experience based tiered pricing • Flexibility to bill monthly/weekly/daily basis 18

Client Testimonials (Some examples) Syncorp has developed and supported the business in our emerging markets mainly supports the Global Clinical Trial Solutions, alliances and promote global connections to customers: they significantly increased our footprint in the region. Syncorp is a thorough professional organization, paying great attention to detail while always seeing the bigger picture. A real pleasure to have worked with Syncorp and I would have no hesitation in recommending Syncorp. Assistant Vice President, Leading Global Healthcare BPO Company. SYNCORP clinical trial site management services has demonstrated how a group of willing and dedicated investigators, supported by a team of committed coordinators, can develop towards total compliance with ICH GCP. Head Clinical Operations, Leading Indian Pharmaceutical. Start from the professionalism demonstrated in delivering results, the sheer responsiveness to our concerns and the attitude towards providing proactive solutions, made the experience of working with this SYNCORP team an absolute delight. Snr Manager, Medical Writing. Leading Global Pharma Company. Syncorp has paved way for many aspiring students. The entire team of Syncorp is very dedicated, focus oriented, supportive and caring. The trainers are highly professional and at the same time very friendly to the students. The environment provided by Syncorp is very nice. I am very glad to be a part of the Syncorp family. I want to thank the entire team of Syncorp for showing me the path to my future. Student from our academy. Joined a Leading Global CRO Company 19

Corporate Office: Bangalore: Novel Business Centre, No. 10, 100 Feet Ring Road, BTM Layout, 1 st Stage, Bangalore- 560068 (INDIA) Contact number: +91 -8050072226 / +91 -08065474772 Branch Office: Hebbal (Bangalore), Hyderabad and Bhubaneswar. Website: www. syncorp. in and www. syncorp. org E-mail: subham@syncorp. in/ info@syncorp. in 20
- Slides: 20