1 HNMR spectra interpretation Examples Tables and Problems

1 H-NMR spectra interpretation Examples , Tables and Problems
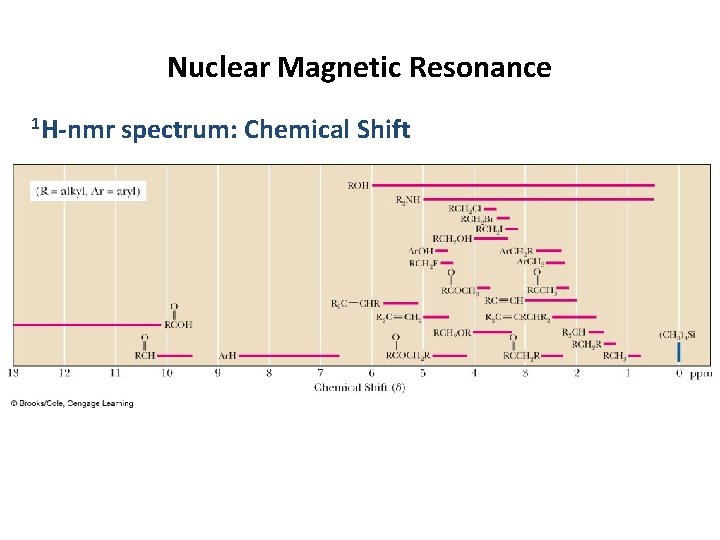
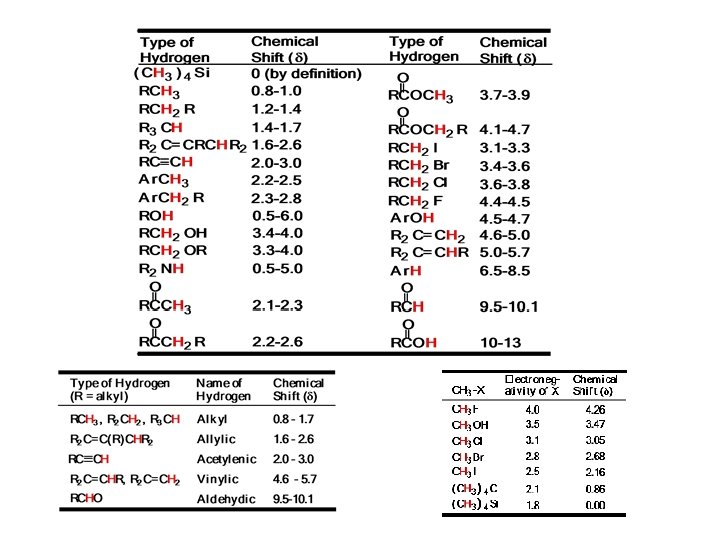
Nuclear Magnetic Resonance 1 H-nmr spectrum: Chemical Shift
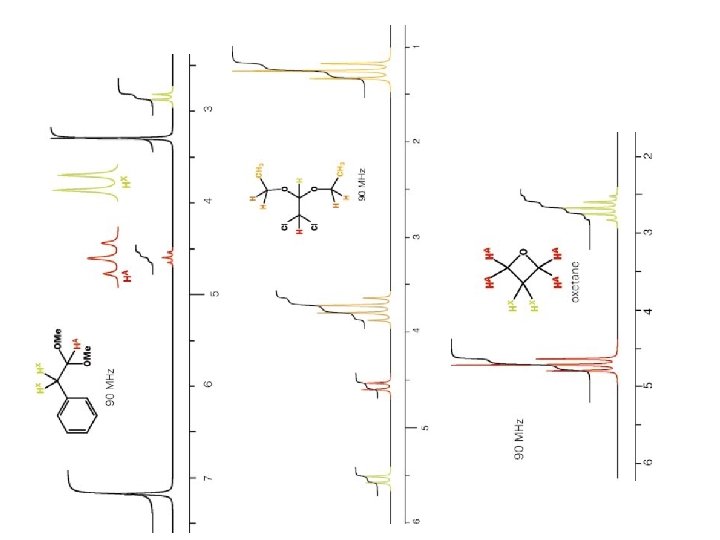
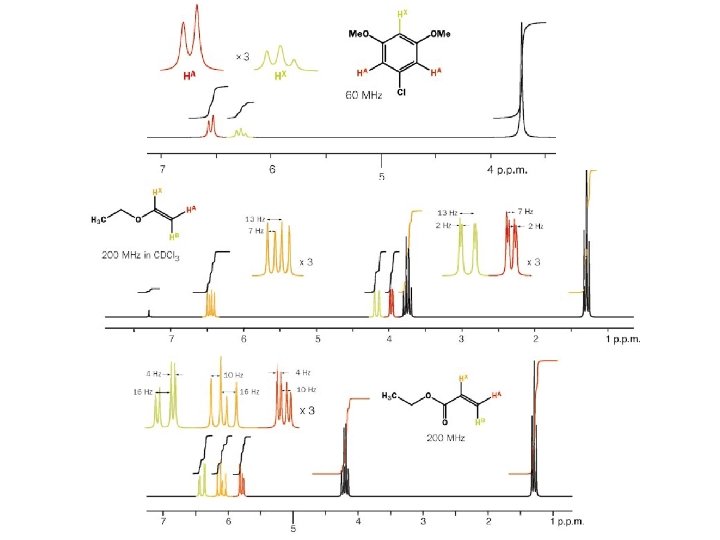

How 1 H NMR spectrum for p-xylene will appear? p-xylene How many H type are their? This will inform us the number of signals in the spectra How many H atoms in each type? This will inform us the area of each signal What are the functional groups near each H type? This will inform us where the signal will appear (chemical shift d) How many H atoms are attached to the carbon atom s nearby each H type? This will inform us how the splitting pattern will appear for each signal
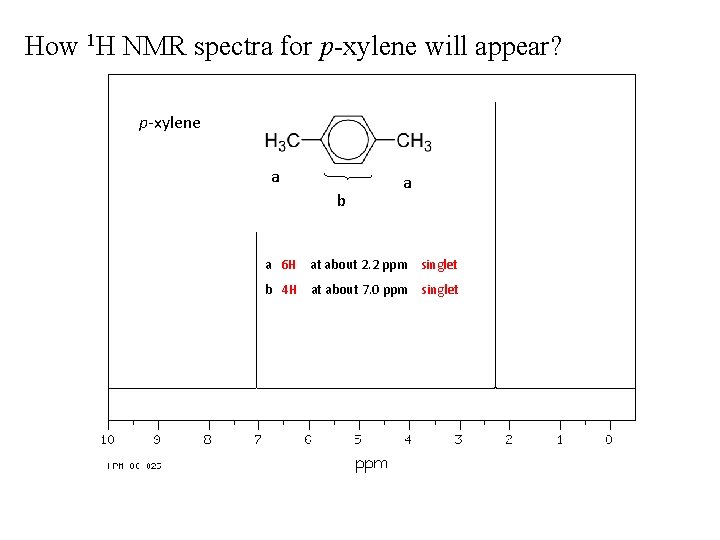
How 1 H NMR spectra for p-xylene will appear? p-xylene a b a a 6 H at about 2. 2 ppm singlet b 4 H at about 7. 0 ppm singlet
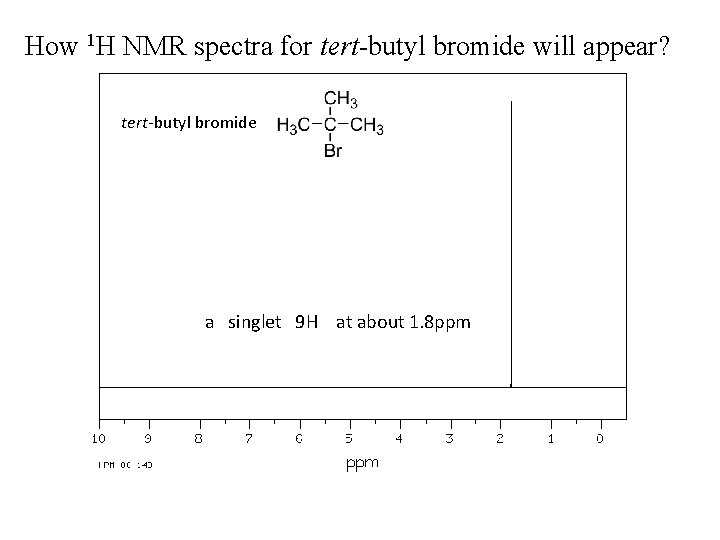
How 1 H NMR spectra for tert-butyl bromide will appear? tert-butyl bromide a singlet 9 H at about 1. 8 ppm
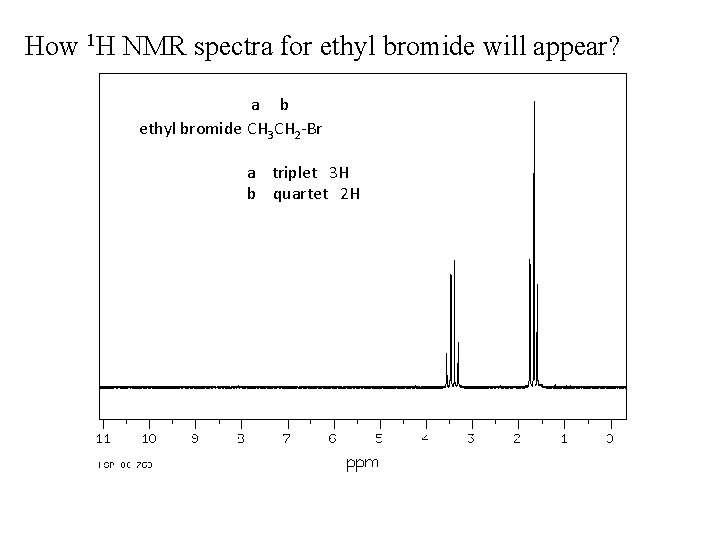
How 1 H NMR spectra for ethyl bromide will appear? a b ethyl bromide CH 3 CH 2 -Br a triplet 3 H b quartet 2 H

Draw the chemical structure for the compound Number of signals in the spectra �will inform us the number of H type in the molecule Area of each signal �will inform us the number of H atoms in each type Chemical shift d of the signal �will inform us about closeness of H type to the functional group Splitting pattern for each signal �will inform us how many H atoms surrounding each type
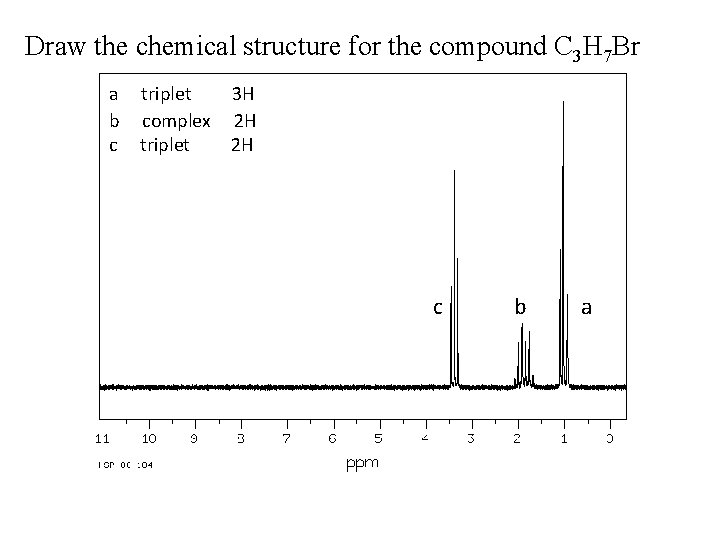
Draw the chemical structure for the compound C 3 H 7 Br a b c triplet complex triplet 3 H 2 H 2 H c b a

Draw the chemical structure for the compound C 3 H 7 Br o C 3 H 7 Br has IHD = 0 �no unsaturation (no double bonds nor cycles), so it is chain molecule (straight or branched). o From the given data, there are three signals (a, b and c) so there are three types of H atoms, each type will be at a unique carbon atom. o It is obvious that there is no symmetry in the molecule, the formula has 7 H atoms and they are distributed on three separate signals (3+2+2). We have CH 3 -, -CH 2 a b and -CH 2 c o c signal has the higher d value (more deshielded atoms), this mean that this H type of atoms are the nearest to the Br atom. We could use tables to check for this, so c signal should be like this -CH 2 -Br c o The splitting pattern shows that a signal is triplet, this mean that it has only 2 H atoms surrounding it, so this side of the molecule will be CH 3 -CH 2 - because it a has only one side to attach with. o Now, the final touch, the like between the tow ends, it is the b hydrogens. This will form CH 3 -CH 2 -Br. This justify the splitting patterns for b and c. a b c
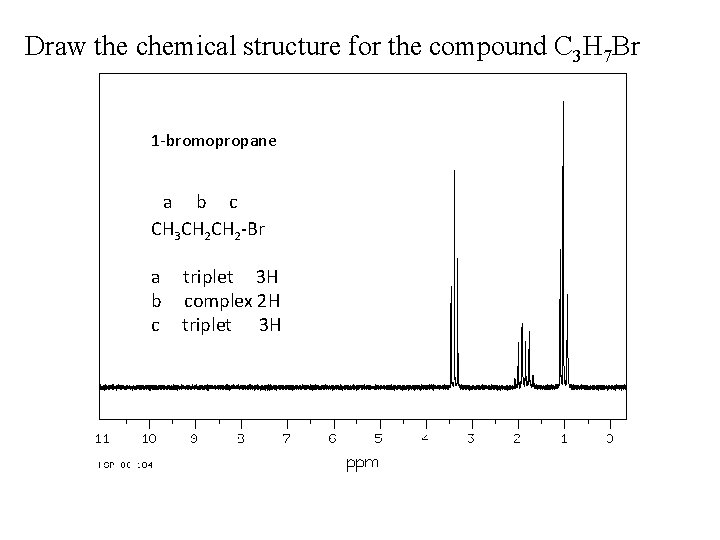
Draw the chemical structure for the compound C 3 H 7 Br 1 -bromopropane a b c CH 3 CH 2 -Br a b c triplet 3 H complex 2 H triplet 3 H
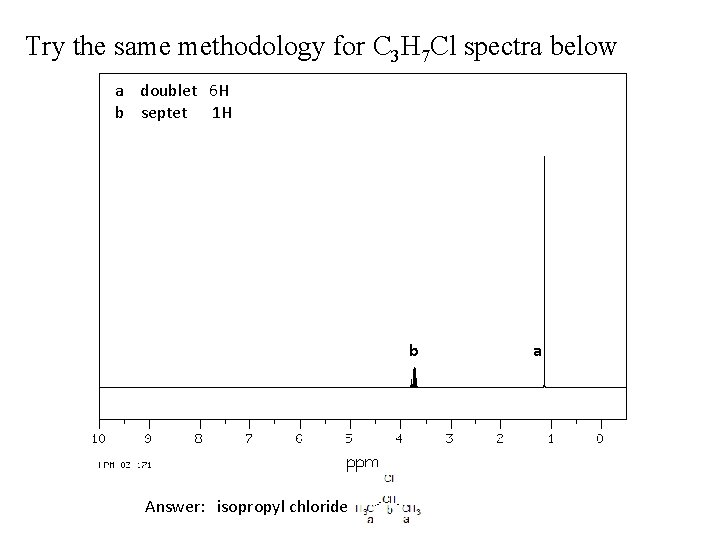
Try the same methodology for C 3 H 7 Cl spectra below a doublet 6 H b septet 1 H b Answer: isopropyl chloride a
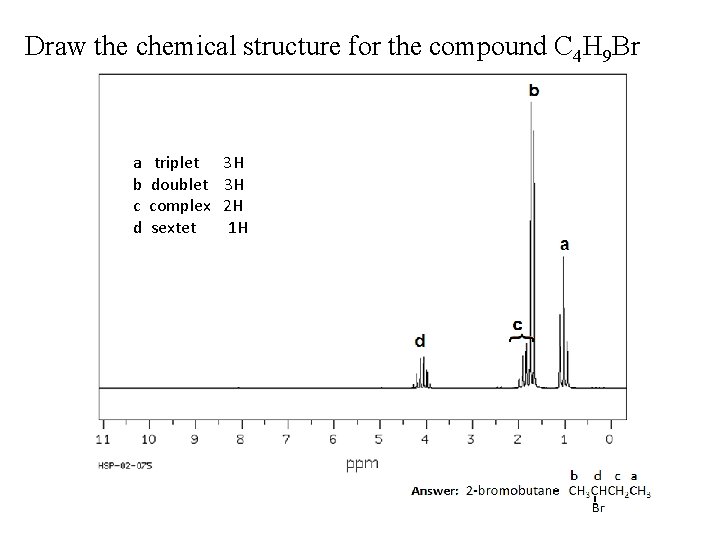
Draw the chemical structure for the compound C 4 H 9 Br a b c d triplet doublet complex sextet 3 H 3 H 2 H 1 H
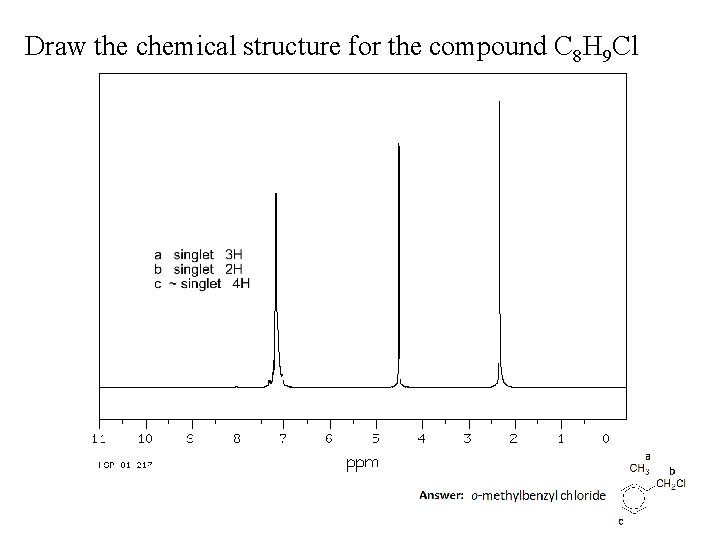
Draw the chemical structure for the compound C 8 H 9 Cl
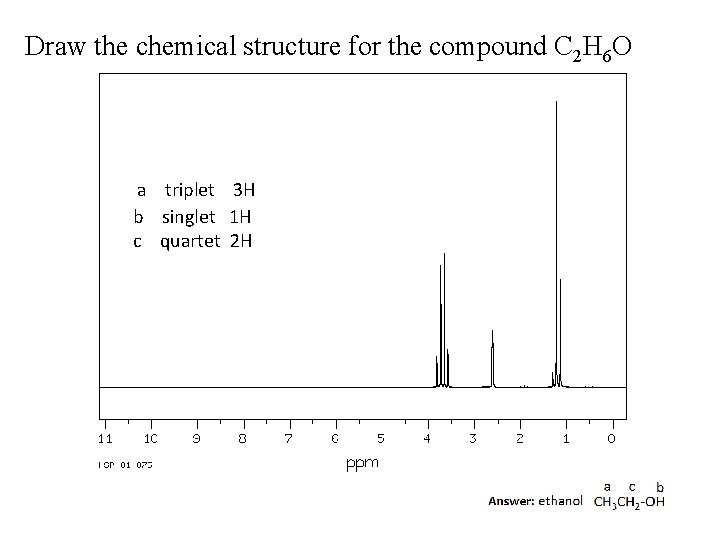
Draw the chemical structure for the compound C 2 H 6 O a triplet 3 H b singlet 1 H c quartet 2 H
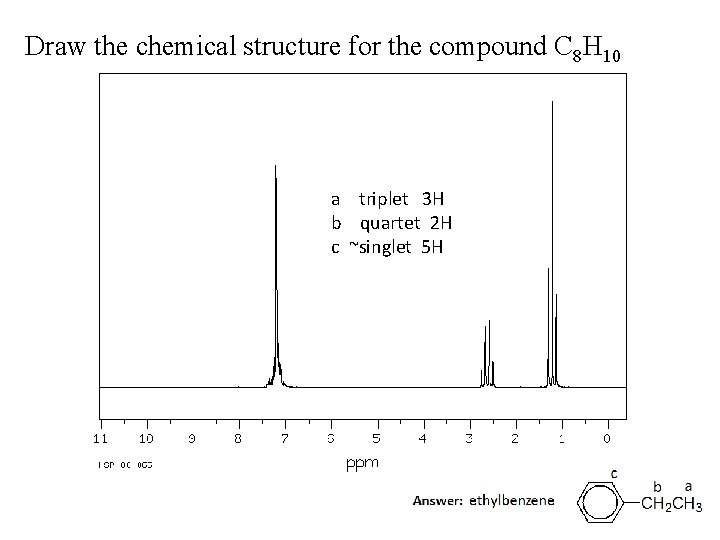
Draw the chemical structure for the compound C 8 H 10 a triplet 3 H b quartet 2 H c ~singlet 5 H
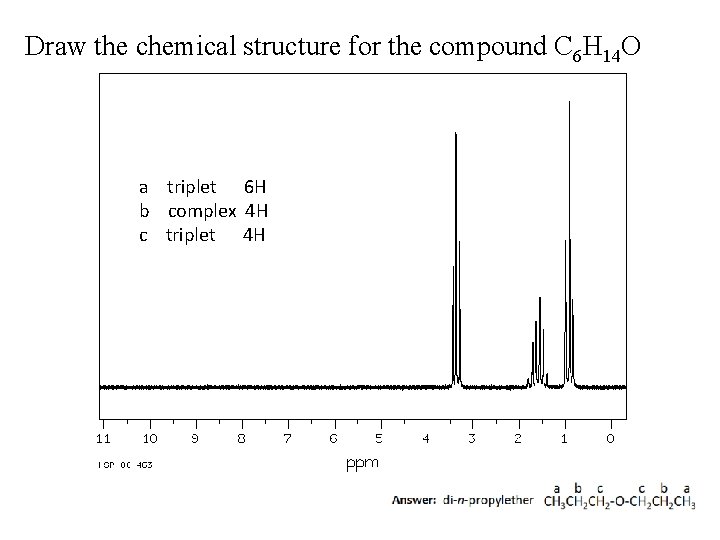
Draw the chemical structure for the compound C 6 H 14 O a triplet 6 H b complex 4 H c triplet 4 H
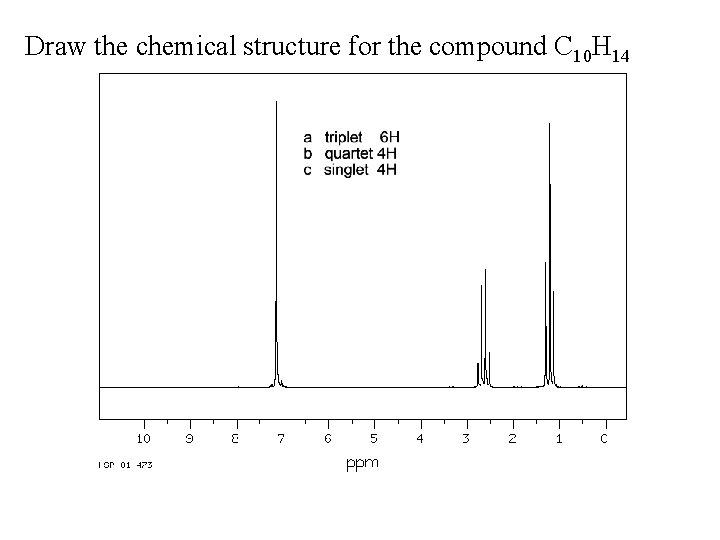
Draw the chemical structure for the compound C 10 H 14
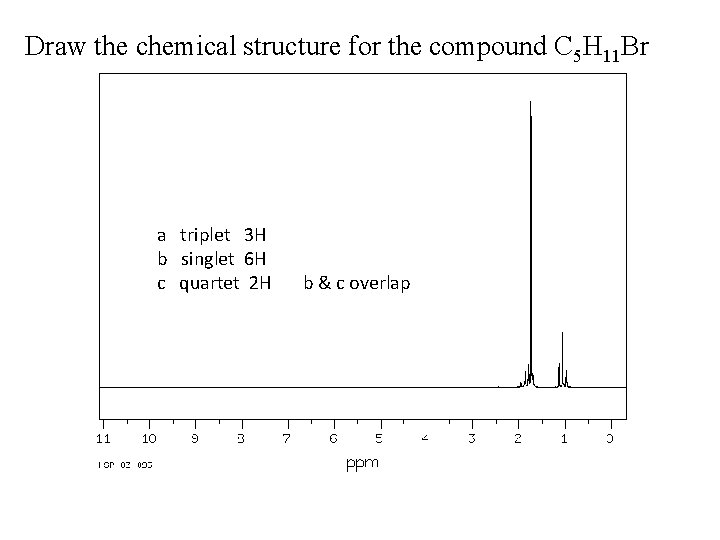
Draw the chemical structure for the compound C 5 H 11 Br a triplet 3 H b singlet 6 H c quartet 2 H b & c overlap
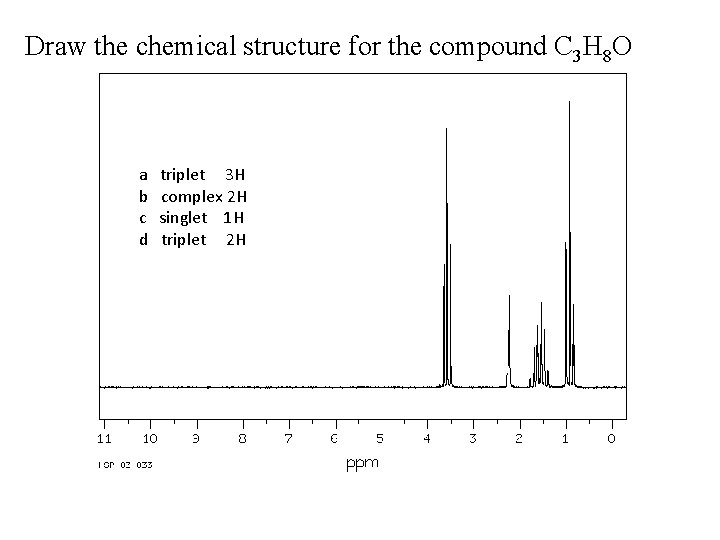
Draw the chemical structure for the compound C 3 H 8 O a b c d triplet 3 H complex 2 H singlet 1 H triplet 2 H
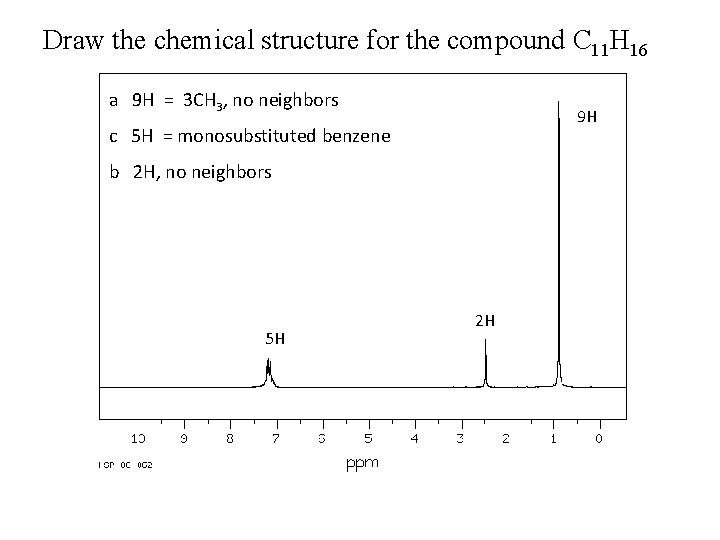
Draw the chemical structure for the compound C 11 H 16 a 9 H = 3 CH 3, no neighbors 9 H c 5 H = monosubstituted benzene b 2 H, no neighbors 5 H 2 H
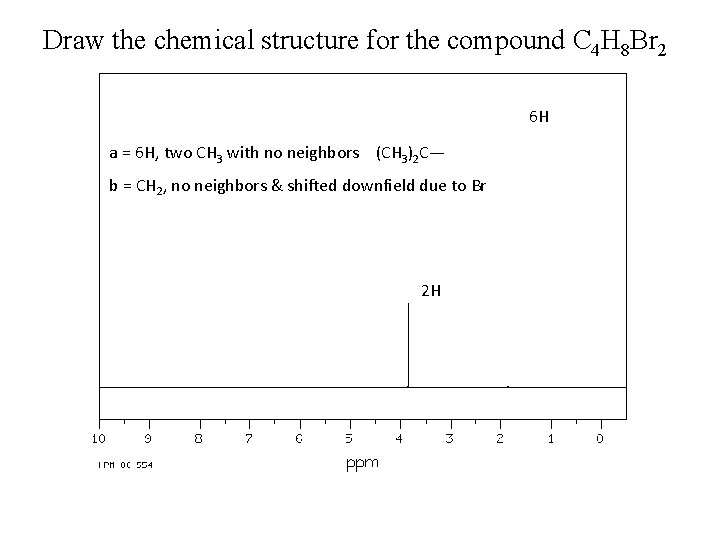
Draw the chemical structure for the compound C 4 H 8 Br 2 6 H a = 6 H, two CH 3 with no neighbors (CH 3)2 C— b = CH 2, no neighbors & shifted downfield due to Br 2 H
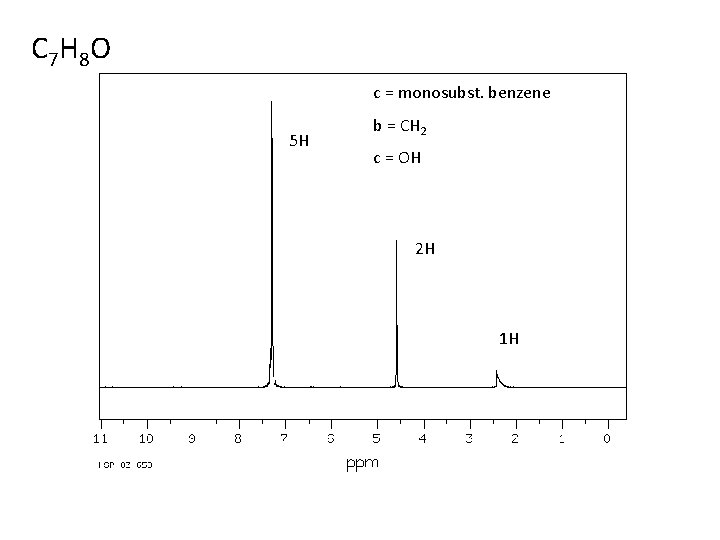
C 7 H 8 O c = monosubst. benzene 5 H b = CH 2 c = OH 2 H 1 H




- Slides: 29