1 Food chemistry Food Rheology Food Science Biochemistry


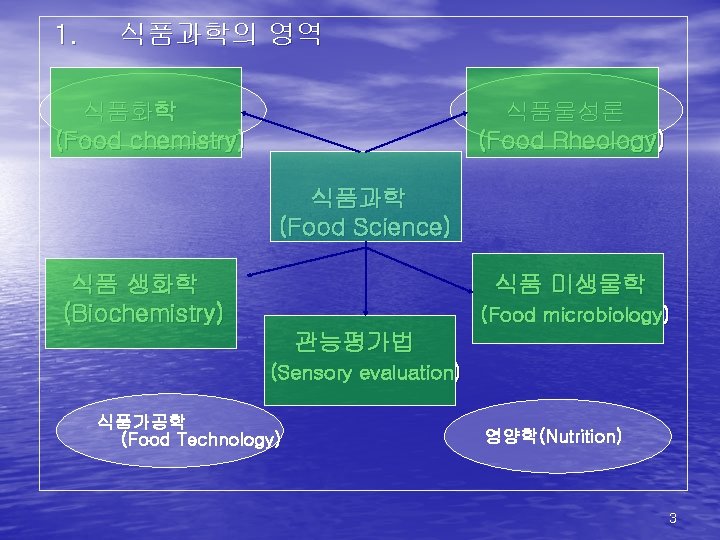
1. 식품과학의 영역 식품화학 (Food chemistry) 식품물성론 (Food Rheology) 식품과학 (Food Science) 식품 생화학 (Biochemistry) 식품 미생물학 (Food microbiology) 관능평가법 (Sensory evaluation) 식품가공학 (Food Technology) 영양학(Nutrition) 3


2. 식품의 조건 Nutritive value (영양) Safety ) (안전성 Organoleptic quality (기호성) Economy (경제성) 5
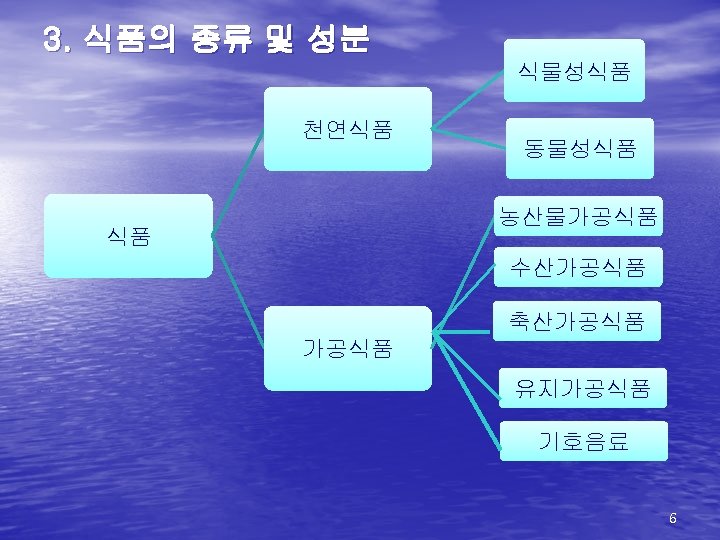

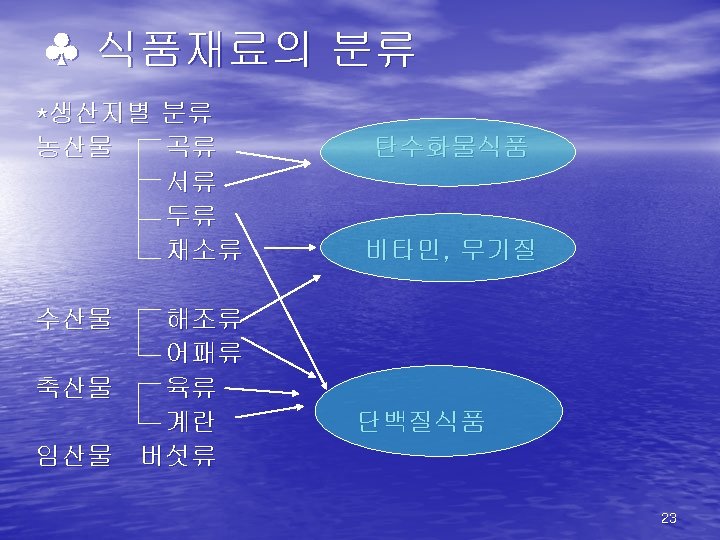
식품의 구성성분 1. 일반성분 Carbohydrate Moisture Protein Organic matters Food Fiber Sugar Lipids Vitamins Solid matters Minerals 2. 특수성분 taste, flavor, color, enzyme 7

8




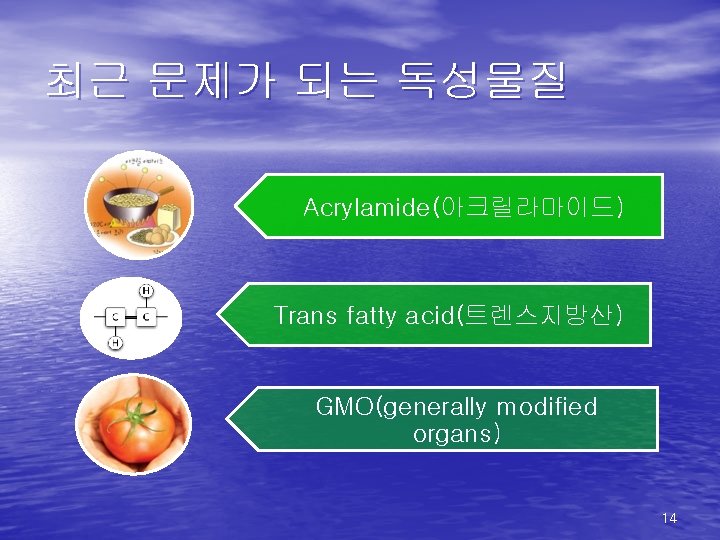
최근 문제가 되는 독성물질 Acrylamide(아크릴라마이드) Trans fatty acid(트렌스지방산) GMO(generally modified organs) 14








3장. Carbohydrate (1) Structure Organic molecules in which carbon, hydrogen and oxygen bond together in the ratio Cn(H 2 O)m 6 CO 2 + 6 H 2 O + energy (from sunlight) C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + energy 24

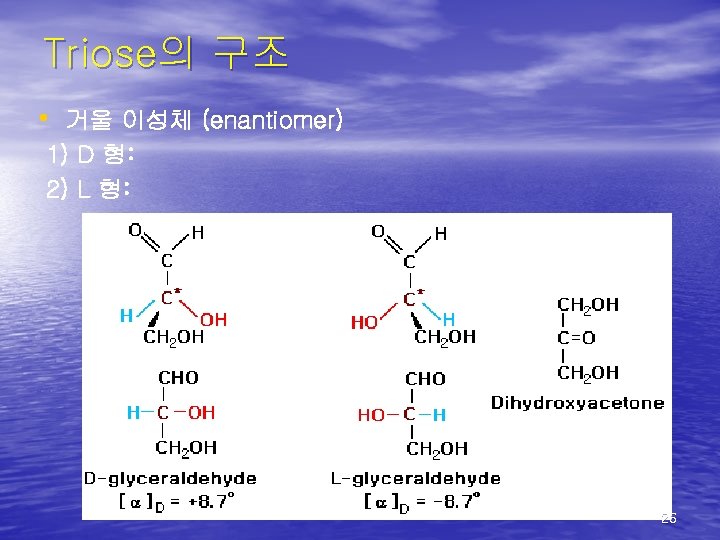
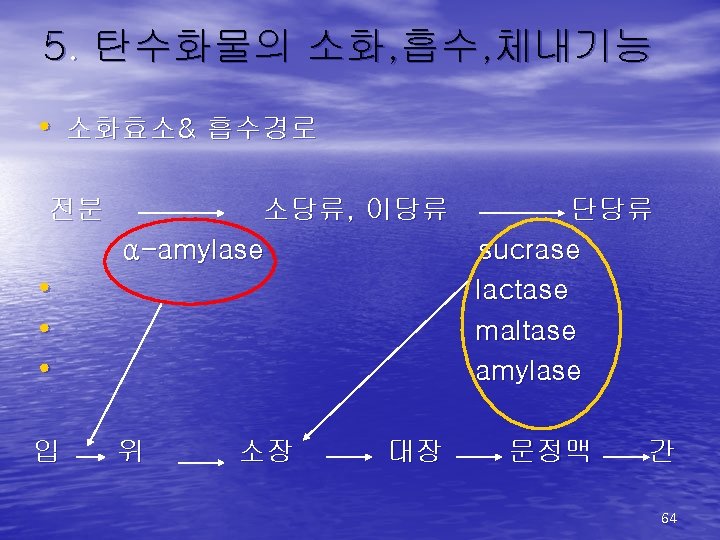
(2)Nomenclature • Carbohydrates are the main energy source for the human body. • Chemically, carbohydrates are organic • • molecules in which carbon, hydrogen and oxygen bond together in the ratio: Cn(H 2 O)m Aldose… bound with CHO(aldehyde) Ketose… bound with CO(ketone) 25
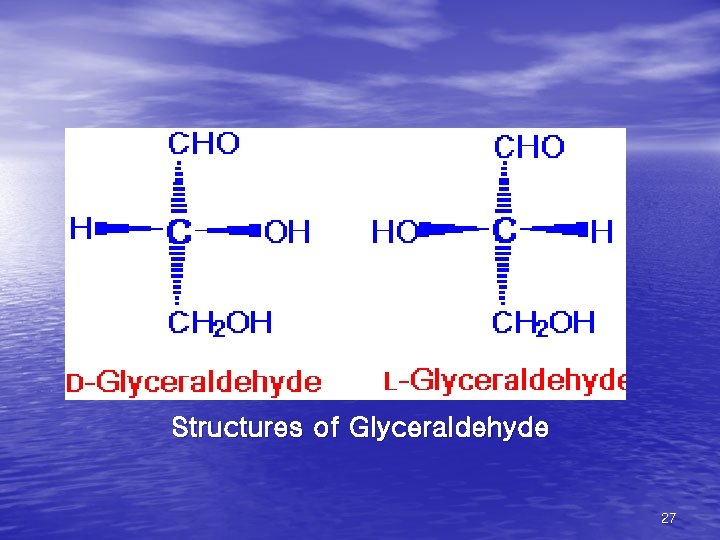
Structures of Glyceraldehyde 27

(3) Classification 28

1)Monosaccharides(단당류) 29

(1) Pentose(5탄당) 1. Arabinose: - 천연에서 L-arabinofuranose형으로 존재 - 침엽수의 arabinogalactan, 식물의 hemicellulose, 아리비아 고무 등의 점질물, pectin의 구성성분 2. D-Ribose & D-deoxyribose: - 동식물 세포의 핵산의 구성성분 - Riboflavin의 보효소 성분으로 존재 (D-Ribose) 3. D-xylose: - Hemicellulose의 주성분인 xylan을 구성 30
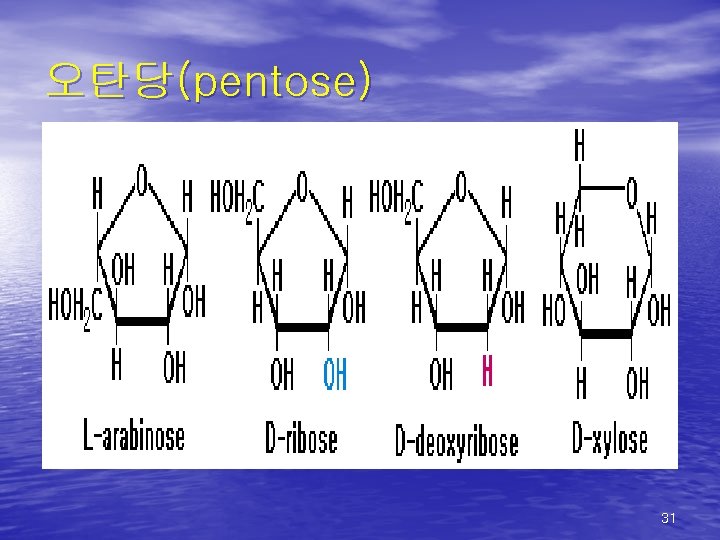
오탄당(pentose) 31

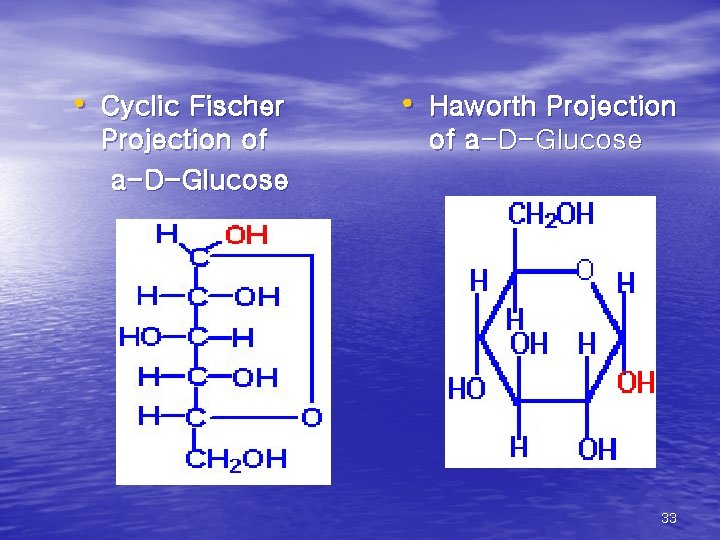
• Cyclic Fischer Projection of a-D-Glucose • Haworth Projection of a-D-Glucose 33
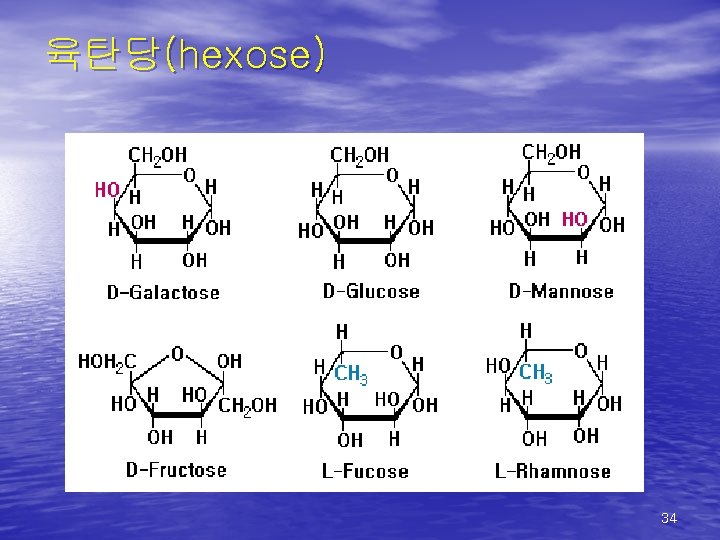
육탄당(hexose) 34

More Monosaccharide Derivatives • Sugar alcohols: mild reduction of sugars • Deoxy sugars: constituents of DNA, etc. • Sugar esters: phosphate esters like ATP • • are important Amino sugars contain an amino group in place of a hydroxyl group Acetals, ketals and glycosides: basis for oligo- and poly-saccharides 35
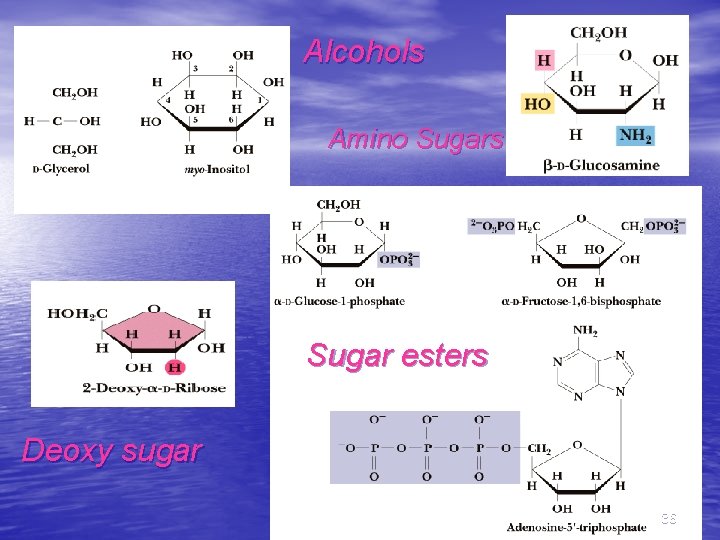
Alcohols Amino Sugars Sugar esters Deoxy sugar 36

당알코올 (Sugar alcohol) • 단당류의 aldehyde기나 ketone기의 환원 - 오탄당 알코올: pentitol - 육탄당 알코올: hexitol • Ribitol & xylitol: 감미료로 이용 - Xylitol: uronic acid 대사경로의 중간산물 • Sorbitol: glucose, fructose의 환원에 의해 생성 • Mannitol: mannose의 환원에 의해 생성 37

2) Disaccharides(이당류) • The linkage of two monosaccharides to form disaccharides involves a glycosidic bond. • Several physiologically important disaccharides are sucrose, lactose and maltose. 38
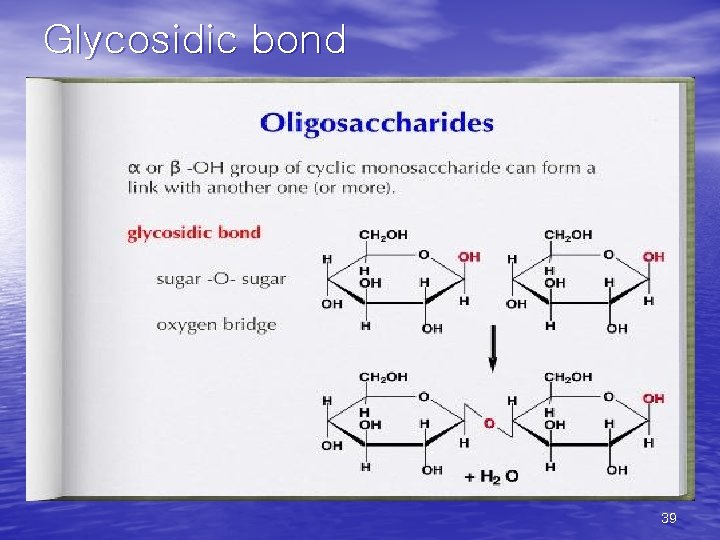
Glycosidic bond 39
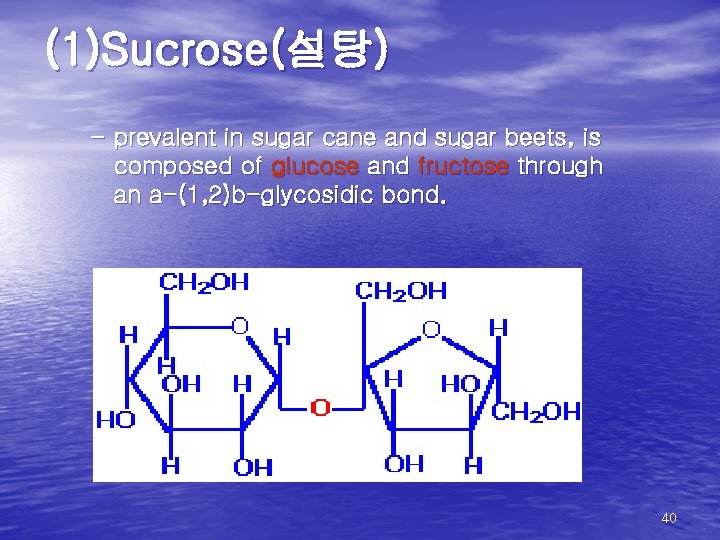
(1)Sucrose(설탕) – prevalent in sugar cane and sugar beets, is composed of glucose and fructose through an a-(1, 2)b-glycosidic bond. 40
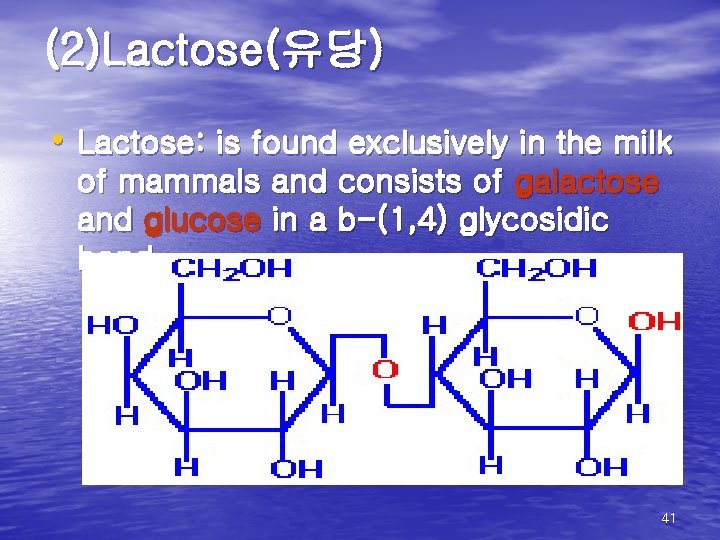
(2)Lactose(유당) • Lactose: is found exclusively in the milk of mammals and consists of galactose and glucose in a b-(1, 4) glycosidic bond 41
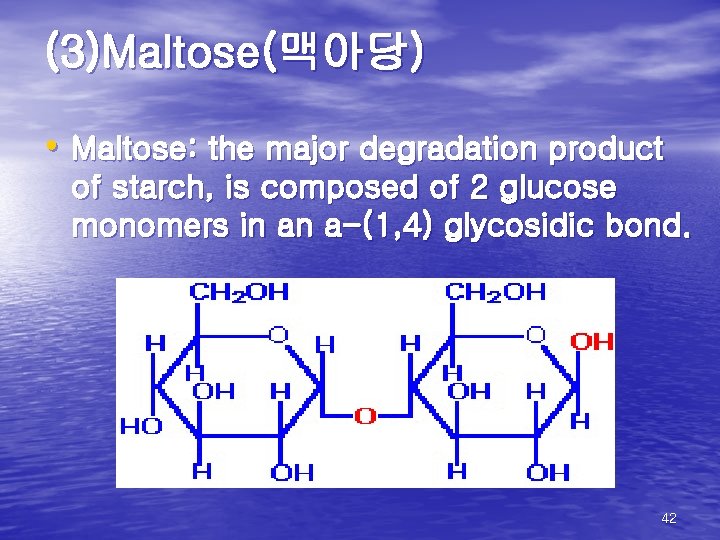
(3)Maltose(맥아당) • Maltose: the major degradation product of starch, is composed of 2 glucose monomers in an a-(1, 4) glycosidic bond. 42


44

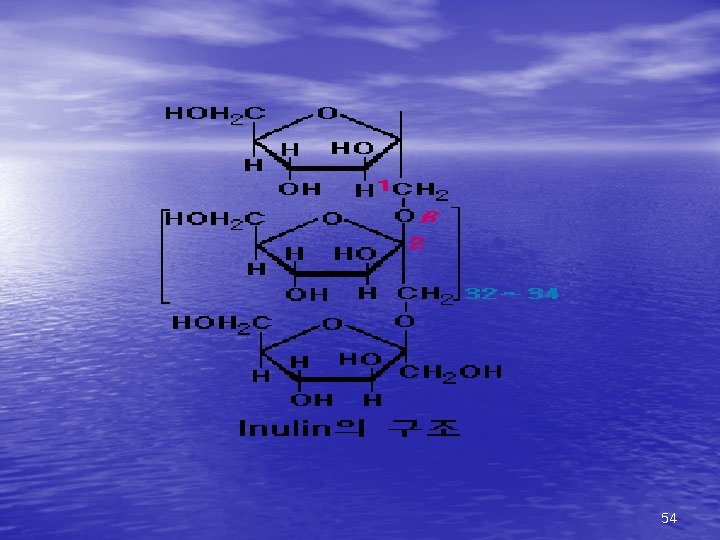
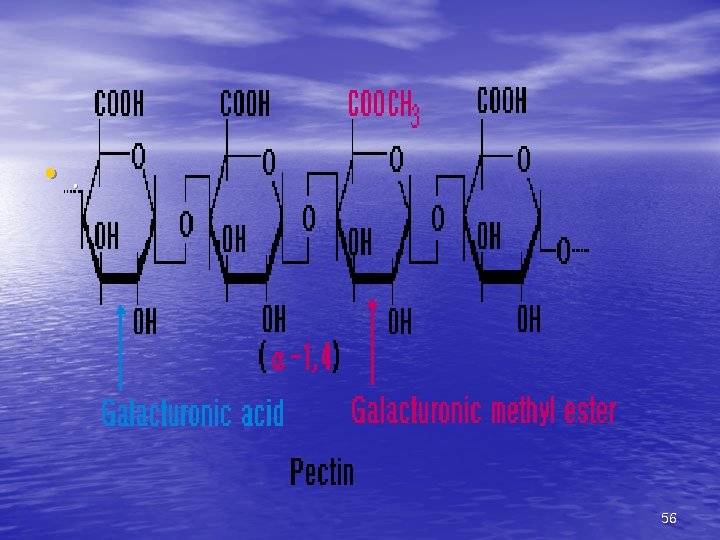
4) Polysaccharides(다당류) Most of the carbohydrates found in nature occur in the form of high molecular weight polymers(200~300)called polysaccharides. Homopolysaccharides are composed of a single monosaccharide building block ex) Starch, Dextrin, Glycogen, Fiber, Pentosan, Inulin Heteropolysaccharides. are composed of more than one type of monosaccharide ex) Glucomannan, Pectin 45

• (1)Starch • the major form of stored carbohydrate in plant • • cells. lower degree of branching (about every 20 -30 residues). Amylose + amylopectin. • (2) Glycogen • the major form of stored carbohydrate in • • animals. homopolymer of glucose in a-(1, 4) linkage; it is also highly branched, with a-(1, 6) branch linkages occurring every 8 -10 residues. compact structure that results from the coiling of the polymer chains 46
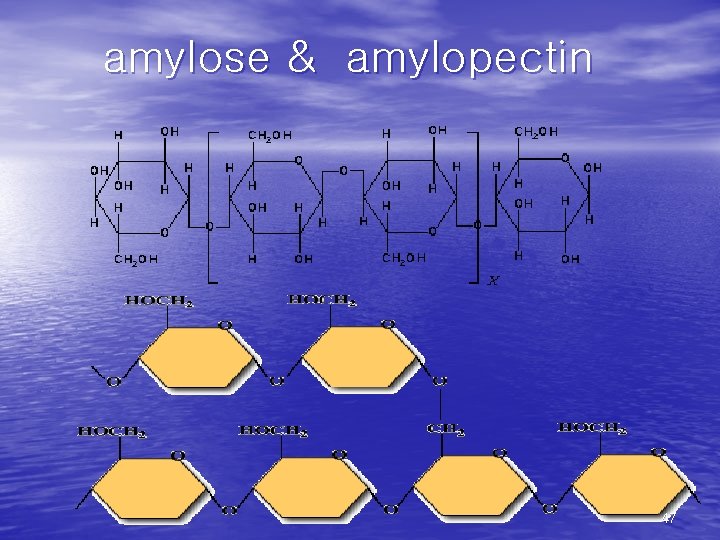
amylose & amylopectin 47
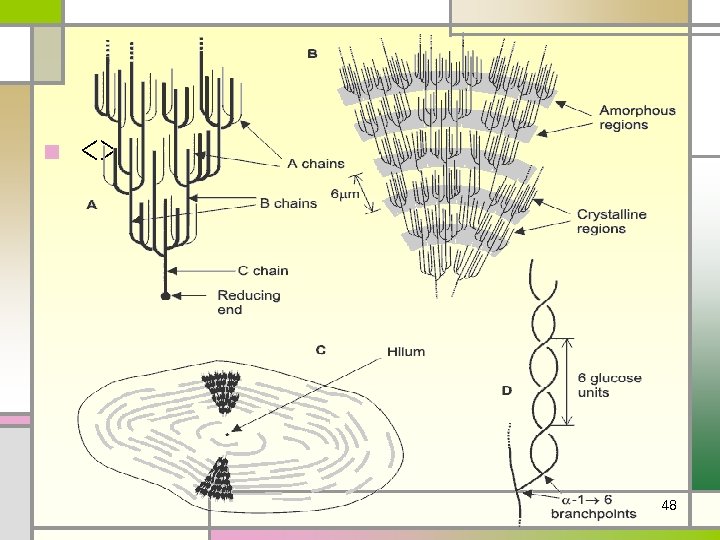
n <> 48
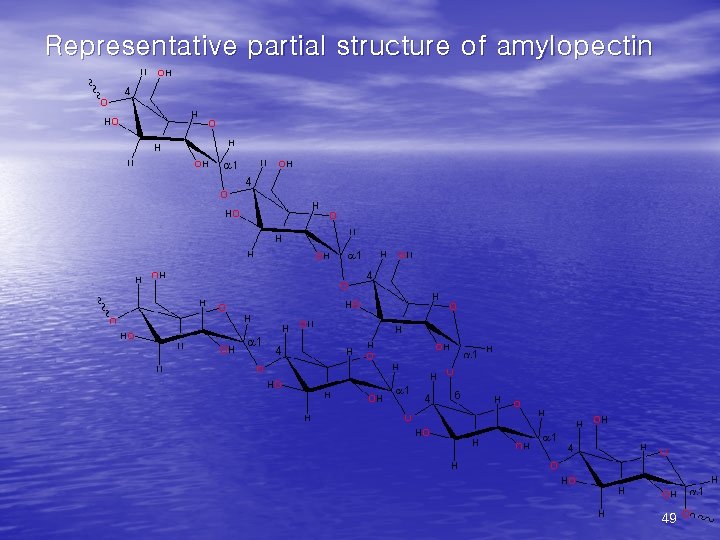
Representative partial structure of amylopectin 49
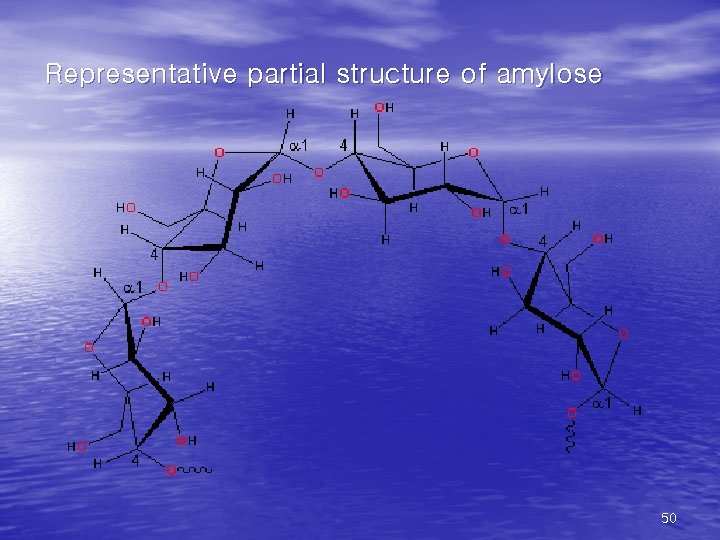
Representative partial structure of amylose 50


52

54


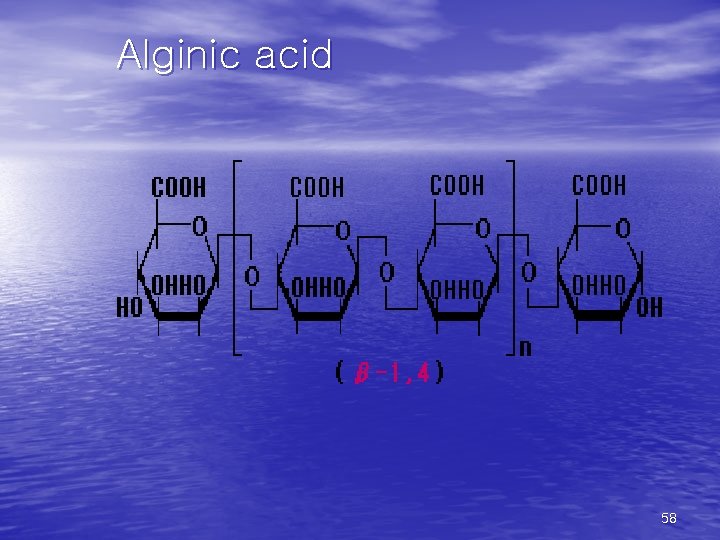
Alginic acid 58

탄수화물의 변화 • Caramelization • Gelatinization • Retrogradation 59






- Slides: 66