1 Epidemiology Prevalence of HBV by Region Epidemiology

1
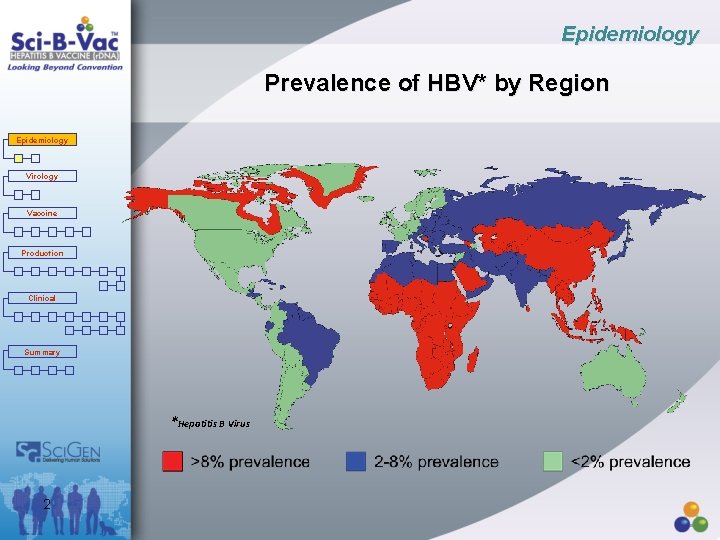
Epidemiology Prevalence of HBV* by Region Epidemiology Virology Vaccine Production Clinical Summary *Hepatitis B Virus 2

Epidemiology Worldwide HBV Infection Epidemiology • More than 2 billion people infected during lifetime Virology • Up to 2 million die each year from HBV infection Vaccine • Worldwide there are ~350 -400 million HBs. Ag carriers Production Clinical Summary • In the WHO European Region, 14 million people are estimated to live with chronic hepatitis B; Up to 36, 000 deaths are attributable to HBV every year in Europe • Annual infection in US ~320, 000, ~32, 000 HBs. Ag carriers • Persistent HBV is considered a significant risk factor for development of primary liver cancer 3
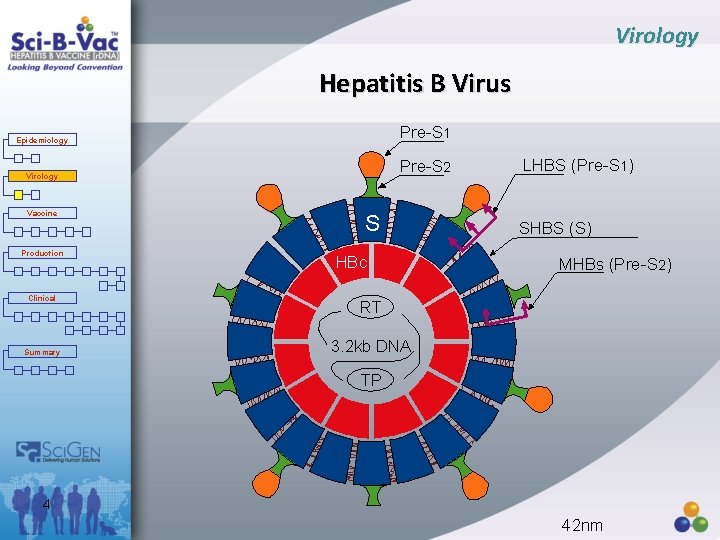
Virology Hepatitis B Virus Pre-S 1 Epidemiology Pre-S 2 Virology Vaccine Production Clinical Summary S HBc LHBS (Pre-S 1) SHBS (S) MHBs (Pre-S 2) RT 3. 2 kb DNA TP 4 42 nm
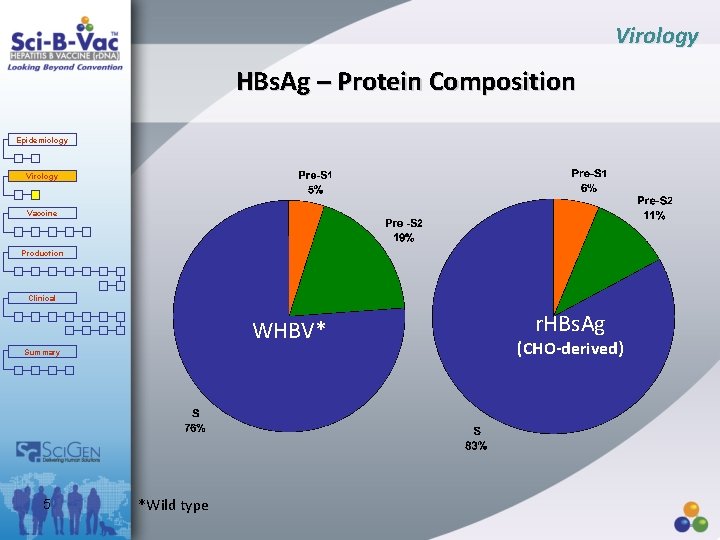
Virology HBs. Ag – Protein Composition Epidemiology Virology Vaccine Production Clinical WHBV* Summary 5 *Wild type r. HBs. Ag (CHO-derived)

Vaccines Factors Which Affect Non-Response to Immunization enhancing Virology • Genetically determined resistance Vaccine • Advanced age Production • Overweight • Gender Clinical Summary • Smoking • Chronic liver diseases Pre-S Epidemiology • Systemic diseases • Immune suppression 6 Craven DE, et al. Ann Int Med 1986; Alper CA, et al. N Eng J Med 1989, Milich DR Immunol Today 1988, Hohler T, et al. Hum Immunol 1998 attenuating
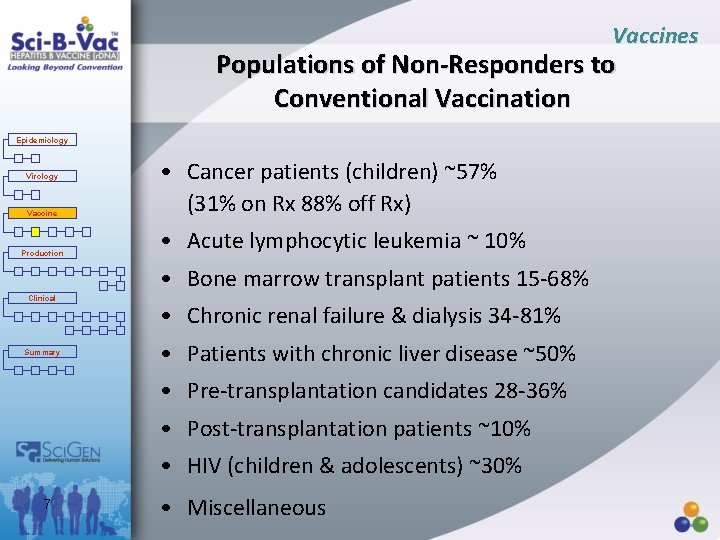
Vaccines Populations of Non-Responders to Conventional Vaccination Epidemiology Virology Vaccine Production • Cancer patients (children) ~57% (31% on Rx 88% off Rx) • Acute lymphocytic leukemia ~ 10% • Bone marrow transplant patients 15 -68% Clinical Summary • Chronic renal failure & dialysis 34 -81% • Patients with chronic liver disease ~50% • Pre-transplantation candidates 28 -36% • Post-transplantation patients ~10% • HIV (children & adolescents) ~30% 7 • Miscellaneous

Vaccines Why Do We Need Better HBV Vaccines? Epidemiology Virology Vaccine Production Clinical Summary • Non–response to conventional HBV vaccines in special populations • Fast induction of immunity to HBV in defined populations • Low compliance with the 3 dose regimen of conventional HBV vaccines • Possible protection against HBV envelope mutant(s) 8

Vaccines History of Hepatitis B Vaccines Epidemiology Virology Vaccine Production Clinical Summary 9 1 st Generation: Plasma-derived -HBs. Ag, Pre-S 2 nd Generation: Yeast-derived, (recombinant) -HBs. Ag, (S Antigen only) 3 rd Generation: Mammalian cell derived (recombinant) -HBs. Ag, Pre-S 2, Pre-S 1
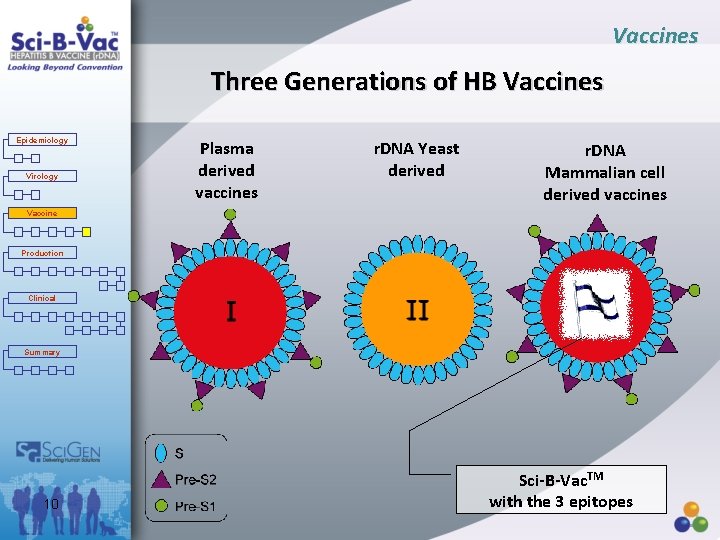
Vaccines Three Generations of HB Vaccines Epidemiology Virology Plasma derived vaccines r. DNA Yeast derived r. DNA Mammalian cell derived vaccines Vaccine Production Clinical Summary 10 Sci-B-Vac. TM with the 3 epitopes
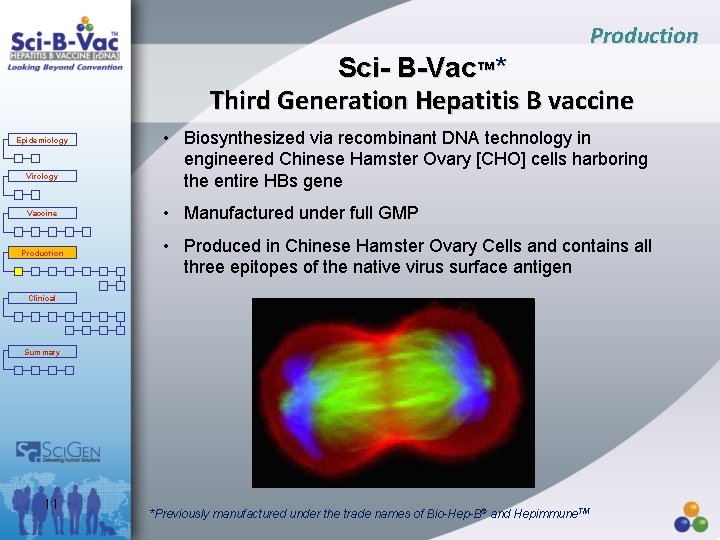
Production Sci- B-Vac™* Third Generation Hepatitis B vaccine Epidemiology Virology Vaccine Production • Biosynthesized via recombinant DNA technology in engineered Chinese Hamster Ovary [CHO] cells harboring the entire HBs gene • Manufactured under full GMP • Produced in Chinese Hamster Ovary Cells and contains all three epitopes of the native virus surface antigen Clinical Summary 11 *Previously manufactured under the trade names of Bio-Hep-B® and Hepimmune. TM
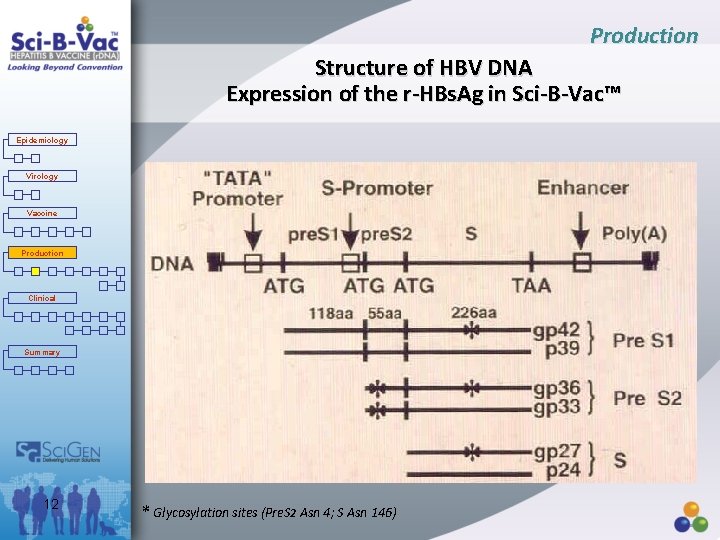
Production Structure of HBV DNA Expression of the r-HBs. Ag in Sci-B-Vac™ Epidemiology Virology Vaccine Production Clinical Summary 12 * Glycosylation sites (Pre. S 2 Asn 4; S Asn 146)

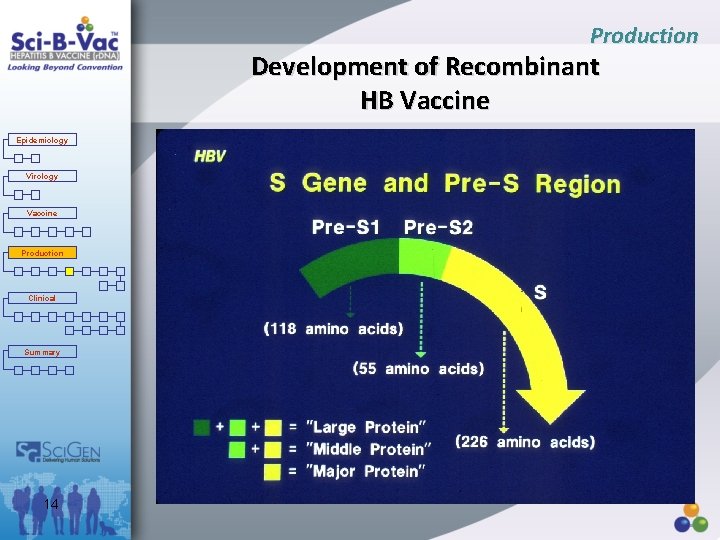
Production Development of Recombinant HB Vaccine Epidemiology Virology HBs gene fragment + Expression Vector (recombinant plasmid) Vaccine Production Clinical r. DNA Transfection Summary Host (CHO cells) Expression 13 HBs Proteins (Pre. S 1, Pre. S 2, S)
Production Development of Recombinant HB Vaccine Epidemiology Virology Vaccine Production Clinical Summary 14
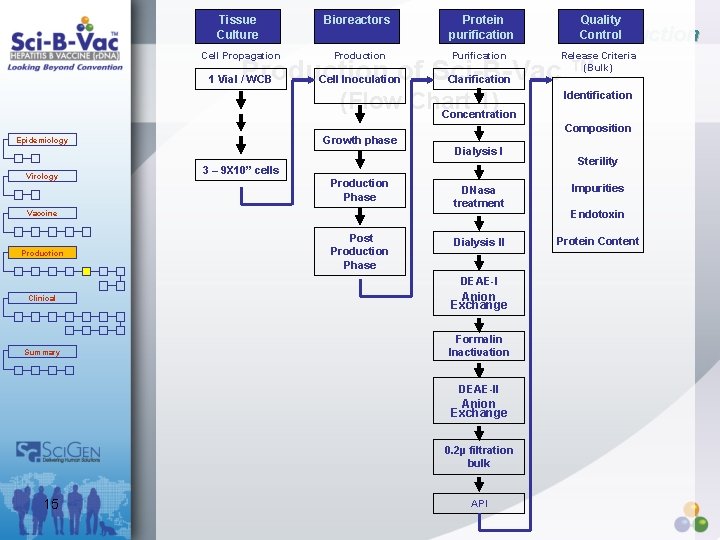
Tissue Culture Bioreactors Protein purification Quality Control Cell Propagation Production Purification 1 Vial / WCB Cell Inoculation Clarification Release Criteria (Bulk) Production of Sci-B-Vac ™ (Flow Chart 1) Concentration Growth phase Epidemiology Virology Vaccine Production Composition Dialysis I 3 – 9 X 10” cells Production Phase Post Production Phase DNasa treatment Dialysis II DEAE-I Clinical Summary Anion Exchange Formalin Inactivation DEAE-II Anion Exchange 0. 2µ filtration bulk 15 Identification API Sterility Impurities Endotoxin Protein Content
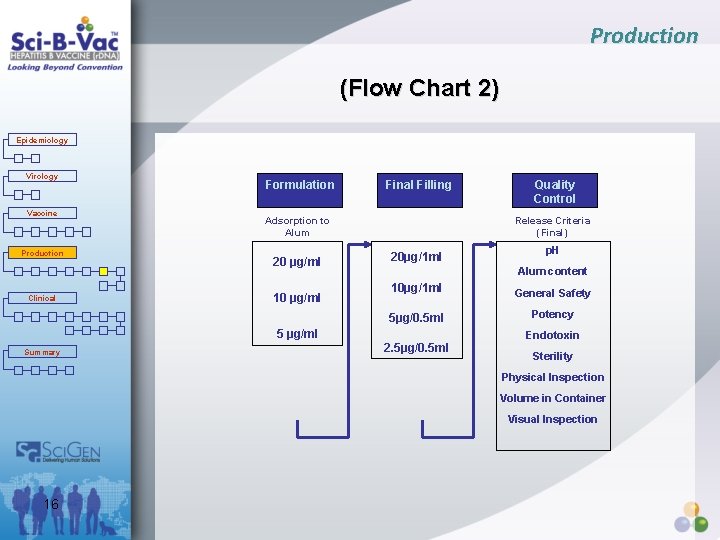
Production (Flow Chart 2) Epidemiology Virology Vaccine Production Clinical Formulation Final Filling Adsorption to Alum 20 µg/ml 10 µg/ml Release Criteria (Final) 20µg/1 ml p. H Alum content 10µg/1 ml General Safety 5µg/0. 5 ml Potency 5 µg/ml Summary Quality Control 2. 5µg/0. 5 ml Endotoxin Sterility Physical Inspection Volume in Container Visual Inspection 16
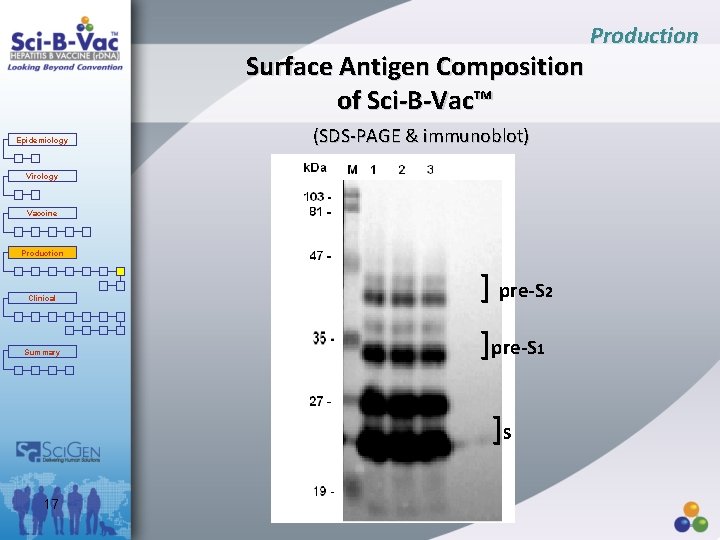
Production Surface Antigen Composition of Sci-B-Vac™ Epidemiology (SDS-PAGE & immunoblot) Virology Vaccine Production Clinical Summary ] pre-S 2 ]pre-S 1 ]S 17
Production CHO Cells in Production Phase Epidemiology Virology Vaccine Production Clinical Summary 18
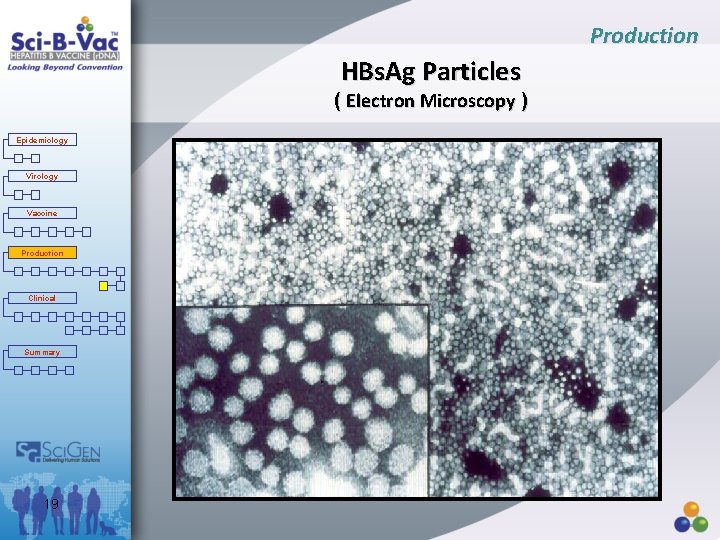
Production HBs. Ag Particles ( Electron Microscopy ) Epidemiology Virology Vaccine Production Clinical Summary 19
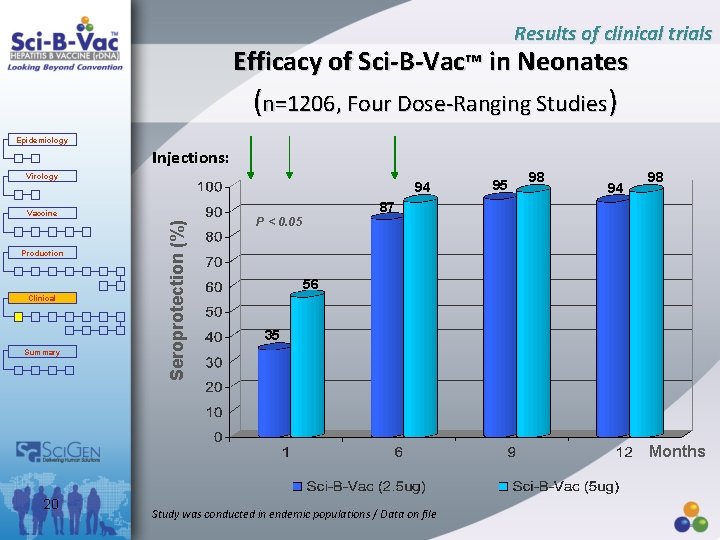
Results of clinical trials Efficacy of Sci-B-Vac™ in Neonates (n=1206, Four Dose-Ranging Studies) Epidemiology Injections: Virology 94 Production Clinical Summary Seroprotection (%) Vaccine 95 98 94 98 87 P < 0. 05 56 35 Months 20 Study was conducted in endemic populations / Data on file
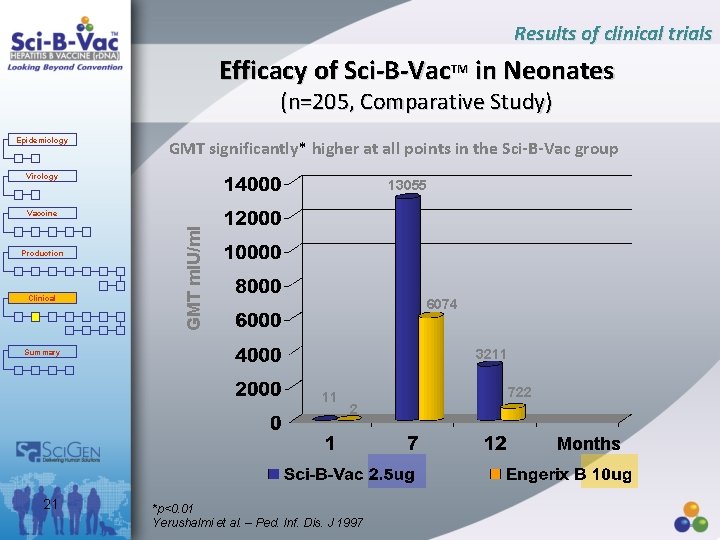
Results of clinical trials Efficacy of Sci-B-Vac. TM in Neonates (n=205, Comparative Study) Epidemiology GMT significantly* higher at all points in the Sci-B-Vac group Virology 13055 Production Clinical GMT m. IU/ml Vaccine 6074 3211 Summary 11 722 2 Months 21 *p<0. 01 Yerushalmi et al. – Ped. Inf. Dis. J 1997
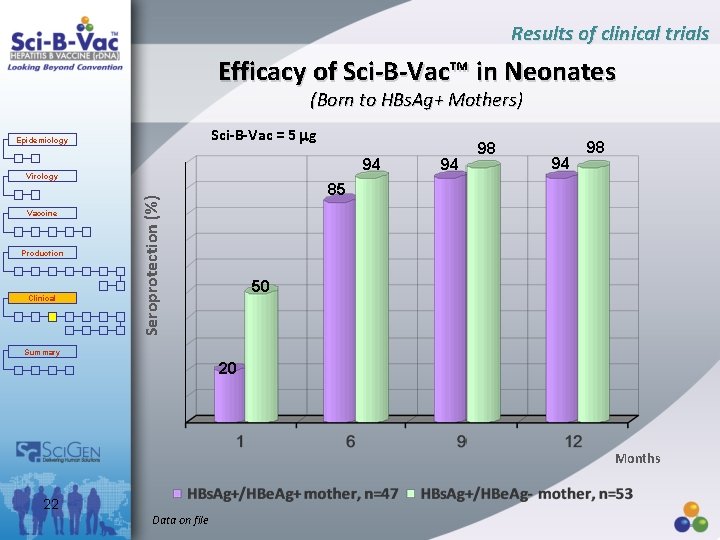
Results of clinical trials Efficacy of Sci-B-Vac™ in Neonates (Born to HBs. Ag+ Mothers) Sci-B-Vac = 5 g Epidemiology 94 Virology Production Clinical 94 98 85 Seroprotection (%) Vaccine 94 98 50 Summary 20 Months 22 Data on file
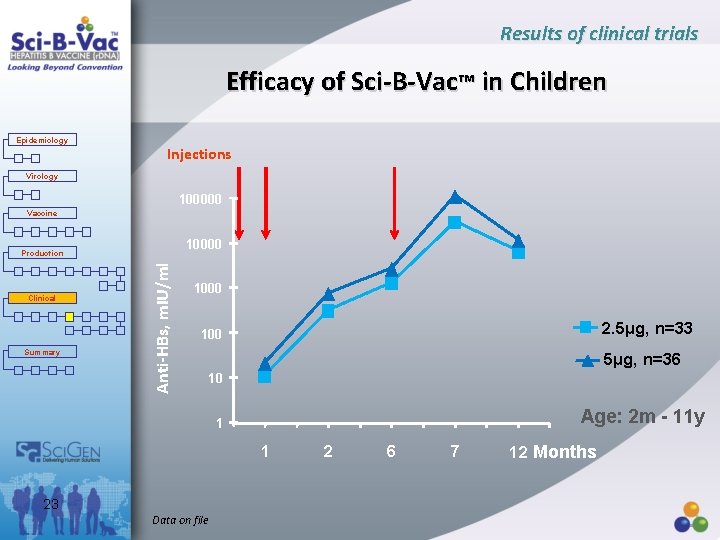
Results of clinical trials Efficacy of Sci-B-Vac™ in Children Epidemiology Injections Virology 100000 Vaccine 10000 Clinical Summary Anti-HBs, m. IU/ml Production 1000 2. 5µg, n=33 100 5µg, n=36 10 Age: 2 m - 11 y 1 1 23 Data on file 2 6 7 12 Months
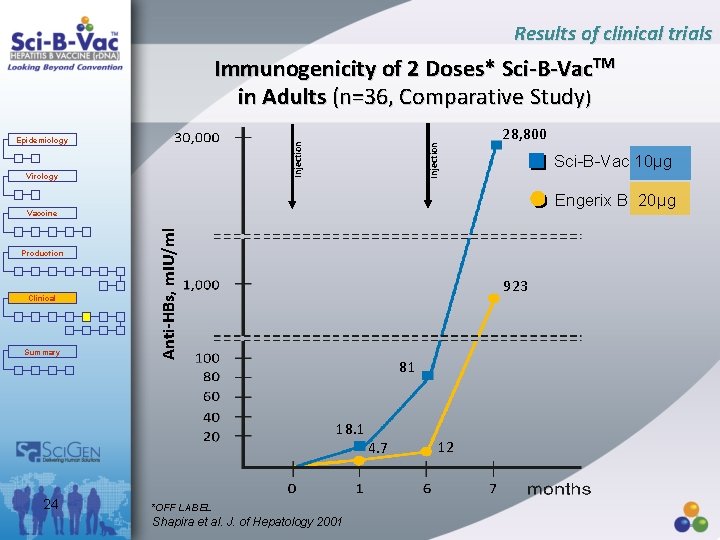
Results of clinical trials Immunogenicity of 2 Doses* Sci-B-Vac. TM in Adults (n=36, Comparative Study) Virology Injection Epidemiology 28, 800 Sci-B-Vac 10µg Engerix B 20µg Production Clinical Summary Anti-HBs, m. IU/ml Vaccine 923 81 18. 1 24 *OFF LABEL Shapira et al. J. of Hepatology 2001 4. 7 12
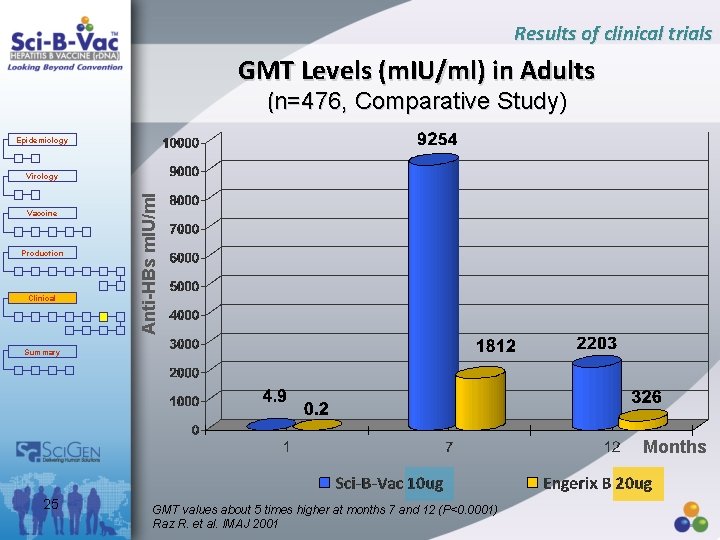
Results of clinical trials GMT Levels (m. IU/ml) in Adults (n=476, Comparative Study) Epidemiology Vaccine Production Clinical Summary Anti-HBs m. IU/ml Virology Months 25 GMT values about 5 times higher at months 7 and 12 (P<0. 0001) Raz R. et al. IMAJ 2001
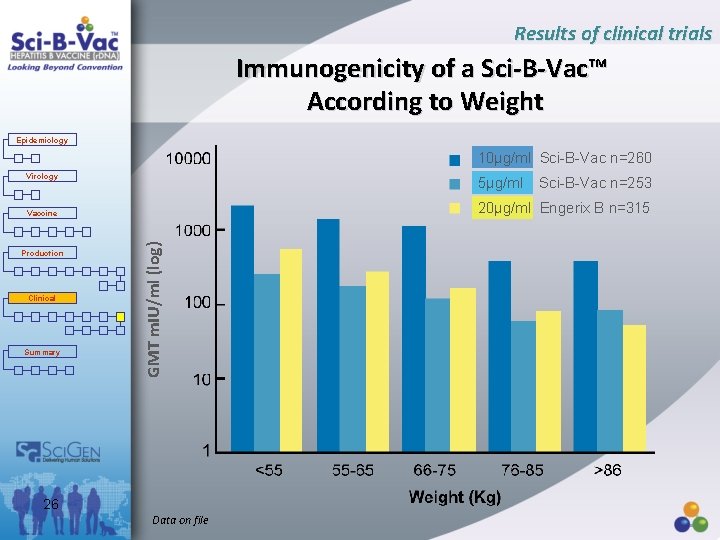
Results of clinical trials Immunogenicity of a Sci-B-Vac™ According to Weight Epidemiology 10µg/ml Sci-B-Vac n=260 Virology 5µg/ml 20µg/ml Engerix B n=315 Clinical Summary GMT m. IU/ml (log) Vaccine Production Sci-B-Vac n=253 26 Data on file

Results of clinical trials Efficacy of Sci-B-Vac™ in Non-and Low Responders to Standard Vaccine (N=719, Comparative Study) Epidemiology Virology • Open, randomized, comparative, controlled, multicenter international parallel group study. Vaccine Production Clinical • To demonstrate superiority of Sci-B-Vac over standard vaccine after one or two additional doses in non-responders ≥ 4 prior injections of standard vaccine. Summary • To evaluate efficacy of the two vaccines in low responders after ≥ 4 and non responders after 3 prior injections of the standard vaccine. 28 Rendi-Wagner P. et al. Vaccine 2006

Results of clinical trials Study Parameters Epidemiology Virology Vaccine Production Clinical Summary • Age: ≥ 18 years • Randomized: 719 • Injected with Sci-B-Vac: 479 • Injected with Engerix B: 237 • Injections: on days 1 and 85 • Adverse Events: 3% Sci-B-Vac vs 0. 8% Engerix B 29 Rendi-Wagner P. et al. Vaccine 2006
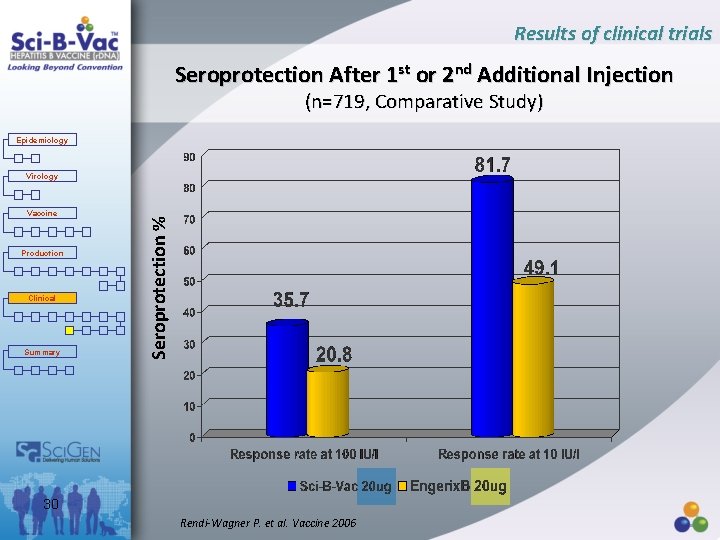
Results of clinical trials Seroprotection After 1 st or 2 nd Additional Injection (n=719, Comparative Study) Epidemiology Vaccine Production Clinical Summary Seroprotection % Virology 30 Rendi-Wagner P. et al. Vaccine 2006

Analyses of 10, 815 Individual Anti-HBs Measurements for 1, 923 Vaccinees Epidemiology Virology Vaccine Production Clinical • The 3 rd generation HBV vaccine Sci-B-Vac and HBV/MF 59 appear the more immunogenic vaccines • Hevac B and HB Vax vaccines appear less immunogenic • Priming of immune memory is successful after a single dose • Immune memory is maintained after anti-HBs antibody decay • Antibody boosts following completion of conventional Summary immunization are unnecessary Vaccines tested: Engerix B, SL*, Bio-Hep- B, China HBV MF/59, Recombivax, Herbiovac, Hevac B, HB Vax 31 *Wilson J. et. al. 2002

Performance of Sci- B-Vac™ in > 20 Clinical Trials Epidemiology Virology Vaccine Production Clinical Summary • Sci-B-Vac is safe and highly immunogenic • Rapid (earlier/higher) immune responses • High seroprotection rates • Tested in adults and children in over 5, 000 subjects • Protects neonates of HBe. Ag positive and negative mothers • Induces seroconversion in high-risk patients and in non-responders 32

Possible Future Applications For Sci-B-Vac™ Epidemiology Virology • Non / hypo responders to standard vaccines Vaccine • Vaccination of health care workers Production Clinical Summary • Immune suppressed patients • Organ donors • Universal vaccination for neonates in endemic countries using less than three doses 33

THANK YOU 34
- Slides: 33