1 Dynamic Processes Irreversible process A B A
1
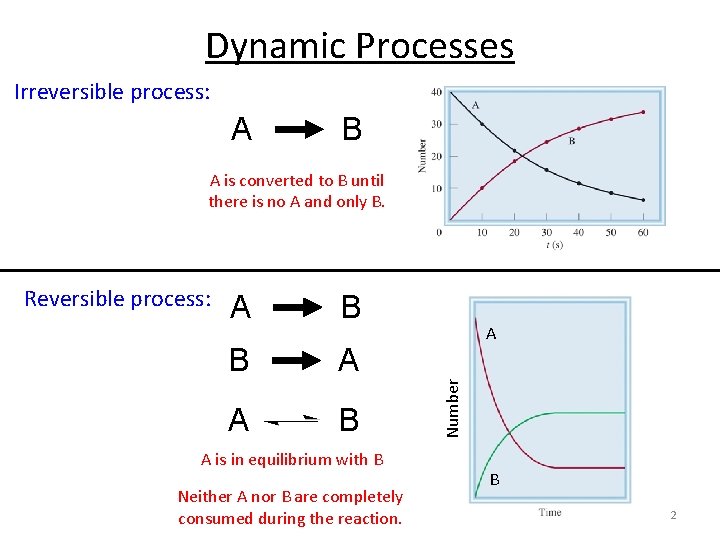
Dynamic Processes Irreversible process: A B A is converted to B until there is no A and only B. A B B A A B A is in equilibrium with B Neither A nor B are completely consumed during the reaction. A Number Reversible process: B 2

• The change is reversible Equilibrium A B • The system is “closed”—no substance can enter or leave • The system is dynamic - At the macroscopic level, it appears as if nothing is happening, but at the particulate level, reversible changes are occurring continuously. • Can be at physical equilibrium or chemical equilibrium Physical Equilibrium- forward and reverse processes occur at the same rate but there is no chemical change. Solubility Vapor Pressure Liquid At time = 0 At time > 0 rate of dissolution = At time = ∞ rate of precipitation At time = 0 At time > 0 rate of = vaporization At time = ∞ rate of condensation 3

• The change is reversible Equilibrium A B • The system is “closed”—no substance can enter or leave • The system is dynamic - At the macroscopic level, it appears as if nothing is happening, but at the particulate level, reversible changes are occurring continuously • Can be at physical equilibrium or chemical equilibrium Chemical Equilibrium • the rates of the forward and reverse reactions are equal. • a chemical change is occurring (intramolecular bonds broken/formed) • concentrations of the reactants and products remain constant. • this does not mean [conc] of reactants and products are equal! NO 2 N 2 O 4 (g) 2 NO 2 (g) 4
![Equilibrium and Rate A B rate. A B = k. A [A] B A Equilibrium and Rate A B rate. A B = k. A [A] B A](http://slidetodoc.com/presentation_image/a61a499e800a4f0a64789c851d81ed5e/image-5.jpg)
Equilibrium and Rate A B rate. A B = k. A [A] B A rate. B A = k. B [B] Number B A 0 1 2 4 n time At equilibrium! At time ~0: [B] = 0, [A] > 0 B formation = k. A[A] At time 1 [B] > 0, [A] > 0 rate. A B >> rate. B A At time 2 [B] = [A] k. A/rate. A B = k. B / rate. B A At time 4 [B] > 0, [A] > 0 rate. A B > rate. B A At time n ∞ [B] unchanged, [A] unchanged rate. A B = rate. B A 5
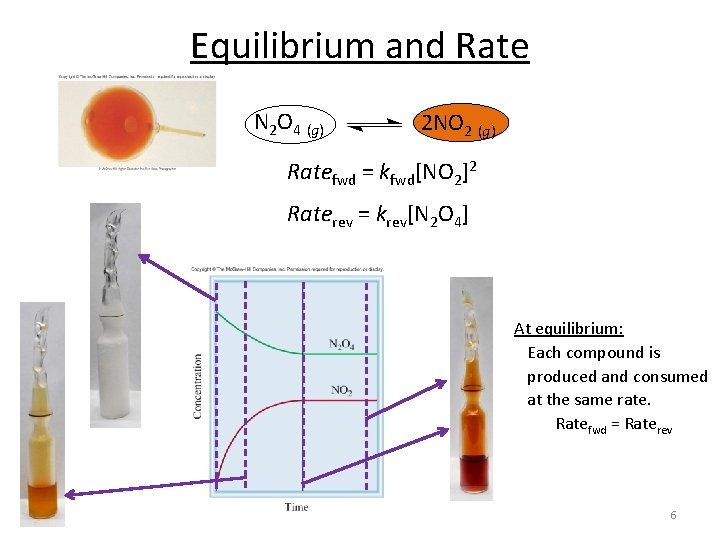
Equilibrium and Rate N 2 O 4 (g) 2 NO 2 (g) Ratefwd = kfwd[NO 2]2 Raterev = krev[N 2 O 4] At equilibrium: Each compound is produced and consumed at the same rate. Ratefwd = Raterev 6
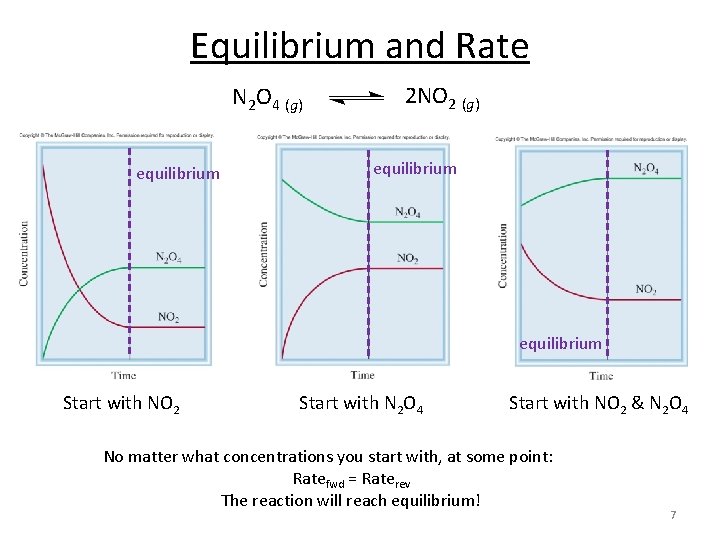
Equilibrium and Rate N 2 O 4 (g) equilibrium 2 NO 2 (g) equilibrium Start with NO 2 Start with N 2 O 4 Start with NO 2 & N 2 O 4 No matter what concentrations you start with, at some point: Ratefwd = Raterev The reaction will reach equilibrium! 7
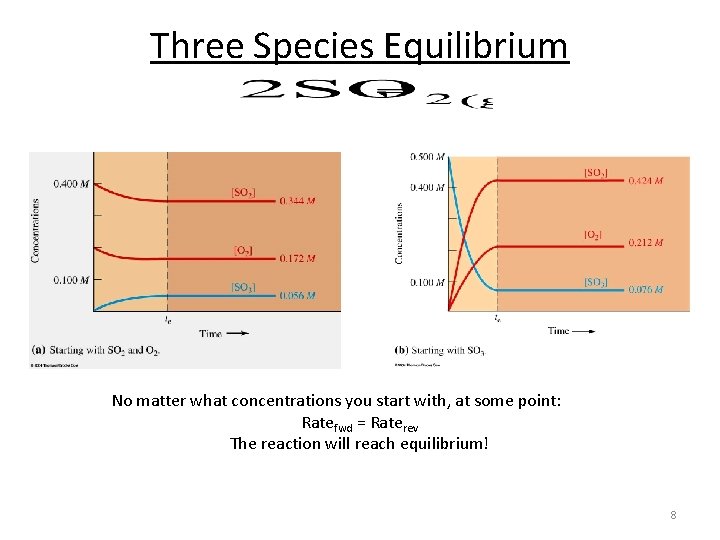
Three Species Equilibrium No matter what concentrations you start with, at some point: Ratefwd = Raterev The reaction will reach equilibrium! 8
9

Equilibrium Concentrations A B • Equilibrium does not mean that concentrations are all equal!! • However, concentrations at equilibrium are related to the fwd/rev rate constants. • Every equilibrium has its own equilibrium constant. rate. A B = k. A [A] rate. B A = k. B [B] At equilibrium: rate. A B = rate. B A Rate constant A B Rate constant B A k. A [A] = k. B [B] k. A k. B = [B] [A] = K 10
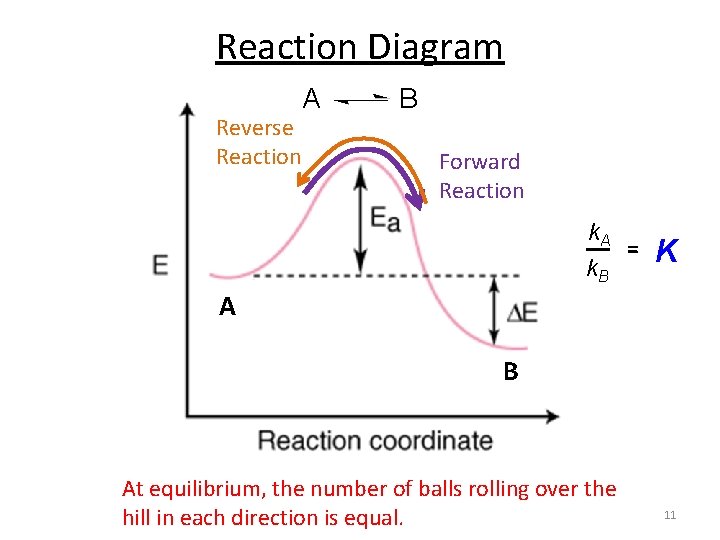
Reaction Diagram Reverse Reaction A B Forward Reaction k. A k. B = K A B At equilibrium, the number of balls rolling over the hill in each direction is equal. 11

Equilibrium Constant For a general reversible reaction such as: a. A + b. B c. C + d. D the equilibrium constant is written as Law of Mass Action The equilibrium constant for the overall reaction includes stoichiometric coefficients. General practice is not to include units for the equilibrium constant. 12
![Equilibrium Constant N 2 O 4 (g) 2 NO 2 (g) [NO 2]2 K Equilibrium Constant N 2 O 4 (g) 2 NO 2 (g) [NO 2]2 K](http://slidetodoc.com/presentation_image/a61a499e800a4f0a64789c851d81ed5e/image-13.jpg)
Equilibrium Constant N 2 O 4 (g) 2 NO 2 (g) [NO 2]2 K = [N 2 O 4] K = 4. 6 x 10 -3 13
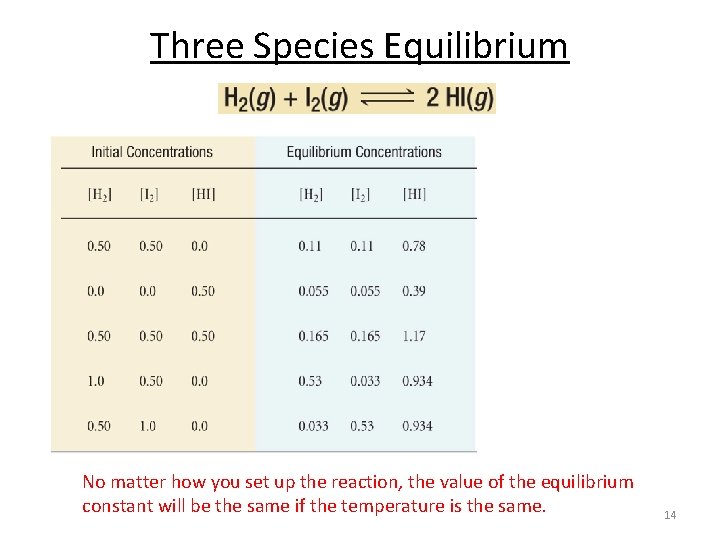
Three Species Equilibrium No matter how you set up the reaction, the value of the equilibrium constant will be the same if the temperature is the same. 14
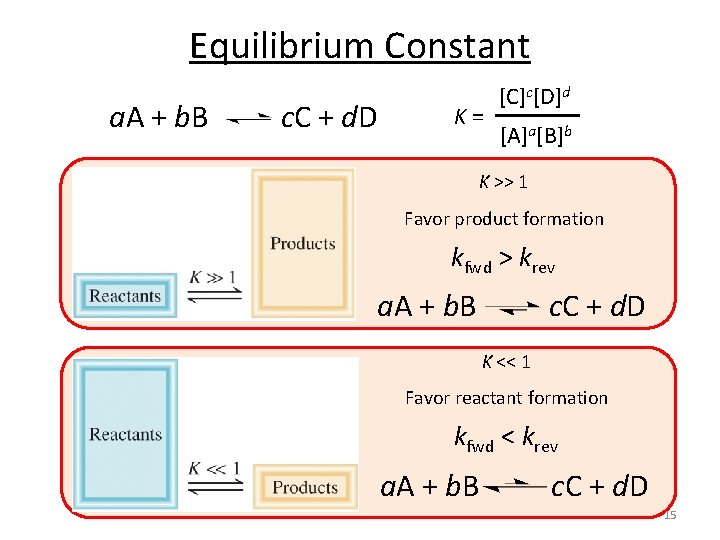
Equilibrium Constant a. A + b. B c. C + d. D K = [C]c[D]d [A]a[B]b K >> 1 Favor product formation kfwd > krev a. A + b. B c. C + d. D K << 1 Favor reactant formation kfwd < krev a. A + b. B c. C + d. D 15

Equilibrium Constant A) B) C) D) Question 1) Which reaction most favors the formation of products? 2) Which reaction most favors the formation of reactants? 16

Equilibrium Constant For a general reversible reaction such as: a. A + b. B c. C + d. D • Equilibrium constants can be expressed using Kc or Kp. • Kc uses the concentration of reactants and products. [C]c [D]d Kc = a b [A] [B] • Kp uses the pressure of the gaseous reactants and products. P Cc P Dd Kp = a b PA PB 17

Equilibrium N 2 O 4 (g) In terms of pressure (KP) In terms of concentration (Kc) Kc = 2 NO 2 (g) [NO 2]2 PNO 2 Kp = P 2 NO [N 2 O 4] 2 4 Kc Kp …at least not always! 18
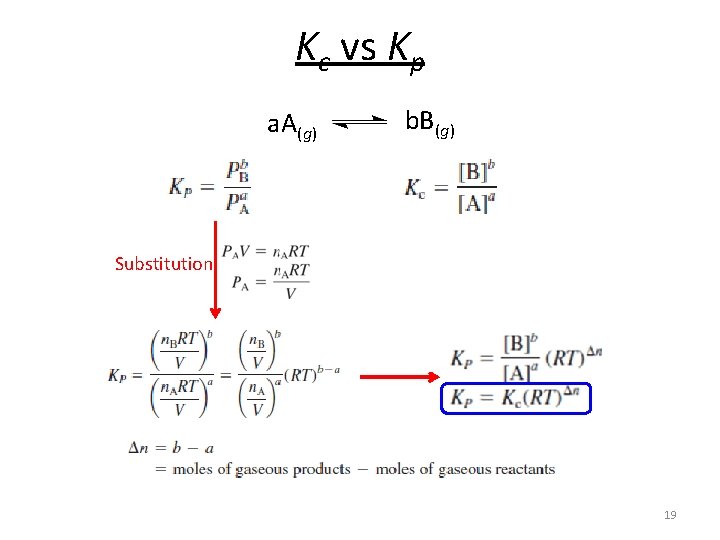
Kc vs Kp a. A(g) b. B(g) Substitution 19
![Kc vs Kp a. A + b. B c. C + d. D [C]c Kc vs Kp a. A + b. B c. C + d. D [C]c](http://slidetodoc.com/presentation_image/a61a499e800a4f0a64789c851d81ed5e/image-20.jpg)
Kc vs Kp a. A + b. B c. C + d. D [C]c [D]d Kc = a b [A] [B] P Cc P Dd Kp = a b PA PB Kp = Kc(RT)Dn Dn = moles of gaseous products – moles of gaseous reactants Dn = (c + d) – (a + b) When does Kp equal Kc? 20
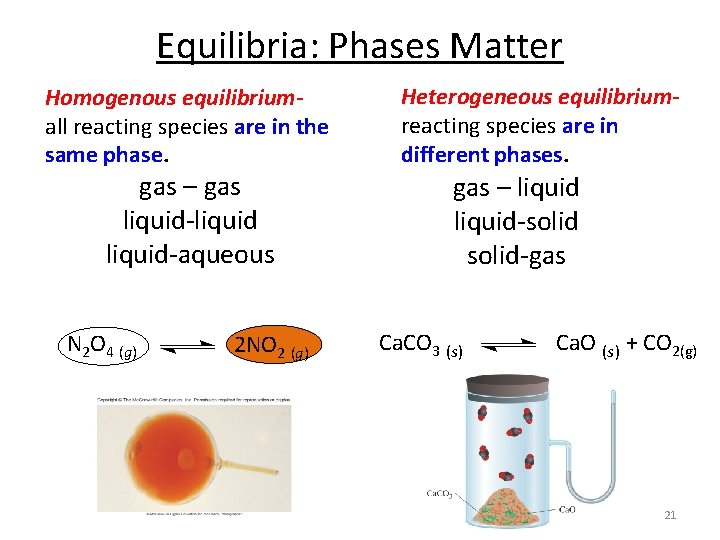
Equilibria: Phases Matter Homogenous equilibriumall reacting species are in the same phase. gas – gas liquid-liquid-aqueous N 2 O 4 (g) 2 NO 2 (g) Heterogeneous equilibriumreacting species are in different phases. gas – liquid-solid-gas Ca. CO 3 (s) Ca. O (s) + CO 2(g) 21

Homogenous Equilibria -all reacting species are in the same phase. gas-gas equilibria N 2(g) + 3 H 2(g) ⇄ 2 NH 3(g) Kc = H 2 O is often used as a solvent(l). However, if H 2 O is written as a gas (g), then its concentration must be considered in Kc and Kp. [NH 2]2 [N 2][H 2]3 PNH 32 Kp = PN 2 PH 23 CH 4(g) + 3 H 2 O(g) ⇄ CO(g) + 3 H 2(g) Kc = [CO][H 2]2 [CH 4][H 2 O]3 PCO PH 2 Kp = PCH 4 PH 2 O 3 22
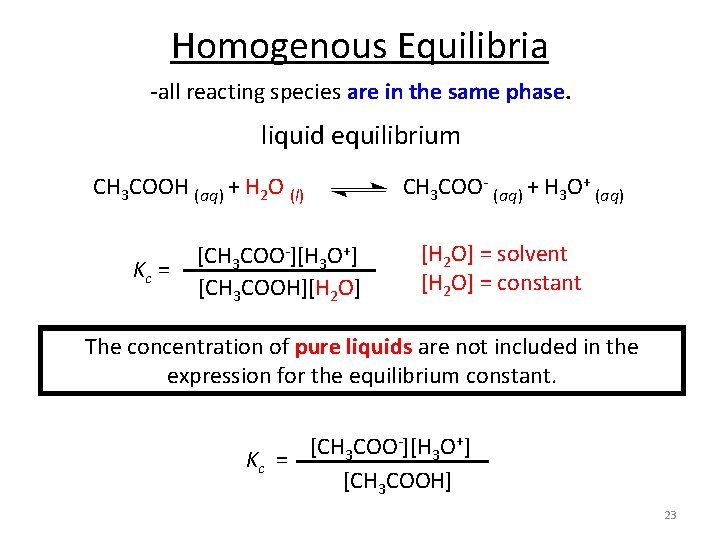
Homogenous Equilibria -all reacting species are in the same phase. liquid equilibrium CH 3 COOH (aq) + H 2 O (l) CH 3 COO- (aq) + H 3 O+ (aq) Kc = [CH 3 COO-][H 3 O+] [CH 3 COOH][H 2 O] = solvent [H 2 O] = constant The concentration of pure liquids are not included in the expression for the equilibrium constant. [CH 3 COO-][H 3 O+] Kc = [CH 3 COOH] 23

Question Write expressions for the following reversible reactions at equilibrium: 1) HF(aq) + H 2 O(l) H 3 O+(aq) + F-(aq) A) Kc = [H 3 O+][F-] [HF][H 2 O] B) Kc = [HF] [F-][H 3 O+ ] C) Kc = [F-][H 3 O+] [HF] 2) 2 NO(g) + O 2(g) 2 NO 2(g) A) Kc = [NO 2]2 [O 2 ][NO]2 B) Kc = [NO 2] [O 2][NO] C) KP = PNO 22 PO 2 PNO 2 3) CH 3 COOH(aq) + C 2 H 5 OH(aq) CH 3 COOC 2 H 5(aq) + H 2 O(l) A) Kc = [CH 3 COOC 2 H 5][H 2 O] [CH 3 COOH][C 2 H 5 OH] B) Kc = [CH 3 COOC 2 H 5] [CH 3 COOH][C 2 H 5 OH] 24

Heterogenous Equilibria -reacting species are in different phases. • Can include liquids, gases and solids as either reactants or products. Ca. CO 3 (s) Ca. O (s) + CO 2 (g) Note Ca. O will not form a neat separated pile! time 25
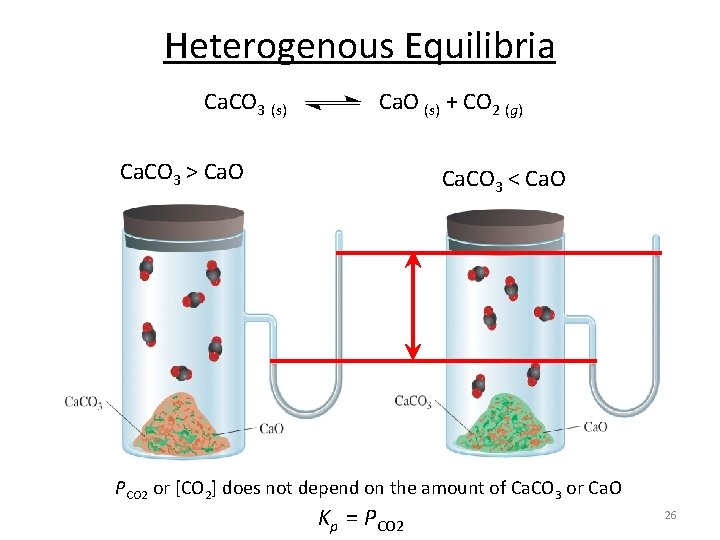
Heterogenous Equilibria Ca. CO 3 (s) Ca. O (s) + CO 2 (g) Ca. CO 3 > Ca. O Ca. CO 3 < Ca. O PCO 2 or [CO 2] does not depend on the amount of Ca. CO 3 or Ca. O Kp = PCO 2 26

Heterogenous Equilibria Ca. CO 3 (s) Ca. O (s) + CO 2 (g) Kc = [Ca. O][CO 2] [Ca. CO 3] Activity- is a measure of the “effective concentration” of a species in a mixture. Activity of a solid = 1 Kc = [CO 2] [1] Kp = PCO 2 The concentration of solids are not included in the expression for the equilibrium constant. 27

Consider the following heterogeneous equilibrium: Ca. CO 3(s) Ca. O(s) + CO 2(g) At 800°C, the pressure of CO 2 is 0. 236 atm. Calculate: (a) KP Given T and PCO 2 Kp = PCO 2 Find Kp KP = 0. 236 (b) Kc Have T and Kp Find Kc Kp = Kc(RT)Dn R = 0. 0821 L atm/K mol T = 273 + 800 = 1073 K Dn = 1 – 0 = 1 0. 236 = Kc(0. 0821 x 1073)1 Kc = 2. 68 x 10 -3 28

Liquids and Solids The concentration of pure liquids are not included in the expression for the equilibrium constant. CH 3 COOH (aq) + H 2 O (l) CH 3 COO- (aq) + H 3 O+ (aq) [CH 3 COO-][H 3 O+] Kc = [CH 3 COOH] The concentration of solids are not included in the expression for the equilibrium constant. Ca. CO 3 (s) Ca. O (s) + CO 2 (g) Kc = [CO 2] Kp = PCO 2 29

Writing Equilibrium Constant Expressions 1. The concentrations of the reacting species in the condensed phase are expressed in M. In the gaseous phase, the concentrations can be expressed in M or in atm. 2. The concentrations of pure solids, pure liquids and solvents do not appear in the equilibrium constant expressions. 3. The equilibrium constant is a dimensionless quantity. 4. In quoting a value for the equilibrium constant, you must specify the balanced equation and the temperature. 30
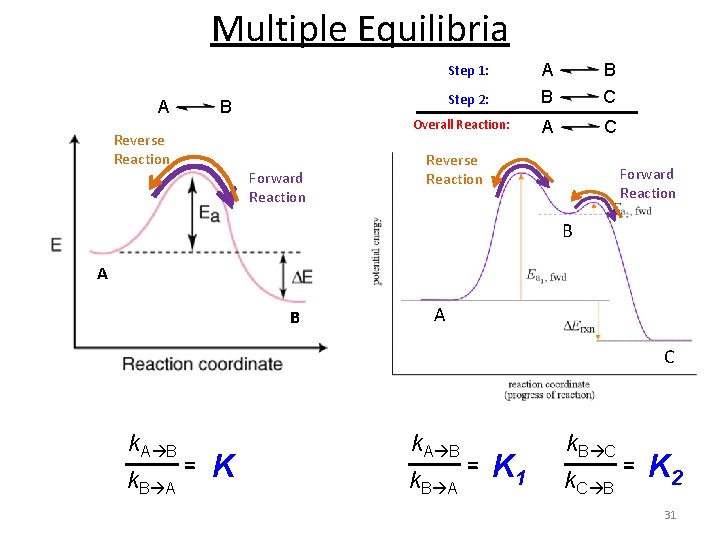
Multiple Equilibria Step 1: A Step 2: B Reverse Reaction Overall Reaction: Forward Reaction A B B C A C Reverse Reaction Forward Reaction B A C k. A B k. B A = K 1 k. B C k. C B = K 2 31

Multiple Equilibria – A reaction can be an individual reaction step or a multistep reaction. – If the overall reaction is the sum of two or more reactions, the overall reaction Equilibrium Constant is the product of the Equilibrium Constants for the steps 32

Multiple Equilibria Product molecules of one equilibrium constant are involved in a second equilibrium process. A + B C + D E + F A + B E + F Kc‘‘ Kc [C][D] x [E][F] Kc = Kc‘ x Kc‘‘ = [A][B] [C][D] Kc‘ = [A][B] [E][F] Kc‘‘ = [C][D] [E][F] Kc = [A][B] If a reaction can be expressed as the sum of two or more reactions, the equilibrium constant for the overall reaction is given by the product of the equilibrium constants of the individual reactions. 33

Practice Problem Determine the overall equilibrium constant for the reaction between Nitrogen & Oxygen to form the toxic gas Nitrogen Dioxide – a component of atmospheric smog N 2(g) + 2 NO(g) + Overall: O 2(g) ⇆ 2 NO(g) O 2(g) ⇆ 2 NO 2(g) N 2(g) + 2 O 2(g) ⇆ Kc 1 Kc 2 2 NO 2(g) 34

Multiple Equilibria Overall Equilibrium: Kc Equilibrium 1: Equilibrium 2: Given K’c and K”c what is Kc? x 35

Rules for Manipulating K • The overall reaction equilibrium constant (K) is the product of the equilibrium constants for the steps. • If the equation is reversed, the equilibrium constant is inverted. • If the equation is multiplied by a factor, the equilibrium constant is raised to the same factor.

Writing Equilibrium Constant Expressions 1. The concentrations of the reacting species in the condensed phase are expressed in M (Kc). In the gaseous phase, the concentrations can be expressed in M (Kc) or in atm (Kp). 2. The concentrations of pure solids, pure liquids and solvents DO NOT appear in the equilibrium constant expressions. 3. The equilibrium constant is a dimensionless quantity. 4. In quoting a value for the equilibrium constant, you must specify the balanced equation and the temperature. 5. If a reaction can be expressed as a sum of two or more reactions, the equilibrium constant for the overall reaction is given by the product of the equilibrium constants of the individual reactions. 37
38
- Slides: 38