1 Define SI Base Units for Mass Length


1. Define SI Base Units for Mass, Length, Time, Temperature, Amount of a substance, Electric Current, and Luminous Intensity 2. Explain how adding a prefix changes a unit. 3. Perform Density and Temperature Calculations. 4. Perform Unit Conversions using Factor Label Method. 5. Convert data from standard form to scientific notation and from scientific notation to standard form. 6. Use various measuring devices to determine precision and accuracy.
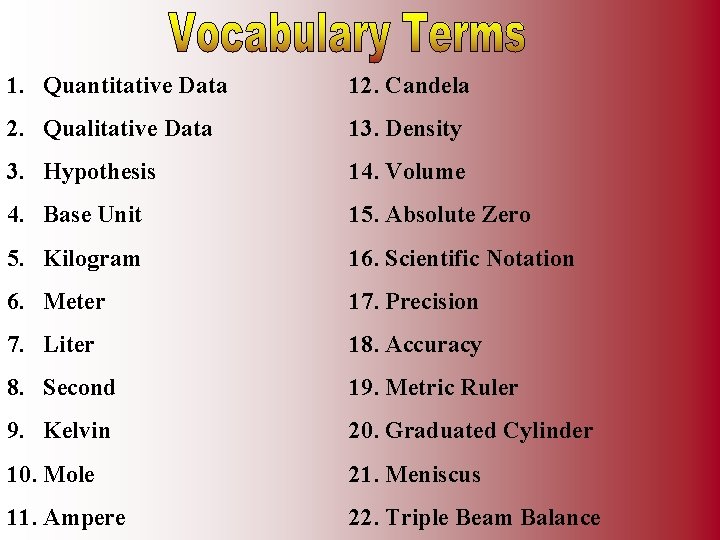
1. Quantitative Data 12. Candela 2. Qualitative Data 13. Density 3. Hypothesis 14. Volume 4. Base Unit 15. Absolute Zero 5. Kilogram 16. Scientific Notation 6. Meter 17. Precision 7. Liter 18. Accuracy 8. Second 19. Metric Ruler 9. Kelvin 20. Graduated Cylinder 10. Mole 21. Meniscus 11. Ampere 22. Triple Beam Balance


Scientific Law – A rule or principle describing a physical relationship or event that is observed and experimented, and NEVER changes. Scientific Theory – A set of widely accepted explanations of observations and phenomena, CAN change if there is consistent evidence. Scientific Hypothesis – A tentative explanation for an observation or phenomenon, used to make predictions.

Qualitative Data: Observations made by scientists that describes the information using the five senses – sight, smell, taste, sound, touch. Examples: Mr. A is wearing a green shirt, Mr. A has a loud voice, A Lemon has a sour taste, Tape feels sticky, etc. Quantitative Data: Observations made by scientists that describes the numerical value of information. Examples: Mr. A is 6’ 1” tall, Mr. A has 10 fingers and toes, The classroom is 78 degrees Fahrenheit, You got 9 out 10 correct on the Lab Safety Test, etc.

When you do something with an observation, like draw a conclusion, offer an explanation or decide that a chemical reaction occurred, then you are making an inference. An observation is the awareness of some condition; inference is the result of a mental process which attempts to explain or catalog or speculate about that observation.
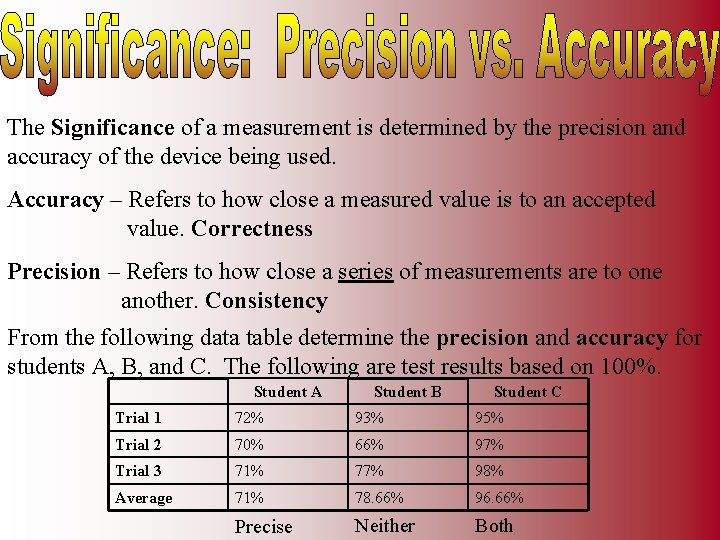
The Significance of a measurement is determined by the precision and accuracy of the device being used. Accuracy – Refers to how close a measured value is to an accepted value. Correctness Precision – Refers to how close a series of measurements are to one another. Consistency From the following data table determine the precision and accuracy for students A, B, and C. The following are test results based on 100%. Student A Student B Student C Trial 1 72% 93% 95% Trial 2 70% 66% 97% Trial 3 71% 77% 98% Average 71% 78. 66% 96. 66% Precise Neither Both

Base Unit: A defined unit in a system of measurement based on an object or event in the physical world. Quantity Mass Base Unit Kilogram (kg) Length Meter (m) Time Second (s) Temperature Kelvin (K) Amount of a substance Mole (mol) Electric Current Ampere (A) Luminous Intensity Candela (cd)
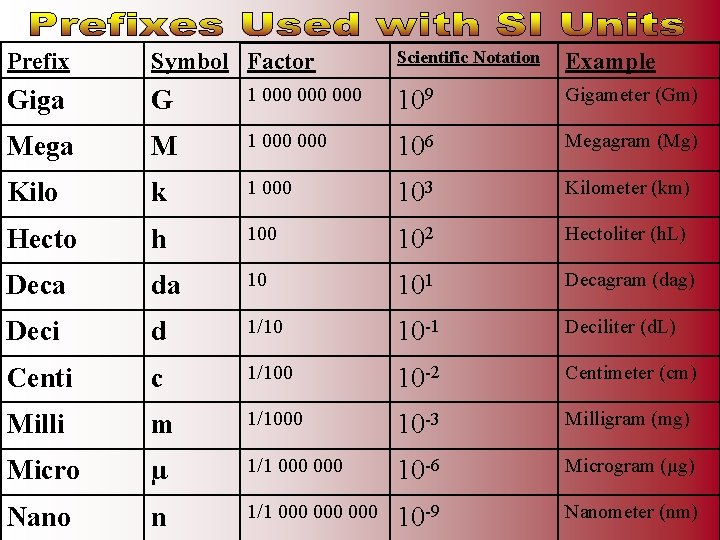
Scientific Notation Example 1 000 000 109 Gigameter (Gm) M 1 000 106 Megagram (Mg) Kilo k 1 000 103 Kilometer (km) Hecto h 100 102 Hectoliter (h. L) Deca da 10 101 Decagram (dag) Deci d 1/10 10 -1 Deciliter (d. L) Centi c 1/100 10 -2 Centimeter (cm) Milli m 1/1000 10 -3 Milligram (mg) Micro µ 1/1 000 10 -6 Microgram (µg) Nano n 1/1 000 000 10 -9 Nanometer (nm) Prefix Symbol Factor Giga G Mega

1 Dozen eggs = 12 eggs 12 inches = 1 foot 60 seconds = 1 minute 16 ounces = 1 pound Factor Label Method: A way to convert a ratio of equivalent values to express the same quantity in different units. Examples: 24 inches = ____? ____ feet 24 inches 1 feet 12 inches Given Units to Get to Units to Cancel = 2 feet Equivalent Ratio
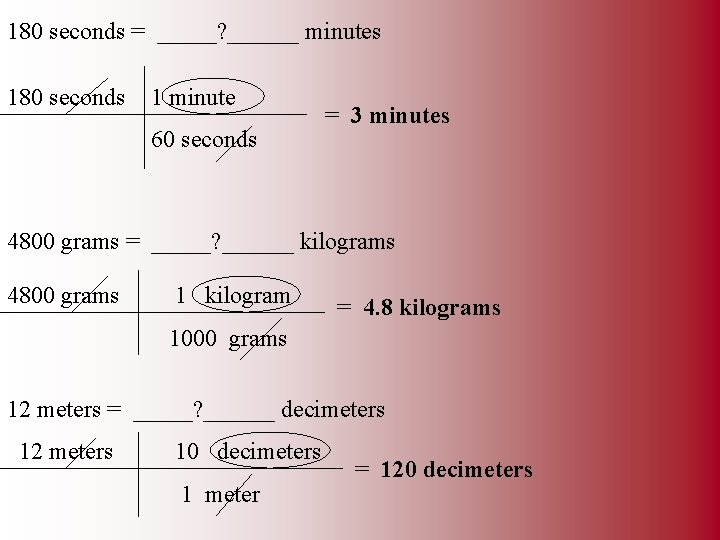
180 seconds = _____? ______ minutes 180 seconds 1 minute 60 seconds = 3 minutes 4800 grams = _____? ______ kilograms 4800 grams 1 kilogram = 4. 8 kilograms 1000 grams 12 meters = _____? ______ decimeters 12 meters 10 decimeters 1 meter = 120 decimeters
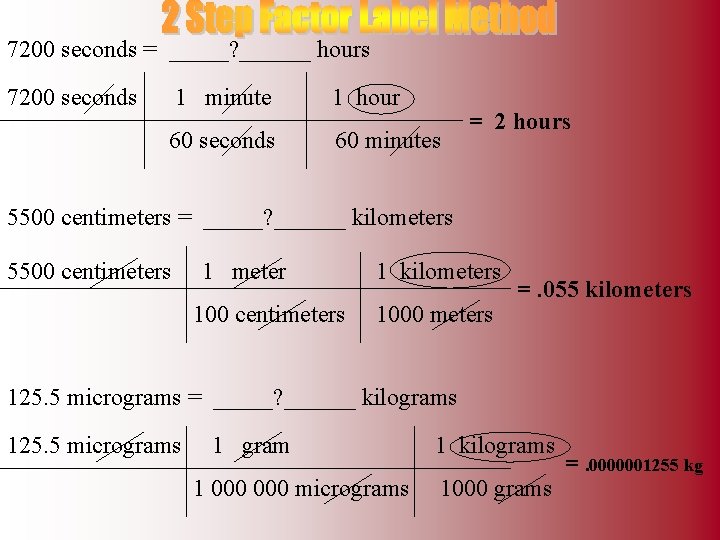
7200 seconds = _____? ______ hours 7200 seconds 1 minute 1 hour 60 seconds 60 minutes = 2 hours 5500 centimeters = _____? ______ kilometers 5500 centimeters 1 meter 100 centimeters 1 kilometers 1000 meters =. 055 kilometers 125. 5 micrograms = _____? ______ kilograms 125. 5 micrograms 1 gram 1 000 micrograms 1 kilograms 1000 grams =. 0000001255 kg

Volume: the amount of space occupied by an object. Can measured in Liters, milliliters, ounces, quarts, pints, gallons, and cubic centimeters (cm 3). Volume = length x width x height Density: the mass per unit volume of a given material. Equation: Density = Mass / Volume Examples – Water has a density of 1. 0 g/m. L Ice has a density of 0. 92 g/m. L Aluminum has density of 2. 7 g/m. L What has a greater density oil or water? What has a greater density, a brick or a pillow?

Hot and Cold are qualitative descriptions of temperature. Using the three different temperature scales describes a quantitative observation. The three temperature scales include; Celsius, Fahrenheit, and Kelvin 1. °C = (°F – 32) / 1. 8 Known Values for Water 2. °F = 1. 8(°C) + 32 Freezing Point of Water = 0 °C, 32 °F, 273 K 3. K = °C + 273 Boiling Point of Water = 100 °C, 212 °F, 373 K Absolute Zero – The temperature at which a substance would have zero kinetic energy, particles are not moving at all. (0 K, -273 °C, - 459. 4 °F) Do the following Temperature Conversions: 111. 2 °F 40 °C a. 44 °C = _______ b. 104 °F = _______ 398 c. 125 °C = ______K

Scientific Notation: expresses numbers as a multiple of two factors; a number between 1 and 10 (M); and 10 raised to a power, or exponent (n) General Formula: M x 10 n M = a number between 1 -10 but cannot be 10 (1 - 9. 999) n = the number of decimal places moved Examples: 1, 000 = 1. 0 x 103 150, 000 = 1. 5 x 105 498 = 4. 98 x 102 8, 542, 327 = 8. 542327 x 106 0. 005 = 5. 0 x 10 -3 0. 04005 = 4. 005 x 10 -2 0. 0000652 = 6. 52 x 10 -5 100 = 1 so 4. 1 x 100 = 4. 1
- Slides: 16