1 Covalent Bonding Takes place between two nonmetals

1

Covalent Bonding ● ● ● Takes place between two non-metals Electrons are shared by both nuclei to help each attain 8 valence electrons There are several electrostatic interactions in these bonds: ● ● Attractions between electrons and nuclei Repulsions between electrons Repulsions between nuclei A molecule is formed when two or more atoms bond covalently Covalent Bond Video (25 sec) Ionic and Covalent (2 min) 2

Drawing the Lewis Dot Diagram of a molecule helps determine the specific properties. Properties of Covalent Compounds ● ● ● Most have low melting points and low boiling points. Many are liquids and gases at room temperature. Nonconductors of electricity. AKA Nonelectrolytes
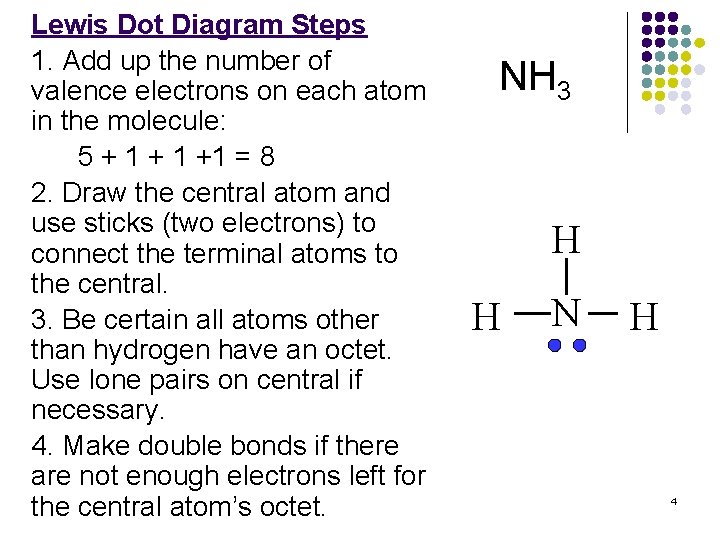
Lewis Dot Diagram Steps 1. Add up the number of valence electrons on each atom in the molecule: 5 + 1 +1 = 8 2. Draw the central atom and use sticks (two electrons) to connect the terminal atoms to the central. 3. Be certain all atoms other than hydrogen have an octet. Use lone pairs on central if necessary. 4. Make double bonds if there are not enough electrons left for the central atom’s octet. NH 3 H H N H 4
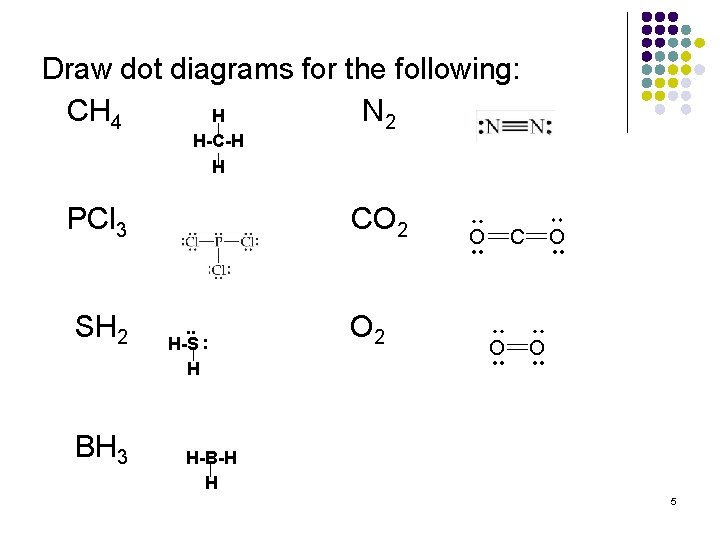
Draw dot diagrams for the following: CH 4 N 2 H H-C-H H PCl 3 SH 2 BH 3 CO 2 . . H-S : H O 2 H-B-H H 5
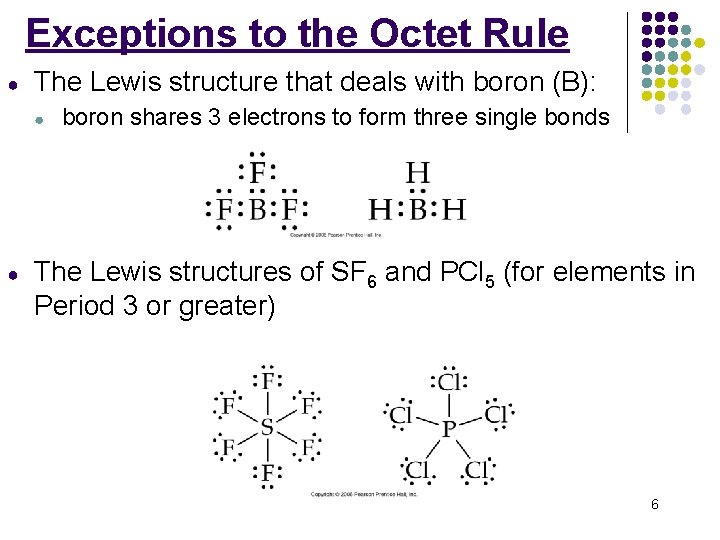
Exceptions to the Octet Rule ● The Lewis structure that deals with boron (B): ● ● boron shares 3 electrons to form three single bonds The Lewis structures of SF 6 and PCl 5 (for elements in Period 3 or greater) 6
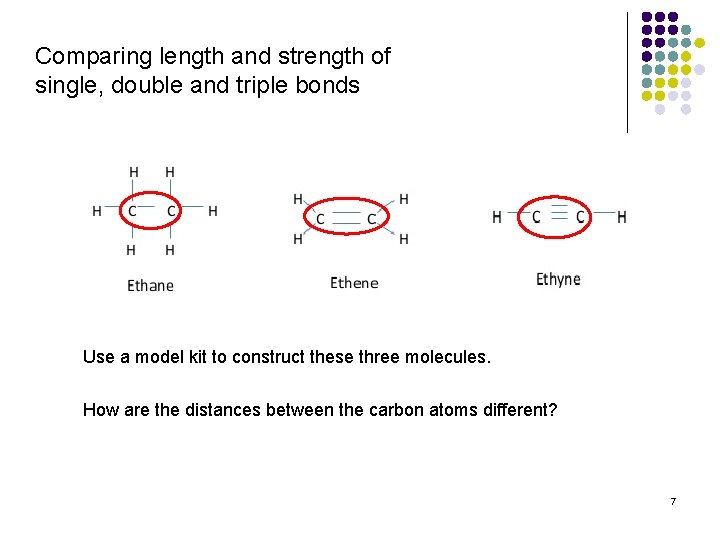
Comparing length and strength of single, double and triple bonds Use a model kit to construct these three molecules. How are the distances between the carbon atoms different? 7

Relative lengths: single bond ˃ double bond ˃ triple bond Relative strengths: triple bond ˃ double bond ˃ single bond 8

Naming Covalent Bonds ● ● Write names of both elements Add -ide to the last name Add prefixes at beginning of each word to indicate how many of each element. Exception- do not use mono in front of the first element N 2 O 5 Dinitrogen Pentaoxide 9

Covalent Bonding Prefixes 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. (mono-) ditritetrapentahexaheptaoctanonadeca 10

Practice Naming ● ● ● ● Name the following: ● CCl 4 ● PCl 3 ● SH 2 ● CO ● NO 3 ● N 2 O 4 Carbon tetrachloride Phosphorus trichloride Sulfur dihydride Carbon monoxide Nitrogen trioxide Dinitrogen tetraoxide 11

Molecular Elements ● ● Certain elements occur as 2 atom molecules Rule of 7’s ● ● there are 7 common diatomic elements find the element with atomic number 7, N make a figure 7 by going over to Group 7 A, then down don’t forget to include H 2 VIIA H 2 N 2 7 Common Covalent Compounds ● ● ● H 2 O Water NH 3 Ammonia CH 4 Methane O 2 F 2 Cl 2 Br 2 I 2 12

Acid Nomenclature

How to identify an acid: The formula of an acid starts with “H” HCl hydrochloric acid HNO 3 nitric acid H 3 PO 4 phosphoric acid

Two types of acids: 1. Binary Acids (Two elements, one of which is hydrogen) HCl HBr H 2 S 2. Oxyacids (contain oxygen, usually a polyatomic ion bonded to hydrogen) H 2 SO 4 HCl. O 3 HCl. O 4

If it is a binary acid: Prefix = Hydroroot = second element’s name Suffix = -ic acid HCl = hydro- chlor -ic acid→ hydrochloric acid HBr= hydro- brom -ic acid →hydrobromic acid H 2 S = hydro – sulfur – ic acid →hydrosulfuric acid

If it is an oxyacid 1. 2. 3. Prefix = none root = anion (polyatomic name) Suffix =depends! + acid If anion ends in –ate replace with –ic acid If anion ends in –ite replace with –ous acid Example: HNO₃ Example: HNO₂ Since NO₃⁻ = Nitrate Since NO₂⁻ = Nitrite HNO₃ = Nitric Acid HNO₂ = Nitrous acid *Sulfate/ite root goes to sulfur (sulfuric and sulfurous acid). Same is true for phosphate

Examples 1. 2. 3. 4. 5. 6. HNO 2 H 2 SO 3 H 3 PO 4 HNO 3 HCl H 2 SO 4

Examples- answers 1. 2. 3. 4. 5. 6. nitrous acid sulfurous acid phosphoric acid nitric acid hydrochloric acid sulfuric acid

Try on your own ● ● ● ● ● 1. HCl 2. HBr 3. H₂SO₄ 4. H₂S 5. H₂CO₃ 6. HC₂H₃O₂ 7. H₃PO₄ 8. H₃P 9. H₂Cr. O₄

Answers ● ● ● ● ● 1. HCl- hydrochloric acid 2. HBr- hydrobromic acid 3. H₂SO₄- sulfuric acid 4. H₂S- hydrosulfuric acid 5. H₂CO₃- carbonic acid 6. HC₂H₃O₂- acetic acid 7. H₃PO₄- phosphoric acid 8. H₃P- hydrophosphoric acid 9. H₂Cr. O₄- chromic acid

Try writing the formulas: 1. 2. 3. 4. 5. 6. 7. 8. 9. Sulfuric acid Nitric acid Hydrochloric acid Acetic acid Hydrofluoric acid Carbonic acid Nitrous acid Phosphoric acid Hydrosulfuric acid

Answers 1. 2. 3. 4. 5. 6. 7. 8. 9. H₂SO₄ HNO₃ HCl HC 2 H 3 O 2 HF H₂CO₃ HNO₂ H₃PO₄ H₂S
- Slides: 23