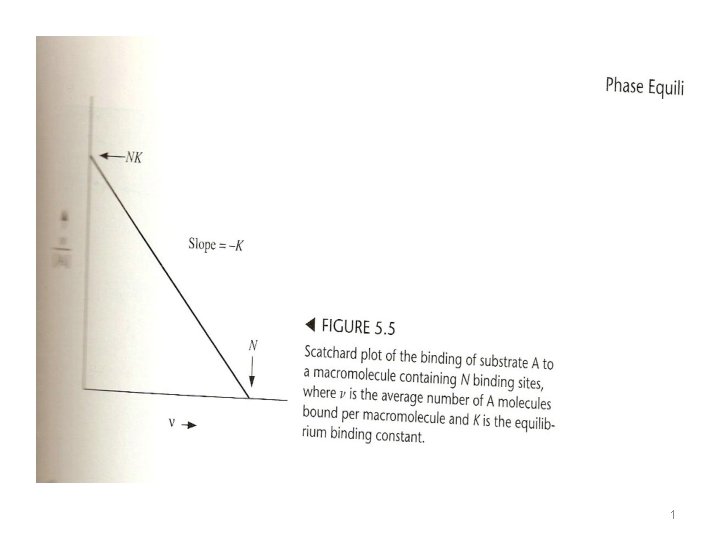
1 Cooperative binding and Anticooperative Binding Although the
1

Cooperative binding and Anticooperative Binding • Although the sites are identical, there is significant interaction among them. • The binding of a ligand to one site affects the binding of ligands to other sites. • The first ligand bound affects the binding of the next one, which affects the binding of the next one, and so on. • In cooperative binding, the first ligand bound makes it easier for the next one to be bound. (case: all- or- none binding) • In anticooperative binding, each succeeding ligand is bound less strongly than the previous one. 2

Donnan Effect and Donnan potential • We use equilibrium dialysis to measure binding of a small molecule ligand by a macromolecule. • The method becomes more complicated if the macromolecule and ligand are charged. • The solutions on each side of the dialysis membrane must be electrically neutral – means that there can be an apparent increase in binding of a ligand with the opposite charge to that of the macromolecule and a decrease in binding of a ligand with the same charge. • These effect depends on the net charge on the macromolecule. • When equilibrium (except for the macromolecule) is reached for charged species, a voltage is developed across the membrane. • The asymmetric distribution of ions caused by the charged macromolecules is called the Donnan potential 3

Membrane p 213 • Whenever there are two phases in contact, there is also a surface, or interface between them. • This surface has different properties and behavior than the phases. • The surface has thermodynamic properties specified by its free energy, enthalpy and so on, just as the bulk phases have. 4

Lipid molecules • Amphiphilic molecule consists of two parts, one of which interacts primarily with the hydrophobic phase and the other with the aqueous phase. • Surfactant, molecules that have a hydrocarbon tail and a polar head; (surface active molecules) • Fig. 5. 14 There is a large variety of hydrophilic head groups: 1 - Some are polar owing to exposed –OH or –NH 2 2 - Others are negatively charged 3 - Or zwitterionic, bearing both positive and negative charges at PH 7 There is variability in the number of hydrocarbon tails (usually one or two) attached to a head group→ variations are important for defining the properties of the surfaces, especially in membranes and vesicles. They determine the : 1 - Thermal stability, 2 - Fluidity 3 - Curvature of the membrane 4 - The interactions with proteins and other molecules associated with or embedded in the membrane} Lipid bilayers p 214 • • • 5

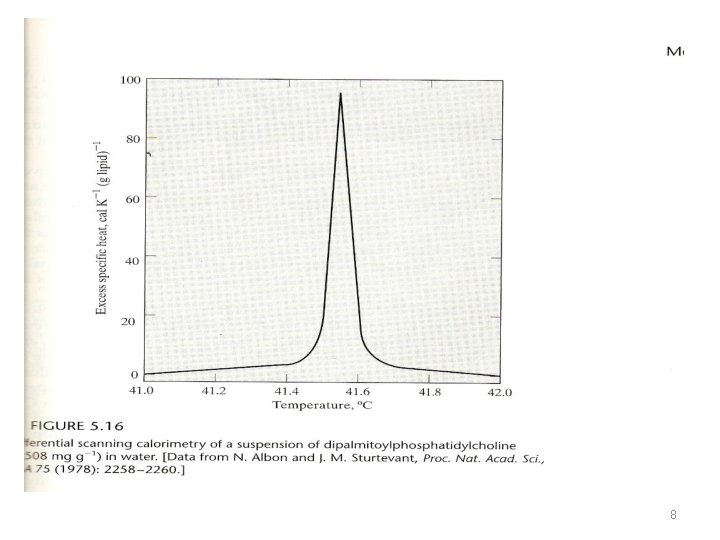
Phase Transitions in Lipids, Bilayers, and Membranes The lipid bilayers are thought to have a fluidity something like that of olive oil. The behavior is liquidlike in two dimensions. As the temperature is decreased, transitions to a solidlike membrane may occur. Under these conditions, the two-dimensional mobility is greatly decreased. Liquid-phase transitions can be studied using technique called differential thermal analysis {DTA}, using differential scanning calorimeter (DSC). 6

Phase Transitions in Lipids, Bilayers, and Membranes • Heat is added to the sample electrically at a constant rate, and the temperature is continuously monitored using a small probe. • When the temperature reaches the “melting point” of the lipid, extra heat is required to induce the transition from solidlike to liquidlike behavior. • Once the transition is completed, the temperature once again rises smoothly in proportion to heat capacity • Fig. 5. 16 • The peak of the curve at 41. 55 o. C indicates the transition temperature, Ttrans, for the lipid. 7
8

• Factors that influence the transition temperatures for lipids include: 1 - the length of the hydrocarbon tail (increased for longer hydrocarbon tails) 2 - the nature of the head group 3 - the presence of unsaturation (double bonds) in the hydrocarbons (decreased) 4 - additional molecules like cholesterol that incorporated into bilayer (increased) Transition temperatures reflecting the hydrocarbon fluidity that is important for biological function (in the -30 o to 100 o. C temperature range) 9

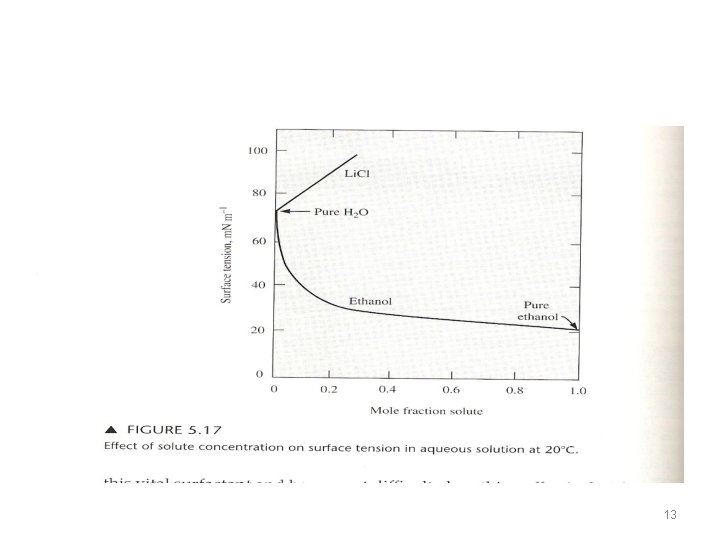
Surface Tension • Molecules at the surface (exposed to air) are attracted inward. • This creates a force in the surface that tends to minimize the surface area. • If the surface is stretched, the free energy of the system is increased. • The surface tension : =The free energy per unit surface area (J/m 2) = force per unit length (N/m) 10

Surface Tension • When a substance is dissolved in the liquid or added to the surface, the surface tension either decrease or does not change very much. (it never increases greatly) • The size of the drop is directly proportional to the surface tension. • Substance that tends to raise the surface tension of a liquid raises the free energy of the surface; they concentrate less at the surface. • Substances that lower the surface tension also lower the free energy of the surface; they migrate to the surface. 11

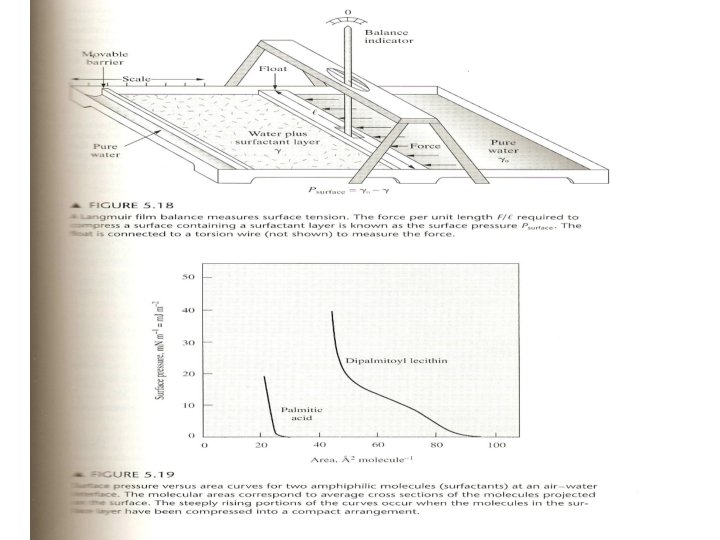
• The surface tension = (∂G/∂A)T, P , A=surface area Surfactant – Langmuir film balance fig. 5. 18 Fig. 5. 19 Psurface(m. Nm-1) =γo – γ , γo is the surface tension of pure water γ is the surface tension of coated water As the film is compressed, the surface pressure rises. In some cases, discontinuities appear in the plots owing to phase transitions that occur in some surfactants. The high surface pressures at low area per molecule (high surface concentration) result from the low surface tension produced by the surface-active molecules. 12
13
14

Biological membrane p 225 • The composition and orientation of the molecules determine their function. • Lipid bilayer • The proteins may be • 1 -Intrinsic or integral proteins (in which they are incorporated directly into the membrane structure • 2 - extrinsic proteins (if they are attached to the membrane surface or interact strongly with it. • Hydrophobic and hydrophilic sites are located in different regions of the membrane proteins. 15

The variety of membrane composition results in a range of physical properties. • For example 1 - the mobility of the molecules in two dimensions in a membrane may be that of a typical liquid or that of a solid. (phase transition) 2 - The transition temperature or melting point is sensitive to the lipid composition. 3 - Many proteins are amphiphilic and interact with both the hydrophobic lipids and the polar aqueous interface. 16

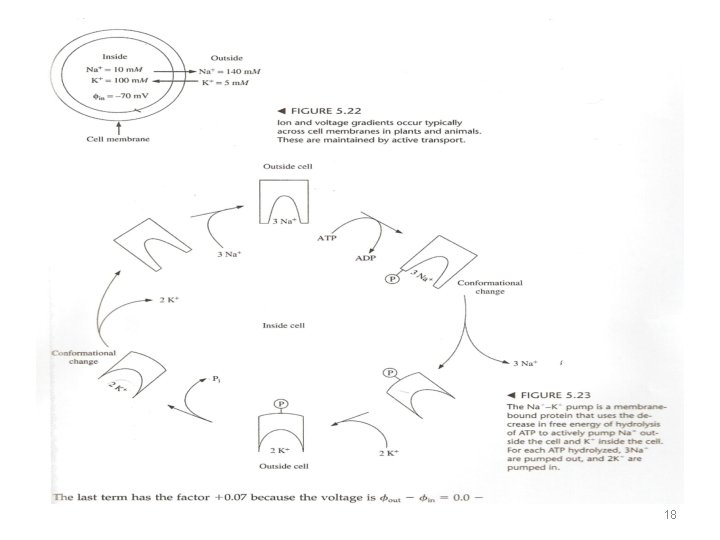
Active and Passive Transport • Passive flow (concentration-electric field) {in equilibrium dialysis flow of ligand to bind with the macromolecule} • Active transport (Na-K pump) • {the transport of a substance from a lower to higher chemical potential} • F=k. QQ/r 2 V=E. L V=KQ/r • Electric field lines - direction • E= KQ/r 2 17
18

• A protein complex called the sodium-potassium pump uses the free energy of hydrolysis of ATP to pump Na ions out of the cell and K ions into the cell. • The free energy of the net reaction must be negative. • This means that the positive free energy of the actively transporting ions against concentration and voltage gradient must be more than balanced by the negative free energy of ATP hydrolysis Δμ = RT ln a. Na+ (outside) + ZFV a. Na+ (inside) , R= gas constant =8. 3145 J/K mol T = temperature in Kelvin a. Na+ (outside)= concentration of sodium ions outside a. Na+ (inside)= concentration of sodium ions inside Z = charge on ion ± 1, ± 2, ± 3, . . F= faraday constant =9. 6485 x 104 C/mol V= Φ 2 - Φ 1 is the potential difference in volts between the two phases 19

20
- Slides: 20