1 Clinical Practice Guidelines Decompensated cirrhosis About these

1


Clinical Practice Guidelines Decompensated cirrhosis

About these slides • These slides give a comprehensive overview of the EASL clinical practice guidelines on the management of decompensated cirrhosis • The guidelines were first presented at the International Liver Congress 2018 and are published in the Journal of Hepatology – A full copy of the publication can be downloaded from the Clinical Practice Guidelines section of the EASL website • Please feel free to use, adapt, and share these slides for your own personal use; however, please acknowledge EASL as the source

About these slides • Definitions of all abbreviations shown in these slides are provided within the slide notes • When you see a home symbol like this one: , you can click on this to return to the outline or topics pages, depending on which section you are in These slides are intended for use as an educational resource and should not be used in isolation to make patient management decisions. All information included should be verified before treating patients or using any therapies described in these materials • Please send any feedback to: slidedeck_feedback@easloffice. eu

Guideline panel • Chair – Paolo Angeli • Panel – Càndid Villanueva, Claire Francoz, Rajeshwar P Mookerjee, Jonel Trebicka, Aleksander Krag, Wim Laleman, Pere Gines, Mauro Bernardi (EASL Governing Board Representative) • Reviewers – Alexander Gerbes, Thierry Gustot, Guadalupe Garcia-Tsao EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Outline Methods Background Guidelines • Grading evidence and recommendations • Definition and pathophysiology of decompensated cirrhosis • Key recommendations EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Methods Grading evidence and recommendations

Grading evidence and recommendations • Grading is adapted from the GRADE system 1 Grade of evidence I Randomized, controlled trials II-1 Controlled trials without randomization II-2 Cohort or case-control analytical studies II-3 Multiple time series, dramatic uncontrolled experiments III Opinions of respected authorities, descriptive epidemiology Grade of recommendation 1 Strong recommendation: Factors influencing the strength of the recommendation included the quality of the evidence, presumed patient-important outcomes, and cost 2 Weaker recommendation: Variability in preferences and values, or more uncertainty: more likely a weak recommendation is warranted Recommendation is made with less certainty: higher cost or resource consumption 1. Guyatt GH, et al. BMJ. 2008: 336: 924– 6; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Background Definition and pathophysiology of decompensated cirrhosis
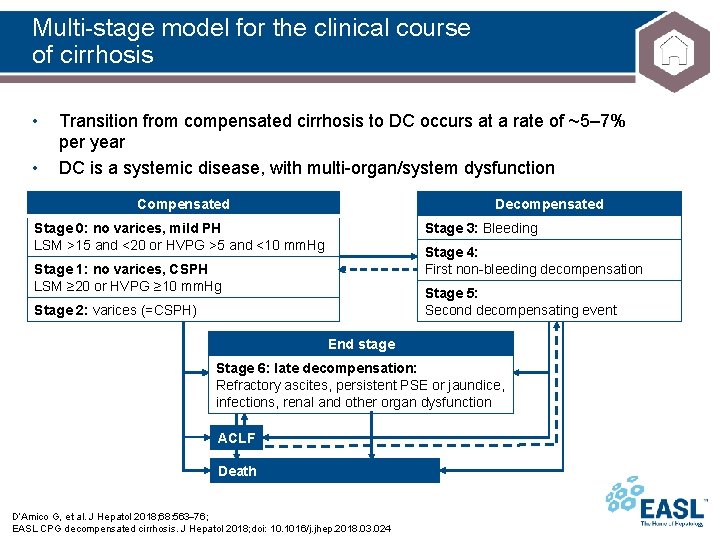
Multi-stage model for the clinical course of cirrhosis • • Transition from compensated cirrhosis to DC occurs at a rate of ~5– 7% per year DC is a systemic disease, with multi-organ/system dysfunction Decompensated Compensated Stage 3: Bleeding Stage 0: no varices, mild PH LSM >15 and <20 or HVPG >5 and <10 mm. Hg Stage 4: First non-bleeding decompensation Stage 1: no varices, CSPH LSM ≥ 20 or HVPG ≥ 10 mm. Hg Stage 5: Second decompensating event Stage 2: varices (=CSPH) End stage Stage 6: late decompensation: Refractory ascites, persistent PSE or jaundice, infections, renal and other organ dysfunction ACLF Death D’Amico G, et al. J Hepatol 2018; 68: 563 76; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
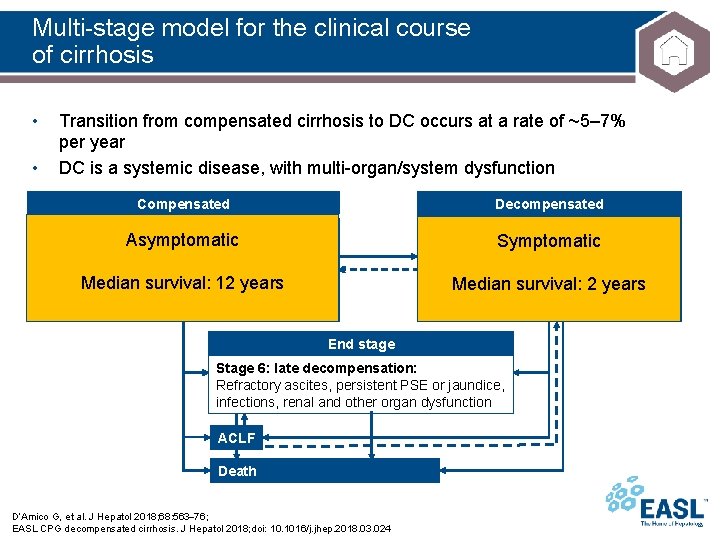
Multi-stage model for the clinical course of cirrhosis • • Transition from compensated cirrhosis to DC occurs at a rate of ~5– 7% per year DC is a systemic disease, with multi-organ/system dysfunction Decompensated Compensated Stage 3: Bleeding Stage 0: no varices, mild PH Asymptomatic LSM >15 and <20 or HVPG >5 and <10 mm. Hg Stage 1: no varices, CSPH Median LSM ≥ 20 or HVPGsurvival: ≥ 10 mm. Hg 12 Symptomatic Stage 4: First non-bleeding decompensation years Median survival: 2 years Stage 5: Second decompensating event Stage 2: varices (=CSPH) End stage Stage 6: late decompensation: Refractory ascites, persistent PSE or jaundice, infections, renal and other organ dysfunction ACLF Death D’Amico G, et al. J Hepatol 2018; 68: 563 76; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
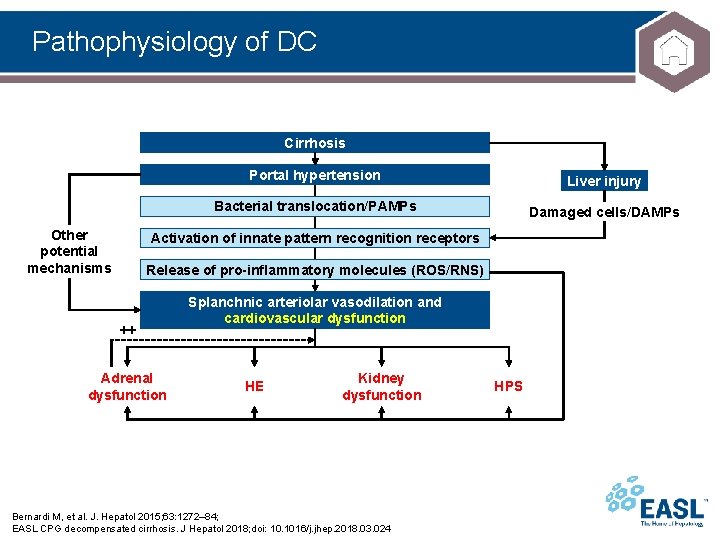
Pathophysiology of DC Cirrhosis Other potential mechanisms Portal hypertension Liver injury Bacterial translocation/PAMPs Damaged cells/DAMPs Activation of innate pattern recognition receptors Release of pro-inflammatory molecules (ROS/RNS) ++ Adrenal dysfunction Splanchnic arteriolar vasodilation and cardiovascular dysfunction HE Kidney dysfunction Bernardi M, et al. J. Hepatol 2015; 63: 1272– 84; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024 HPS
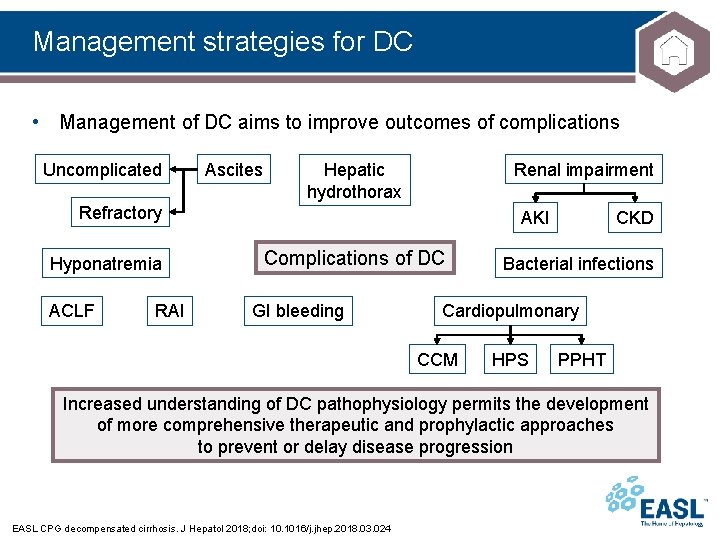
Management strategies for DC • Management of DC aims to improve outcomes of complications Uncomplicated Ascites Hepatic hydrothorax Renal impairment Refractory Hyponatremia ACLF RAI AKI Complications of DC GI bleeding CKD Bacterial infections Cardiopulmonary CCM HPS PPHT Increased understanding of DC pathophysiology permits the development of more comprehensive therapeutic and prophylactic approaches to prevent or delay disease progression EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Key recommendations

Topics 1. Overall management of DC – – Suppression of aetiological factor(s) Treatment of key pathogenic factors 2. Management of specific complications of DC – – – – – Ascites Refractory ascites Hepatic hydrothorax Hyponatremia Gastrointestinal bleeding Bacterial infections Renal impairment Acute-on-chronic liver failure Relative adrenal insufficiency Cardiopulmonary complications EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024 Click on a topic to skip to that section

Overall management of DC • Management should aim to prevent progression, not treat complications • No treatment exists that can act on cirrhosis progression directly • Two alternative approaches can be taken: – Suppress aetiological factor(s) that cause liver inflammation and cirrhosis development – Target key factors in the pathogenesis of cirrhosis decompensation and progression EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Suppression of aetiological factor(s) • Impact is variable • Probably depends on the status of liver disease at the time Recommendation Grade of evidence Grade of recommendation In patients with DC the aetiological factor should be removed, particularly alcohol consumption and hepatitis B or C virus infection, as this strategy is associated with decreased risk of decompensation and increased survival EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024 II-2 1

Treatment of key pathogenic factors • Several strategies have been evaluated to prevent disease progression in patients with DC – Targeting microbiome abnormalities and bacterial translocation to improve the gut–liver axis (i. e. rifaximin) – Improving the disturbed circulatory function (i. e. long-term albumin) – Treating the inflammatory state (i. e. statins) – Targeting portal hypertension (i. e. β-blockers) Further clinical research is needed to confirm the safety and potential benefits of these therapeutic approaches to prevent cirrhosis progression in patients with DC EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Ascites • Most common complication of decompensation in cirrhosis – Develops in 5– 10% of patients with compensated cirrhosis per year • Significant impact on patients – – – Impairs patient working and social life Frequently leads to hospitalization Requires chronic treatment Direct cause of further complications Poor prognosis (5 -year survival, ~30%) • Ascites can be uncomplicated or refractory – Ascites is uncomplicated when not infected, refractory or associated with impairment of renal function EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Uncomplicated ascites: evaluation and diagnosis • Cirrhosis is responsible for 80% of cases of ascites • Initial patient evaluation: – – History Physical examination Abdominal ultrasound Laboratory assessment • Liver and renal function, serum and urine electrolytes, analysis of ascitic fluid • Ascites is graded based on amount of fluid in the abdominal cavity Grading of ascites* Grade 1 Mild ascites: only detectable by ultrasound examination Grade 2 Moderate ascites: manifest by moderate symmetrical distension of abdomen Grade 3 Large or gross ascites: provokes marked abdominal distension *Ascites recurring on ≥ 3 occasions within a 12 -month period despite dietary sodium restriction and adequate diuretic dosage are considered recurrent EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Uncomplicated ascites: evaluation and diagnosis • Diagnostic paracentesis is indicated in: * – All patients with new-onset grade 2 or 3 ascites – Patients hospitalized for worsening ascites or any complication of cirrhosis Recommendation Grade of evidence Grade of recommendation Neutrophil count and culture of ascitic fluid culture† should be performed to exclude bacterial peritonitis • Neutrophil count >250 cells/µl denotes SBP II-2 1 Ascitic total protein concentration should be performed to identify patients at higher risk of developing SBP‡ II-2 1 The SAAG should be calculated when the cause of ascites is not immediately evident, and/or when conditions other than cirrhosis are suspected§ II-2 1 Cytology should be performed to differentiate malignancy-related from non-malignant ascites II-2 1 *Grade of evidence II-2, grade of recommendation 1; †Bedside inoculation blood culture bottles with 10 ml fluid each; ‡A total protein concentration <1. 5 g/dl is generally considered a risk factor for SBP; §SAAG ≥ 1. 1 g/dl indicates that portal hypertension is involved in ascites formation with an accuracy of about 97% EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Uncomplicated ascites: prognosis • Development of ascites in patients with cirrhosis is associated with a poor prognosis – 1 -year mortality: 40% – 2 -year mortality: 50% • Patients with ascites should be considered for referral for LT Recommendation Grade of evidence Grade of recommendation Since the development of grade 2 or 3 ascites in patients with cirrhosis is associated with reduced survival, LT should be considered as a potential treatment option II-2 1 • Patients may not receive adequate priority in transplant lists – Most commonly used prognostic scores can underestimate mortality risk • Improved methods to assess prognosis in these patients are needed EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
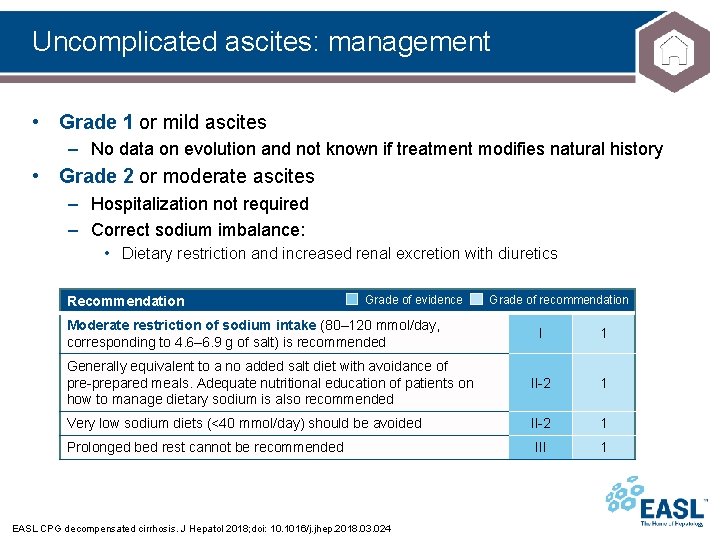
Uncomplicated ascites: management • Grade 1 or mild ascites – No data on evolution and not known if treatment modifies natural history • Grade 2 or moderate ascites – Hospitalization not required – Correct sodium imbalance: • Dietary restriction and increased renal excretion with diuretics Recommendation Grade of evidence Moderate restriction of sodium intake (80– 120 mmol/day, corresponding to 4. 6– 6. 9 g of salt) is recommended Grade of recommendation I 1 Generally equivalent to a no added salt diet with avoidance of pre-prepared meals. Adequate nutritional education of patients on how to manage dietary sodium is also recommended II-2 1 Very low sodium diets (<40 mmol/day) should be avoided II-2 1 III 1 Prolonged bed rest cannot be recommended EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Uncomplicated ascites: recommended diuretics • Mainstay of medical treatment are anti-mineralocorticoid drugs* • Loop diuretics may be added in patients with long-standing ascites Recommendation Grade of evidence Grade of recommendation First episode of grade 2 ascites • Anti-mineralocorticoid drug alone (from 100 mg/day with 100 mg stepwise increased every 72 hours to a maximum of 400 mg/day if no response to lower doses) I 1 In patients who do not respond to anti-mineralocorticoids† or who develop hyperkalaemia, furosemide should be added (from 40 mg/day with 40 mg stepwise increases to a maximum of 160 mg/day) I 1 Long-standing or recurrent ascites • Combination of an anti-mineralocorticoid drug and furosemide (dose increased sequentially according to response) I 1 Torasemide can be given in patients exhibiting a weak response to furosemide I 2 *Spironolactone, canrenone or K-canrenoate; †Body weight reduction <2 kg/week EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
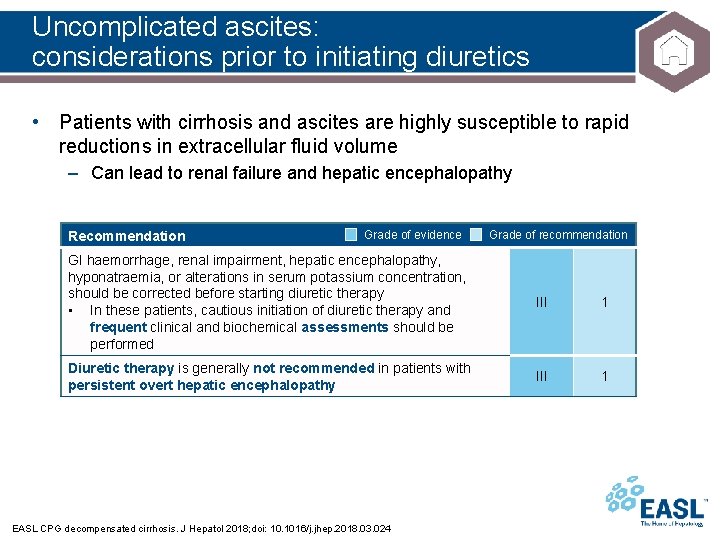
Uncomplicated ascites: considerations prior to initiating diuretics • Patients with cirrhosis and ascites are highly susceptible to rapid reductions in extracellular fluid volume – Can lead to renal failure and hepatic encephalopathy Recommendation Grade of evidence Grade of recommendation GI haemorrhage, renal impairment, hepatic encephalopathy, hyponatraemia, or alterations in serum potassium concentration, should be corrected before starting diuretic therapy • In these patients, cautious initiation of diuretic therapy and frequent clinical and biochemical assessments should be performed III 1 Diuretic therapy is generally not recommended in patients with persistent overt hepatic encephalopathy III 1 EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Uncomplicated ascites: monitoring of patients receiving diuretics • Loop diuretics can lead to potassium and magnesium depletion and hyponatraemia • Muscle cramps can impair quality of life in patients receiving diuretics Recommendation Grade of evidence Grade of recommendation Frequent clinical and biochemical monitoring during the first weeks of treatment (particularly on first presentation) I 1 II-2 1 Once ascites have largely resolved, the dose of diuretics should be reduced to the lowest effective dose III 1 Discontinue diuretics in case of severe hyponatraemia, * AKI, worsening hepatic encephalopathy, or incapacitating muscle cramps III 1 Discontinue furosemide for severe hypokalaemia (<3 mmol/L ) Discontinue anti-mineralocorticoids for hyperkalaemia (>6 mmol/L) III 1 Albumin infusion or baclofen administration† are recommended in patients with muscle cramps I 1 Recommended maximum weight loss: 0. 5 kg/day in patients without oedema, 1 kg/day in patients with oedema *Serum sodium <125 mmol/L; † 10 mg/day, with a weekly increase of 10 mg/day up to 30 mg/day EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Uncomplicated ascites: management of Grade 3 ascites • Grade 3 or large ascites – LVP, under strict sterile conditions, is the treatment of choice • Ascites should be completely removed in a single session* – Contraindications to LVP include: • Uncooperative patient, abdominal skin infection at puncture sites, pregnancy, severe coagulopathy, severe bowel distention Recommendation Grade of evidence Grade of recommendation LVP should be followed with plasma volume expansion I 1 Plasma volume expansion should be performed by albumin infusion (8 g/L ascites) • For >5 L of ascites: more effective than other plasma expanders • For <5 L of ascites (low risk of PPCD): treat with albumin due to concerns about use of alternative plasma expanders I 1 III 1 After LVP, patients should receive the minimum dose of diuretics necessary to prevent re-accumulation of ascites I 1 When needed, LVP should be performed in patients with AKI or SBP III 1 *Grade of evidence I, grade of recommendation 1 EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Uncomplicated ascites: contraindicated drugs • Patients with DC and ascites are at increased risk of renal impairment from several types of drug Recommendation Grade of evidence Grade of recommendation NSAIDs should not be used (high risk of developing further sodium retention, hyponatraemia, and AKI) II-2 1 Angiotensin-converting enyzme inhibitors, angiotensin II antagonists, or 1 -adrenergic receptor blockers should not generally be used (increased risk of renal impairment) II-2 1 Aminoglycosides are discouraged (increased risk of AKI) • Reserved for patients with severe bacterial infections that cannot be treated with other antibiotics II-2 1 II 2 III 1 Contrast media • In patients with preserved renal function: does not appear to be associated with increased risk of renal impairment • In patients with renal failure: insufficient data, cautious use and preventative measures recommended EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Refractory ascites: definition • International Ascites Club: – “Ascites that cannot be mobilized or the early recurrence of which (after LVP) cannot be satisfactorily prevented by medical therapy” Refractory ascites Diuretic resistant Diuretic intractable Ascites that cannot be mobilized or the early recurrence of which cannot be prevented because of a lack of response to sodium restriction and diuretic treatment Ascites that cannot be mobilized or the early recurrence of which cannot be prevented because of the development of diuretic-induced complications that preclude the use of an effective diuretic dosage EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Refractory ascites: diagnostic criteria Diagnostic criteria Treatment duration Patients must be on intensive diuretic therapy* for at least 1 week and on a salt-restricted diet of less than 90 mmol/day Lack of response Mean weight loss of <0. 8 kg over 4 days and urinary sodium output less than the sodium intake Early ascites recurrence Reappearance of grade 2 or 3 ascites within 4 weeks of initial mobilization • HE: development of encephalopathy in the absence of any other precipitating factor • Renal impairment: increase of serum creatinine by >100% to a value >2 mg/dl (177 µmol/L) in patients with ascites responding to Diuretic-induced treatment complications • Hyponatraemia: a decrease of serum sodium by >10 mmol/L to a serum sodium of <125 mmol/L • Hypo- or hyperkalaemia: a change in serum potassium to <3 mmol/L or >6 mmol/L despite appropriate measures • Incapacitating muscle cramps *Spironolactone 400 mg/day and furosemide 160 mg/day EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Refractory ascites: diagnosis • Refractory ascites is associated with a poor prognosis – Median survival around 6 months Recommendation Grade of evidence Grade of recommendation The diagnosis of refractory ascites relies on the assessment of the response of ascites to diuretic therapy and salt restriction • Evaluation should be done in stable patients without associated complications, such as bleeding or infection, after ascertaining patient compliance to treatment III 1 Patients with refractory ascites should be evaluated for LT III 1 EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Refractory ascites: management • LVP is a safe and effective treatment – Should be associated with albumin administration to prevent PPCD • Drug treatments are controversial or inadequately studied Recommendation Grade of evidence Grade of recommendation Repeated LVP plus albumin (8 g/L of ascites removed) are recommended as first-line treatment for refractory ascites I 1 Diuretics should be discontinued in patients with refractory ascites who do not excrete >30 mmol/day of sodium under diuretic treatment III 1 II-2 1 I 2 Although controversial data exist on the use of NSBBs in refractory ascites, caution should be exercised in severe cases* • High doses of NSBB should be avoided (i. e. propranolol >80 mg/day) • Carvedilol can not be recommended at present *See also section on gastrointestinal bleeding EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Refractory ascites: indications for TIPS • TIPS decompresses the portal system* – Short term: accentuates peripheral arterial vasodilation – Within 4– 6 weeks: improves effective volaemia and renal function to increase renal sodium excretion Recommendation Grade of evidence Grade of recommendation Patients should be evaluated for TIPS insertion when: • There is refractory or recurrent ascites • Paracentesis is ineffective I III 1 1 TIPS insertion is recommended in patients: • With recurrent ascites as it improves survival • With refractory ascites as it improves the control of ascites I I 1 1 II-2 III 1 1 The use of small-diameter PTFE-covered stents is recommended to reduce the risk of TIPS dysfunction and hepatic encephalopathy After TIPS insertion, continue the following until ascites resolution: • Diuretics and salt restriction • Close clinical follow-up *By shunting an intrahepatic portal branch into a hepatic vein EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Hepatic hydrothorax: definition and diagnosis • Definition – Accumulation of transudate in the pleural space • In the absence of cardiac, pulmonary or pleural disease – Ascites moves through small diaphragmatic defects • Negative intrathoracic pressure induced by inspiration – Can lead to respiratory failure – Can be complicated by spontaneous bacterial infections (empyema) – Associated with poor prognosis • Median survival: 8 12 months • Diagnosis – Once pleural effusion has been ascertained, cardiopulmonary and primary pleural diseases should be excluded* – Diagnostic thoracentesis is required to rule out bacterial infection* *Grade of evidence III, grade of recommendation 1 EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
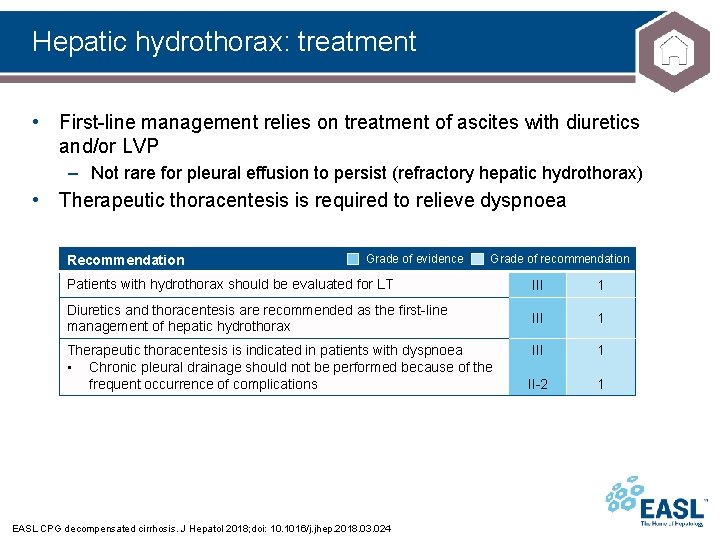
Hepatic hydrothorax: treatment • First-line management relies on treatment of ascites with diuretics and/or LVP – Not rare for pleural effusion to persist (refractory hepatic hydrothorax) • Therapeutic thoracentesis is required to relieve dyspnoea Recommendation Grade of evidence Grade of recommendation Patients with hydrothorax should be evaluated for LT III 1 Diuretics and thoracentesis are recommended as the first-line management of hepatic hydrothorax III 1 II-2 1 Therapeutic thoracentesis is indicated in patients with dyspnoea • Chronic pleural drainage should not be performed because of the frequent occurrence of complications EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
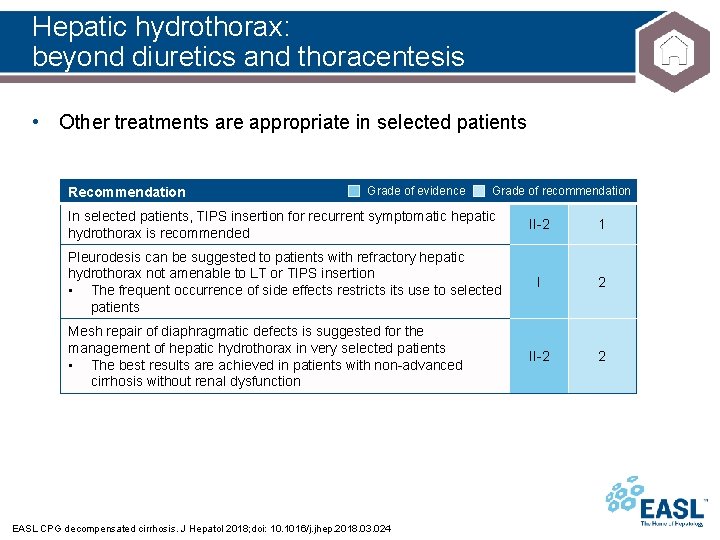
Hepatic hydrothorax: beyond diuretics and thoracentesis • Other treatments are appropriate in selected patients Recommendation Grade of evidence Grade of recommendation In selected patients, TIPS insertion for recurrent symptomatic hepatic hydrothorax is recommended II-2 1 Pleurodesis can be suggested to patients with refractory hepatic hydrothorax not amenable to LT or TIPS insertion • The frequent occurrence of side effects restricts its use to selected patients I 2 II-2 2 Mesh repair of diaphragmatic defects is suggested for the management of hepatic hydrothorax in very selected patients • The best results are achieved in patients with non-advanced cirrhosis without renal dysfunction EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Hyponatraemia • Common in patients with advanced cirrhosis – Arbitrarily defined as serum sodium concentration <130 mmol/L • Hypo- and hypervolaemic hyponatraemia can occur • Associated with: – Increased mortality and morbidity, particularly neurological complications – Reduced survival after LT Recommendation Grade of evidence Grade of recommendation Patients with cirrhosis who develop hyponatraemia should be evaluated for LT II-2 1 Removal of the cause and administration of normal saline are recommended in the management of hypovolaemic hyponatraemia III 1 Fluid restriction* to 1, 000 ml/day is recommended in the management of hypervolaemic hyponatraemia since it may prevent a further reduction in serum sodium levels III 1 *Beyond fluid restriction, hypertonic saline should be limited to rare patients with life-threatening complications. It can be considered in patients with severe hyponatraemia who are expected to undergo LT within days. Correction of serum sodium concentration after attenuation of symptoms should be slow (≤ 8 mmol/L per day) to avoid irreversible neurological sequelae (II-3; 1). Albumin can be administered but data are very limited (II-3; 2). Use of vaptans should be limited to clinical trials (III; 1) EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Variceal haemorrhage: pathophysiology and natural history • Occurs when variceal wall ruptures due to excessive wall tension – Portal pressure is a key factor in both rupture and severity of bleeding • 70% of GI bleeding events result from VH in patients with portal hypertension – Second most common decompensating event – Most severe and immediate life threatening complication Recommendation Grade of evidence Grade of recommendation Patients with DC are at high risk and should have an OGD to screen for varices, unless previously diagnosed and treated II-2 1 If OGD is performed, the presence, size and presence of red wale marks should be reported II-2 1 III 2 In patients without varices in whom aetiological factor persists and/or remain decompensated, screening OGD should be repeated yearly • In other patients the screening interval could be prolonged, but the exact interval is unclear and more data are required EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Variceal haemorrhage: prevention and treatment • High risk of death when VH occurs in patients with DC – Strategies to adequately treat VH and prevent (re)bleeding and death should be actively pursued Recommendation Grade of evidence Grade of recommendation Primary prophylaxis must be initiated upon detection of high-risk varices* because of increased risk of VH I 1 Patients with small varices with red wale marks or Child–Pugh C should be treated with NSBBs III 1 III 2 II-2 2 Patients with medium–large varices should be treated with either NSBBs or EBL • Choice of treatment can be based on local resources and expertise, patient preference, contraindications and AEs • NSBBs could be preferred because in addition to lowering portal pressure, they also exert other potential beneficial effects *High-risk = small varices with red signs, medium or large varices irrespective of Child–Pugh classification or small varices in Child–Pugh C patients EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Variceal haemorrhage: prophylaxis with NSBBs and EBL • NSBBs and EBL are equally effective in preventing first bleeding in patients with high-risk varices – Choice between options depends on factors such as patient preference, contraindications or adverse events Recommendation Grade of evidence Grade of recommendation Ascites is not a contraindication for NSBBs. However, caution should be exercised in cases of severe or refractory ascites • High doses of NSBBs should be avoided • The use of carvedilol can not be recommended at present NSBBs should be discontinued in patients with progressive hypotension or those who develop an acute intercurrent condition* After recovery, reinstatement of NSBBs can be attempted • When NSBB intolerance or contraindications persist, patient’s bleeding risk should be managed by expeditious EBL *Such as bleeding, sepsis, SBP or AKI EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024 I 1 II-2 I 1 2 III 1 III 2 1

Variceal haemorrhage: prevention of variceal rebleeding • NSBBs and EBL in combination reduces the risk of re-bleeding compared with monotherapy Recommendation Combination therapy of NSBBs + EBL is recommended I 1 Covered TIPS placement is recommended in patients who continue to be intolerant to NSBBs* III 1 *Provided that there are no absolute contraindications EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
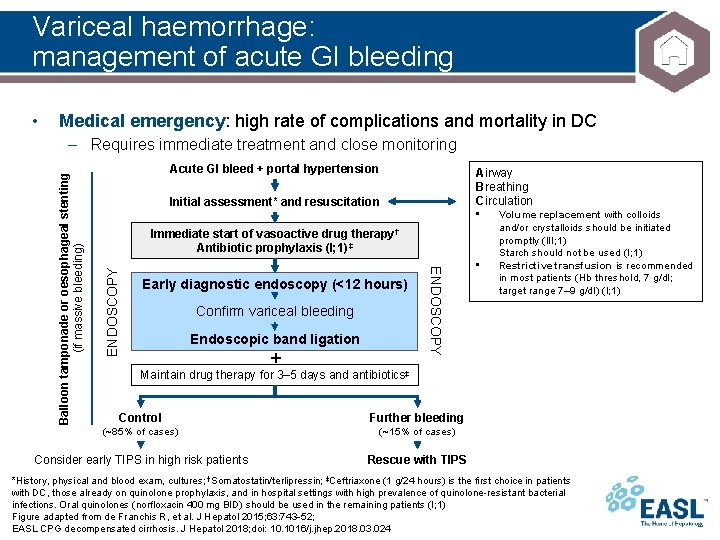
Variceal haemorrhage: management of acute GI bleeding • Medical emergency: high rate of complications and mortality in DC Acute GI bleed + portal hypertension Airway Breathing Circulation Initial assessment* and resuscitation • ENDOSCOPY Immediate start of vasoactive drug therapy† Antibiotic prophylaxis (I; 1) ‡ Early diagnostic endoscopy (<12 hours) Confirm variceal bleeding Endoscopic band ligation + ENDOSCOPY Balloon tamponade or oesophageal stenting (if massive bleeding) – Requires immediate treatment and close monitoring • Volume replacement with colloids and/or crystalloids should be initiated promptly (III; 1) Starch should not be used (I; 1) Restrictive transfusion is recommended in most patients (Hb threshold, 7 g/dl; target range 7– 9 g/dl) (I; 1) Maintain drug therapy for 3– 5 days and antibiotics‡ Control Further bleeding (~85% of cases) (~15% of cases) Consider early TIPS in high risk patients Rescue with TIPS *History, physical and blood exam, cultures; †Somatostatin/terlipressin; ‡Ceftriaxone (1 g/24 hours) is the first choice in patients with DC, those already on quinolone prophylaxis, and in hospital settings with high prevalence of quinolone-resistant bacterial infections. Oral quinolones (norfloxacin 400 mg BID) should be used in the remaining patients (I; 1) Figure adapted from de Franchis R, et al. J Hepatol 2015; 63: 743 52; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Acute variceal haemorrhage: treatment • Vasoactive drugs and ligation are the primary options for acute VH – There may be a role for TIPS in selected high-risk patients Recommendation Grade of evidence Grade of recommendation The combination of vasoactive drugs and ligation is recommended as the first therapeutic option in acute variceal bleeding I 1 Early pre-emptive covered TIPS (placed within 24– 72 hours) can be suggested in selected high-risk patients, such as those with Child–Pugh class C with score <14 • However, the criteria for high-risk patients, particularly Child–Pugh B with active bleeding, remains debatable and needs further study I 2 EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Management of persistent bleeding • Up to 10– 15% of patients have persistent bleeding or early re-bleeding – Despite treatment with vasoactive drugs and EBL, and prophylactic antibiotics Recommendation Grade of evidence Grade of recommendation TIPS should be used as the rescue therapy of choice in cases of persistent bleeding or early re-bleeding I 1 III 1 I 2 In the context of bleeding, where encephalopathy is commonly encountered, prophylactic lactulose may be used to prevent encephalopathy, but further studies are needed I 2 β-blockers and vasodilators should be avoided during the acute bleeding episode III 1 With the pre-requisite of expertise, balloon tamponade should be used in case of uncontrolled bleeding as a temporary ‘‘bridge” (max 24 hours) until definitive treatment can be instituted • Removable, covered and self-expanding oesophageal stents can be used as an alternative to balloon tamponade EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Bacterial infections • Risk of bacterial infection in patients with cirrhosis is caused by multiple factors – – – • Liver dysfunction Portosystemic shunting Gut dysbiosis Increased BT Cirrhosis-associated immune dysfunction Genetic factors Spontaneous bacterial peritonitis – Definition: bacterial infection of ascitic fluid without any intra-abdominal surgically treatable source of infection – Prevalence: all patients with cirrhosis and ascites are at risk • 1. 5– 3. 5% in outpatients; 10% in hospitalized patients – Prognosis: mortality exceeded 90% when first described • Reduced to ~20% with early diagnosis and treatment EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Spontaneous bacterial peritonitis: diagnosis • Diagnosis is based on diagnostic paracentesis • 50% of SBP episodes are present at hospital admission – Signs/symptoms of peritonitis: abdominal pain, tenderness, vomiting or diarrhoea, ileus – Signs of systemic inflammation: hyper- or hypothermia, chills, altered WBC count – Worsening liver function, HE, shock, renal impairment, GI bleeding However: SPB may be asymptomatic, particularly in outpatients EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Spontaneous bacterial peritonitis: diagnosis • Diagnosis is based on diagnostic paracentesis • 50% of SBP episodes are present at hospital admission – Signs/symptoms of peritonitis: abdominal pain, tenderness, vomiting or diarrhoea, ileus – Signs of systemic inflammation: hyper- or hypothermia, chills, altered WBC count – Worsening liver function, HE, shock, renal failure, GI bleeding Recommendation Grade of evidence Grade of recommendation Diagnostic paracentesis should be carried out in: • Patients with cirrhosis and ascites, at admission, to rule out SBP • Patients with GI bleeding, shock, fever or other signs of systemic inflammation, worsening liver and/or renal function, and HE II-2 1 SBP diagnosed by a neutrophil count in ascitic fluid >250/mm 3 • Neutrophil count is determined by microscopy or flow cytometry • No clear evidence to support routine use of reagent strips II-2 1 Ascitic fluid culture positivity is not a prerequisite for SBP diagnosis* II-2 1 *Culture should be performed to guide antibiotic therapy (Grade of evidence II-2, grade of recommendation 1) EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Management of SBP: empirical antibiotics • • Empirical IV antibiotics should be started immediately following diagnosis* Several factors should guide empirical antibiotic use† – Environment (community acquired vs. nosocomial) – Local bacterial resistance profiles – Severity of infection Recommendation Grade of evidence Third-generation cephalosporins are recommended as first-line antibiotic treatment for community-acquired SBP in countries with low rates of antibiotic resistance • In countries with high rates of antibiotic resistance piperacillin/tazobactam or carbapenem should be considered Antibiotic resistance is more likely in healthcare-associated and nosocomial SBP • Piperacillin/tazobactam: in areas with low prevalence of MDR bacteria • Carbapenem: in areas with high prevalence of ESBL-producing Enterobacteriaceae • Carbapenem + glycopeptides, daptomycin linezolid in areas with high prevalence of gram-positive MDR bacteria *Grade of evidence II-2, grade of recommendation 1; †Grade of evidence I, grade of recommendation 1 EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024 Grade of recommendation I 1 II-2 1 I 1

Management of SBP: empirical antibiotics • Antibiotic therapy should be carefully controlled and monitored Recommendation Grade of evidence Grade of recommendation Severe infections by XDR bacteria may require antibiotics known to be highly nephrotoxic in patients with cirrhosis (e. g. vancomycin or aminoglycosides) • In these cases, patients’ plasma levels should be monitored in accordance with local policy thresholds III 1 De-escalation according to bacterial susceptibility based on positive cultures is recommended to minimize resistance selection pressure II-2 1 Antibiotic efficacy should be checked with a second paracentesis at 48 hours from starting treatment • Suspect failure of first-line antibiotic if worsening clinical signs and symptoms and/or increase, or no marked reduction in leucocyte count (at least 25%) in 48 hours II-2 1 III 1 II-2 2 The duration of treatment should be at least 5– 7 days Spontaneous bacterial empyema should be managed similarly to SBP EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
Management of SBP: use of albumin • In patients with SBP treated with a third generation intravenous cephalosporin antibiotic, albumin significantly decreased the incidence of type-1 hepatorenal syndrome and reduced mortality 1 Recommendation Grade of evidence The administration of albumin is recommended in patients with SBP • 1. 5 g/kg at diagnosis and • 1 g/kg on Day 3 1. Sort P, et al. N Engl J Med 1999; 341: 403– 9; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024 Grade of recommendation I 1

Management of SBP: primary prophylaxis • Patients with cirrhosis and low ascitic fluid protein concentration (<10 g/L) and/or high serum bilirubin levels are at high risk of developing a first episode of SBP 1 Recommendation Grade of evidence Grade of recommendation Primary prophylaxsis with norfloxacin (400 mg/day) is recommended in patients with: • Child–Pugh score ≥ 9 and serum bilirubin level ≥ 3 mg/dl, and • Either impaired renal function or hyponatraemia, and • Ascitic fluid protein lower than 15 g/L I 1 Norfloxacin prophylaxis should be stopped in patients with long-lasting improvement of their clinical condition and disappearance of ascites III 1 1. Wiest R, et al. Gut 2012; 61: 297– 310; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Management of SBP: secondary prophylaxis • In patients who survive an episode of SBP, the cumulative recurrence rate at 1 year is approximately 70%1 Recommendation Grade of evidence Grade of recommendation Prophylactic norfloxacin (400 mg/day, orally) is recommended in patients who recover from an episode of SBP I 1 At present, rifaximin cannot be recommended as an alternative to norfloxacin for secondary prophylaxis of SBP • At present, no recommendation can be given to guide prophylaxis of SBP among patients already on rifaximin for the prevention of recurrent HE I 2 Patients who recover from SBP have a poor long-term survival and should be considered for LT II-2 1 PPIs may increase the risk for the development of SBP, their use should be restricted to those with a clear indication II-2 1 1. Rimola A, et al. J Hepatol 2000; 32: 142– 53; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
Management of other infections • Non-SBP infections are frequent in patients with cirrhosis – Associated with increased mortality Recommendation Grade of evidence Grade of recommendation Hospitalized patients with cirrhosis should be monitored closely for the presence of infections to enable early diagnosis and treatment II-1 1 Empirical antibiotic therapy should be commenced promptly II-1 1 Empirical antibiotic therapy should be based on: environment, local resistance profiles, severity and type of infection I 1 In the context of high bacterial resistance to antibiotics, carbapenem alone or in combination with other antibiotics should be preferred* I 1 Severe infections by XDR bacteria may require antibiotics known to be highly nephrotoxic in patients with cirrhosis (e. g. vancomycin or aminoglycosides) • In these cases, patients’ plasma level should be monitored in accordance with local policy thresholds III 1 Routine use of albumin not recommended in infections other than SBP I 1 *Carbapenem alone or in combination with other antibiotics proved to be superior to third-generation cephalosporins in healthcare-associated infections other than SBP EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
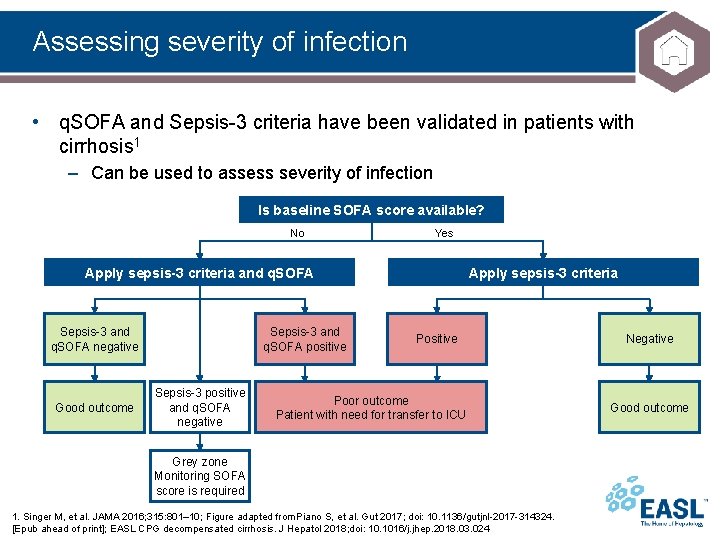
Assessing severity of infection • q. SOFA and Sepsis-3 criteria have been validated in patients with cirrhosis 1 – Can be used to assess severity of infection Is baseline SOFA score available? No Yes Apply sepsis-3 criteria and q. SOFA Sepsis-3 and q. SOFA negative Good outcome Sepsis-3 and q. SOFA positive Sepsis-3 positive and q. SOFA negative Apply sepsis-3 criteria Positive Poor outcome Patient with need for transfer to ICU Grey zone Monitoring SOFA score is required 1. Singer M, et al. JAMA 2016; 315: 801– 10; Figure adapted from Piano S, et al. Gut 2017; doi: 10. 1136/gutjnl-2017 -314324. [Epub ahead of print]; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024 Negative Good outcome
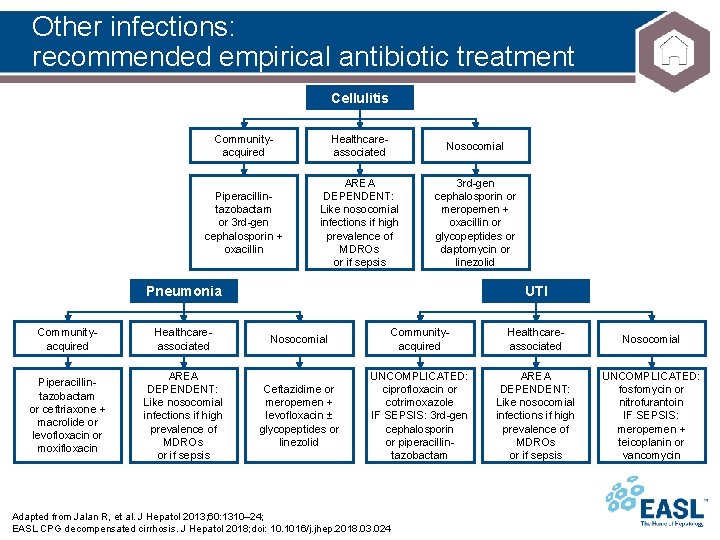
Other infections: recommended empirical antibiotic treatment Cellulitis Communityacquired Healthcareassociated Nosocomial Piperacillintazobactam or 3 rd-gen cephalosporin + oxacillin AREA DEPENDENT: Like nosocomial infections if high prevalence of MDROs or if sepsis 3 rd-gen cephalosporin or meropemen + oxacillin or glycopeptides or daptomycin or linezolid Pneumonia Communityacquired Healthcareassociated Piperacillintazobactam or ceftriaxone + macrolide or levofloxacin or moxifloxacin AREA DEPENDENT: Like nosocomial infections if high prevalence of MDROs or if sepsis UTI Nosocomial Communityacquired Healthcareassociated Nosocomial Ceftazidime or meropemen + levofloxacin ± glycopeptides or linezolid UNCOMPLICATED: ciprofloxacin or cotrimoxazole IF SEPSIS: 3 rd-gen cephalosporin or piperacillintazobactam AREA DEPENDENT: Like nosocomial infections if high prevalence of MDROs or if sepsis UNCOMPLICATED: fosfomycin or nitrofurantoin IF SEPSIS: meropemen + teicoplanin or vancomycin Adapted from Jalan R, et al. J Hepatol 2013; 60: 1310– 24; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
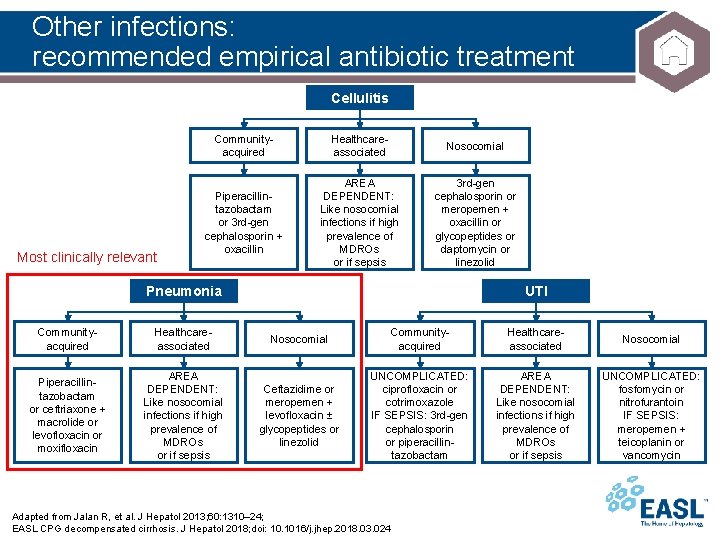
Other infections: recommended empirical antibiotic treatment Cellulitis Most clinically relevant Communityacquired Healthcareassociated Nosocomial Piperacillintazobactam or 3 rd-gen cephalosporin + oxacillin AREA DEPENDENT: Like nosocomial infections if high prevalence of MDROs or if sepsis 3 rd-gen cephalosporin or meropemen + oxacillin or glycopeptides or daptomycin or linezolid Pneumonia Communityacquired Healthcareassociated Piperacillintazobactam or ceftriaxone + macrolide or levofloxacin or moxifloxacin AREA DEPENDENT: Like nosocomial infections if high prevalence of MDROs or if sepsis UTI Nosocomial Communityacquired Healthcareassociated Nosocomial Ceftazidime or meropemen + levofloxacin ± glycopeptides or linezolid UNCOMPLICATED: ciprofloxacin or cotrimoxazole IF SEPSIS: 3 rd-gen cephalosporin or piperacillintazobactam AREA DEPENDENT: Like nosocomial infections if high prevalence of MDROs or if sepsis UNCOMPLICATED: fosfomycin or nitrofurantoin IF SEPSIS: meropemen + teicoplanin or vancomycin Adapted from Jalan R, et al. J Hepatol 2013; 60: 1310– 24; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
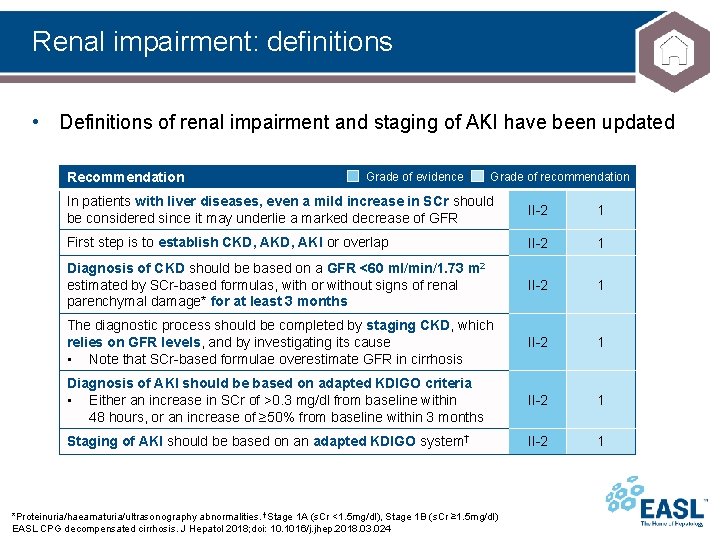
Renal impairment: definitions • Definitions of renal impairment and staging of AKI have been updated Recommendation Grade of evidence Grade of recommendation In patients with liver diseases, even a mild increase in SCr should be considered since it may underlie a marked decrease of GFR II-2 1 First step is to establish CKD, AKI or overlap II-2 1 Diagnosis of CKD should be based on a GFR <60 ml/min/1. 73 m 2 estimated by SCr-based formulas, with or without signs of renal parenchymal damage* for at least 3 months II-2 1 The diagnostic process should be completed by staging CKD, which relies on GFR levels, and by investigating its cause • Note that SCr-based formulae overestimate GFR in cirrhosis II-2 1 Diagnosis of AKI should be based on adapted KDIGO criteria • Either an increase in SCr of >0. 3 mg/dl from baseline within 48 hours, or an increase of ≥ 50% from baseline within 3 months II-2 1 Staging of AKI should be based on an adapted KDIGO system† II-2 1 *Proteinuria/haeamaturia/ultrasonography abnormalities. †Stage 1 A (s. Cr <1. 5 mg/dl), Stage 1 B (s. Cr ≥ 1. 5 mg/dl) EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
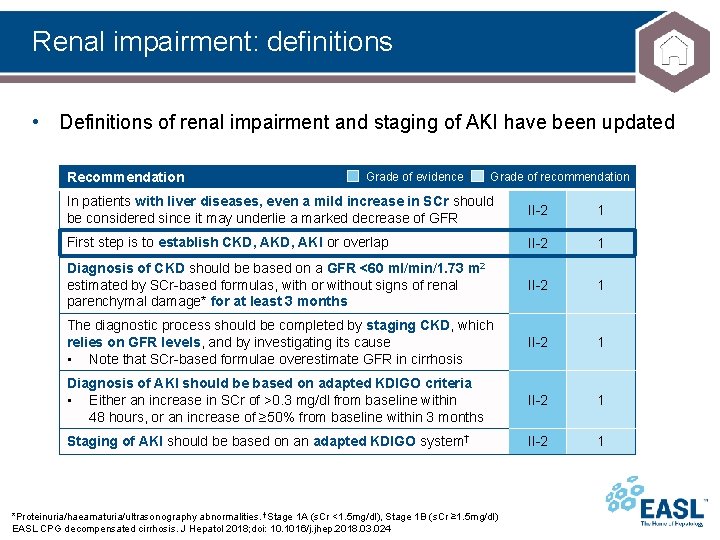
Renal impairment: definitions • Definitions of renal impairment and staging of AKI have been updated Recommendation Grade of evidence Grade of recommendation In patients with liver diseases, even a mild increase in SCr should be considered since it may underlie a marked decrease of GFR II-2 1 First step is to establish CKD, AKI or overlap II-2 1 Diagnosis of CKD should be based on a GFR <60 ml/min/1. 73 m 2 estimated by SCr-based formulas, with or without signs of renal parenchymal damage* for at least 3 months II-2 1 The diagnostic process should be completed by staging CKD, which relies on GFR levels, and by investigating its cause • Note that SCr-based formulae overestimate GFR in cirrhosis II-2 1 Diagnosis of AKI should be based on adapted KDIGO criteria • Either an increase in SCr of >0. 3 mg/dl from baseline within 48 hours, or an increase of ≥ 50% from baseline within 3 months II-2 1 Staging of AKI should be based on an adapted KDIGO system† II-2 1 *Proteinuria/haeamaturia/ultrasonography abnormalities. †Stage 1 A (s. Cr <1. 5 mg/dl), Stage 1 B (s. Cr ≥ 1. 5 mg/dl) EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
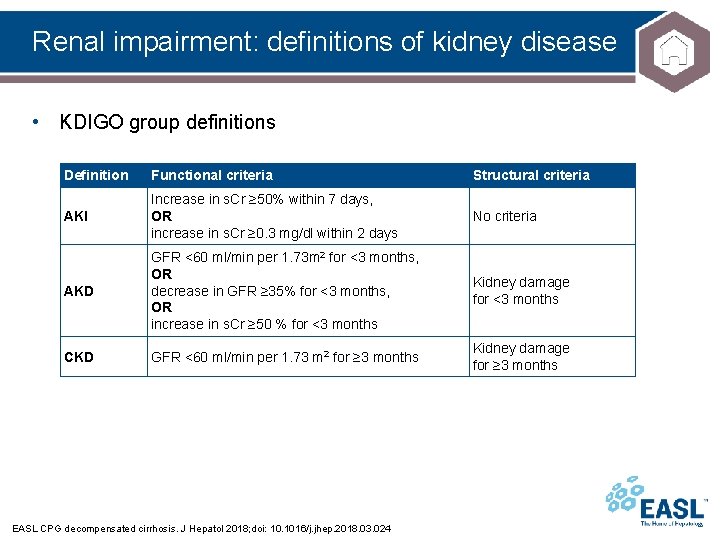
Renal impairment: definitions of kidney disease • KDIGO group definitions Definition Functional criteria Structural criteria AKI Increase in s. Cr ≥ 50% within 7 days, OR increase in s. Cr ≥ 0. 3 mg/dl within 2 days No criteria AKD GFR <60 ml/min per 1. 73 m 2 for <3 months, OR decrease in GFR ≥ 35% for <3 months, OR increase in s. Cr ≥ 50 % for <3 months Kidney damage for <3 months CKD GFR <60 ml/min per 1. 73 m 2 for ≥ 3 months Kidney damage for ≥ 3 months EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
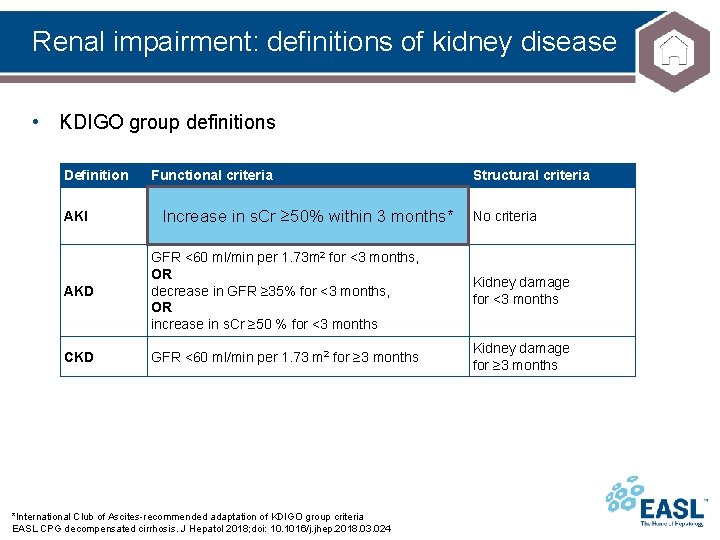
Renal impairment: definitions of kidney disease • KDIGO group definitions Definition Functional criteria Structural criteria AKI Increase in s. Cr ≥ 50% within 7 days, OR Increase in s. Cr ≥ 50% within 3 months* increase in s. Cr ≥ 0. 3 mg/dl within 2 days No criteria AKD GFR <60 ml/min per 1. 73 m 2 for <3 months, OR decrease in GFR ≥ 35% for <3 months, OR increase in s. Cr ≥ 50 % for <3 months Kidney damage for <3 months CKD GFR <60 ml/min per 1. 73 m 2 for ≥ 3 months Kidney damage for ≥ 3 months *International Club of Ascites-recommended adaptation of KDIGO group criteria EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

AKI in patients with cirrhosis: ICA definitions Subject Definition • s. Cr obtained within 3 months prior to admission • If >1 value within the previous 3 months, the value closest to the admission Baseline s. Cr • If no previous s. Cr, the s. Cr on admission should be used Definition of • Increase in s. Cr ≥ 0. 3 mg/dl (≥ 26. 5 µmol/L) within 48 hours or AKI • Increase s. Cr ≥ 50% within the prior 7 days Staging of AKI • Stage 1: increase in s. Cr ≥ 0. 3 mg/dl (≥ 26. 5 µmol/L) or an increase in s. Cr ≥ 1. 5 -fold to 2 -fold from baseline • Stage 2: increase in s. Cr >2 -fold to 3 -fold from baseline • Stage 3: increase of s. Cr >3 -fold from baseline or s. Cr ≥ 4. 0 mg/dl (353. 6 µmol/L) with acute increase ≥ 0. 3 mg/dl (≥ 26. 5 µmol/L) or initiation of renal replacement therapy Progression of AKI to a higher stage and/or of AKI need for RRT Regression of AKI to a lower stage No response Partial response Response to No regression of Regression of AKI stage with a treatment AKI reduction of s. Cr to ≥ 0. 3 mg/dl (≥ 26. 5 µmol/L) above baseline Angeli P, et al. J. Hepatol 2015; 62: 968 74; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024 Full response Return of s. Cr to a value within 0. 3 mg/dl (≥ 26. 5 µmol/L) of baseline

AKI in patients with cirrhosis: ICA definitions Subject Definition • s. Cr obtained within 3 months prior to admission • If >1 value within the previous 3 months, the value closest to the admission Baseline s. Cr • If no previous s. Cr, the s. Cr on admission should be used Definition of • Increase in s. Cr ≥ 0. 3 mg/dl (≥ 26. 5 µmol/L) within 48 hours or AKI • Increase s. Cr ≥ 50% within the prior 7 days Staging of AKI (s. Crµmol/L) <1. 5 mg/dl)* • Stage 1: increase in s. Cr ≥ 0. 3 Stage mg/dl 1 A (≥ 26. 5 or an increase in s. Cr ≥ 1. 5 -fold to 2 Stage 1 B (s. Cr ≥ 1. 5 mg/dl)* fold from baseline • Stage 2: increase in s. Cr >2 -fold to 3 -fold from baseline • Stage 3: increase of s. Cr >3 -fold from baseline or s. Cr ≥ 4. 0 mg/dl (353. 6 µmol/L) with acute increase ≥ 0. 3 mg/dl (≥ 26. 5 µmol/L) or initiation of renal replacement therapy Progression of AKI to a higher stage and/or of AKI need for RRT Regression of AKI to a lower stage No response Partial response Response to No regression of Regression of AKI stage with a treatment AKI reduction of s. Cr to ≥ 0. 3 mg/dl (≥ 26. 5 µmol/L) above baseline *ICA recommended adaptations of KDIGO group criteria Angeli P, et al. J. Hepatol 2015; 62: 968 74; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024 Full response Return of s. Cr to a value within 0. 3 mg/dl (≥ 26. 5 µmol/L) of baseline
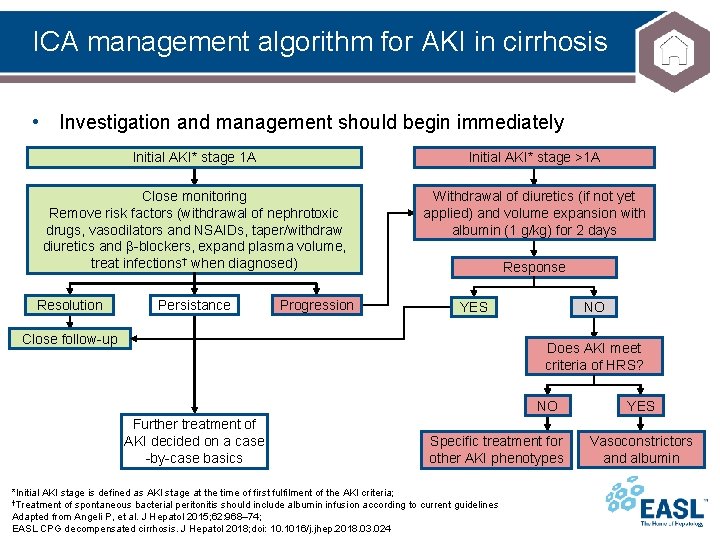
ICA management algorithm for AKI in cirrhosis • Investigation and management should begin immediately Initial AKI* stage 1 A Initial AKI* stage >1 A Close monitoring Remove risk factors (withdrawal of nephrotoxic drugs, vasodilators and NSAIDs, taper/withdraw diuretics and β-blockers, expand plasma volume, treat infections† when diagnosed) Withdrawal of diuretics (if not yet applied) and volume expansion with albumin (1 g/kg) for 2 days Resolution Persistance Progression Response YES Close follow-up NO Does AKI meet criteria of HRS? NO Further treatment of AKI decided on a case -by-case basics Specific treatment for other AKI phenotypes *Initial AKI stage is defined as AKI stage at the time of first fulfilment of the AKI criteria; †Treatment of spontaneous bacterial peritonitis should include albumin infusion according to current guidelines Adapted from Angeli P, et al. J Hepatol 2015; 62: 968– 74; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024 YES Vasoconstrictors and albumin

Differentiating types of AKI • All types of AKI can occur in patients with cirrhosis – Pre-renal, HRS, intrinsic, particularly ATN, and post-renal • • Key point is to differentiate HRS-AKI from ATN Classification of HRS was recently revised by the ICA 1 – Type 1 HRS now corresponds to HRS-AKI – Type 2 HRS includes renal impairment that fulfills the criteria of HRS but not of AKI (non-AKI-HRS or NAKI) Recommendation Grade of evidence Grade of recommendation It is important to differentiate among types of AKI in patients with cirrhosis II-2 1 The diagnosis of HRS-AKI is based on revised ICA criteria (see next slide) • As kidney biopsy is rarely performed in the setting of AKI, biomarkers should be implemented • In clinical practice among the different biomarkers to date, urinary NGAL can be used to distinguish between ATN and HRS II-2 2 1. Angeli et al. J. Hepatol 2015; 62: 968– 74; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

ICA diagnostic criteria for HRS-AKI • • • Cirrhosis and ascites Diagnosis of AKI according to ICA-AKI criteria No response after 2 consecutive days of diuretic withdrawal and plasma volume expansion with albumin 1 g per kg of body weight Absence of shock No current or recent use of nephrotoxic drugs (NSAIDs, aminoglycosides, iodinated contrast media, etc. ) No macroscopic signs of structural kidney injury, * defined as: – Absence of proteinuria (>500 mg/day) – Absence of microhaematuria (>50 RBCs per high power field) – Normal findings on renal ultrasonography *Patients who fulfil these criteria may still have structural damage such as tubular damage Angeli et al. J. Hepatol 2015; 62: 968 74; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Management of HRS-AKI: treatment • First-line therapy is terlipressin plus albumin* Recommendation Grade of evidence Grade of recommendation All patients meeting the current definition of HRS-AKI stage >1 A should be expeditiously treated with vasoconstrictors and albumin III 1 Terlipressin can be administered by IV boluses (1 mg every 4– 6 hours) or by continuous IV infusion (2 mg/day)† • In case of non-response (decrease in SCr <25% from the peak value) after 2 days, the dose of terlipressin should be increased in a stepwise manner to a maximum of 12 mg/day I 1 Albumin solution (20%) should be used at 20– 40 g/day • Serial measures assessing central blood volume can help to titrate the dose of albumin to prevent circulatory overload II-2 1 I I I 2 1 1 Noradrenaline can be an alternative to terlipressin‡ • Requires a central venous line often in an ICU Midodrine + octreotide can be an option when terlipressin or noradrenaline are unavailable (but efficacy is much lower) *Grade of evidence I, grade of recommendation 1; †Continuous IV infusion allows for dose reduction to reduced adverse effects; ‡ Limited data are available EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Management of HRS-AKI: screening and monitoring • Patients receiving treatment for HRS-AKI should be monitored for AEs and treatment response – AEs related to terlipressin or noradrenaline include ischaemic and cardiovascular events Recommendation Grade of evidence Grade of recommendation Careful clinical screening including ECG before starting the treatment is recommended • The decision to treat on a regular ward or transfer to higher dependency care should be case based Close monitoring of patients for the duration of treatment is important • Treatment should be modified or discontinued according to the type and severity of side effects I 1 Response to treatment: • CR: final SCr within 0. 3 mg/dl (26. 5 µmol/L) from baseline • PR: regression of AKI stage to a final SCr ≥ 0. 3 mg/dl (26. 5 µmol/L) from baseline III 1 In case of recurrence a repeat course of therapy should be given I 1 *Continuous IV infusion allows for dose reduction to reduced adverse effects EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Management of HRS: TIPS and LT • In most patients with HRS-AKI TIPS is contraindicated because of severe degree of liver failure Recommendation Grade of evidence Grade of recommendation There is insufficient data to advocate TIPS in HRS-AKI • It could be suggested in selected patients with HRS-NAKI II-2 2 LT is the best therapeutic option for patients with HRS regardless of the response to drug therapy I 1 The decision to initiate RRT should be based on the individual severity of illness I 2 II-2 1 The indication for liver-kidney transplantation remains controversial • Should be considered in patients with significant CKD or sustained AKI including HRS-AKI with no response to drug therapy EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Prevention of HRS • Based on the use of albumin in patients who develop SBP and the prevention of SBP using norfloxacin Recommendation Grade of evidence Grade of recommendation Albumin (1. 5 g/kg at diagnosis and 1 g/kg on Day 3) should be given in patients with SBP to prevent AKI I 1 Norfloxacin (400 mg/day) should be given as prophylaxis of SBP to prevent HRS-AKI I 1 EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Acute-on-chronic liver failure • Frequent occurrence in cirrhotic patients – 30% of admitted patients and 25% of outpatients • Major cause of death in patients with cirrhosis (50% mortality rate) • Develops on a background of acute decompensation • Characterized by hepatic and extrahepatic organ failure, highly activated systemic inflammation and a high 28 -day mortality • Precipitating events vary between populations and may include: – – Bacterial infections (30 57% of cases) Active alcohol intake or alcohol binge Reactivation of HBV Superimposed HAV and HEV infection EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
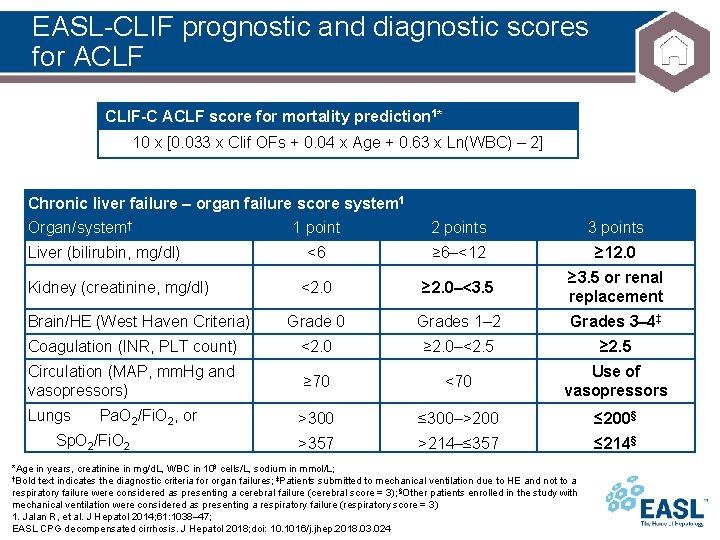
EASL-CLIF prognostic and diagnostic scores for ACLF CLIF-C ACLF score for mortality prediction 1* 10 x [0. 033 x Clif OFs + 0. 04 x Age + 0. 63 x Ln(WBC) – 2] Chronic liver failure – organ failure score system 1 Organ/system† 1 point 2 points 3 points <6 ≥ 6–<12 ≥ 12. 0 <2. 0 ≥ 2. 0–<3. 5 ≥ 3. 5 or renal replacement Grade 0 Grades 1– 2 Grades 3– 4‡ Coagulation (INR, PLT count) <2. 0 ≥ 2. 0–<2. 5 ≥ 2. 5 Circulation (MAP, mm. Hg and vasopressors) ≥ 70 <70 Use of vasopressors Lungs >300 ≤ 300–>200 ≤ 200§ >357 >214–≤ 357 ≤ 214§ Liver (bilirubin, mg/dl) Kidney (creatinine, mg/dl) Brain/HE (West Haven Criteria) Pa. O 2/Fi. O 2, or Sp. O 2/Fi. O 2 *Age in years, creatinine in mg/d. L, WBC in 106 cells/L, sodium in mmol/L; †Bold text indicates the diagnostic criteria for organ failures; ‡Patients submitted to mechanical ventilation due to HE and not to a respiratory failure were considered as presenting a cerebral failure (cerebral score = 3); §Other patients enrolled in the study with mechanical ventilation were considered as presenting a respiratory failure (respiratory score = 3) 1. Jalan R, et al. J Hepatol 2014; 61: 1038– 47; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Classification and grades of ACLF • Even mild renal or brain dysfunction in the presence of another organ failure, is associated with a significant short-term mortality and therefore defines the presence of ACLF Grades of ACLF Clinical characteristics NO ACLF No organ failure, or single non-kidney organ failure, creatinine <1. 5 mg/dl, no HE ACLF 1 a Single renal failure ACLF 1 b Single non-kidney organ failure, creatinine 1. 5– 1. 9 mg/dl and/or HE grade 1– 2 ACLF II Two organ failures ACLF III Three or more organ failures Moreau R, et al. Gastroenterology 2013; 144: 1426– 37; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Diagnosis of ACLF • Diagnosis of ACLF is based on organ failure in the presence of AD in patients with cirrhosis Recommendation Grade of evidence Grade of recommendation ACLF diagnosis: cirrhosis and AD* plus organ failure(s) involving high short-term mortality II-2 1 Diagnosis and grading should be based using the CLIF-C Organ Failure score II-2 1 Potential precipitating factor(s) should be investigated† • Hepatic: heavy alcohol intake, viral hepatitis, DILI, autoimmune hepatitis • Extrahepatic: infections haemodynamic derangements following haemorrhage, surgery II-2 1 *Defined as the acute development or worsening of ascites, overt encephalopathy, GI haemorrhage, non-obstructive jaundice and/or bacterial infections; †Note that in a significant proportion of patients, a precipitant factor may not be identified EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Management of ACLF • There is no specific therapy for ACLF • Treatment is based on organ support and management of complications Recommendation Grade of evidence Grade of recommendation Treatment of ACLF should be based on organ support, management of precipitants and associated complications III 1 Patients should be treated in intermediate care or intensive care settings III 1 ACLF is a dynamic condition and organ function should be monitored frequently and carefully throughout hospitalization • Particularly, liver, kidney, brain, lung, coagulation, and circulation • Monitoring and management should be individualized, mainly according to patients’ age and comorbidities III 1 EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Management of ACLF • • The cause of liver injury can be treated in certain situations, e. g. HBV Early action is crucial to patient survival – Treatment of precipitating factors – Referral for LT before evolution of ACLF makes LT impossible Recommendation Grade of evidence Grade of recommendation Early identification and treatment of precipitating factors of ACLF, particularly bacterial infections, is recommended. However, in some patients ACLF progresses despite treatment of precipitating factors III 1 Nucleoside analogues (tenofovir, entecavir) should be instituted as early as possible in patients with HBV-related ACLF I 1 Early referral of patients with ACLF to LT centres for immediate evaluation is recommended II-3 1 Withdrawal of intensive care support after 1 week can be suggested in patients who are not LT candidates and have ≥ 4 organ failures II-2 2 I 2 Administration of G-CSF cannot be recommended at present EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Relative adrenal insufficiency • Inadequate cortisol response to stress in the setting of critical illness* – Pathophysiology in cirrhosis is not well defined • • Diagnosis is influenced by the method used to measure cortisol It is not known whether cortisol supplementation in clinically stable cirrhosis with RAI is of any value Recommendation Grade of evidence Grade of recommendation Diagnosis of RAI • <248 nmol/L (9 lg/dl) change in total serum cortisol after 250 lg corticotropin injection, or • Random total cortisol of <276 nmol/L (<10 lg/dl) II-2 1 Salivary cortisol determination can be preferred • Serum free cortisol concentration can be influenced by reduced serum levels of CBG and albumin, frequently seen in patients with cirrhosis II-2 2 I 2 Hydrocortisone treatment (at a dose of 50 mg/6 hours) of RAI cannot be recommended *Also known as critical illness-related corticosteroid insufficiency (CIRCI) EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Cirrhotic cardiomyopathy • CCM occurs in patients with established cirrhosis characterized by: – Blunted contractile response to stress (pharmacological/surgery or inflammatory) – Altered diastolic left ventricular relaxation or/and increased left atrial volume – Electrophysiological abnormalities e. g. prolonged QTc – Cardiac output tending to decrease with decompensation – Systolic dysfunction: LVEF <55% • CCM is largely subclinical but its presence influences prognosis in advanced disease EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
Cirrhotic cardiomyopathy • Numerous electrocardiographic criteria, along with transmitral Doppler assessment, are used for the evaluation and diagnosis of diastolic dysfunction – However, there is the need for more controlled studies and correlation with clinical endpoints Recommendation Grade of evidence Grade of recommendation ECG in patients with cirrhosis should be performed with dynamic stress testing* (systolic dysfunction may be masked by hyperdynamic circulation and reduced afterload) • Lack of increased CO after physiological/pharmacological stress† indicates systolic dysfunction II-1 1 Myocardial strain imaging and assessment of GLS may be useful in the assessment of left ventricular systolic function in patients with DC II-2 2 III 2 II-1 1 Cardiac MRI may identify structural changes Diastolic dysfunction may occur as an early sign of CCM in the setting of normal systolic function, and should be diagnosed using ASE criteria: • Average E/e’>14 • Tricuspid velocity >2. 8 m/s • LAVI >34 ml/m 2 *Either pharmacologically, or through exercise; †And in the absence of influence of β-blockade EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Cirrhotic cardiomyopathy • Cardiac evaluation in patients with cirrhosis is important since CCM can influence prognosis Recommendation Grade of evidence Grade of recommendation In patients with AD, reduced CO (as a manifestation of CCM) is associated with the development of AKI (specifically hepatorenal dysfunction) after infections such as SBP II-1 1 QTc interval prolongation is common in cirrhosis and may indicate a poor outcome • Agents that can prolong the QT interval should be used cautiously II-2 2 II-1 2 1 II-2 2 Detailed functional cardiac characterization should be part of the assessment for • TIPS insertion • LT Standardized criteria and protocols for the assessment of systolic and diastolic function in cirrhosis are needed EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Hepatopulmonary syndrome • Four main pulmonary complications may occur in patients with chronic liver disease – – • • Pneumonia Hepatic hydrotorax HPS Portopulmonary hypertension HPS is defined as a disorder in pulmonary oxygenation, caused by intrapulmonary vasodilatation and, less commonly, by pleural and pulmonary arteriovenous communications occurring in the clinical setting of portal hypertension Clinical manifestations of HPS in patients with chronic liver disease primarily involve dyspnoea and platypnoea EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
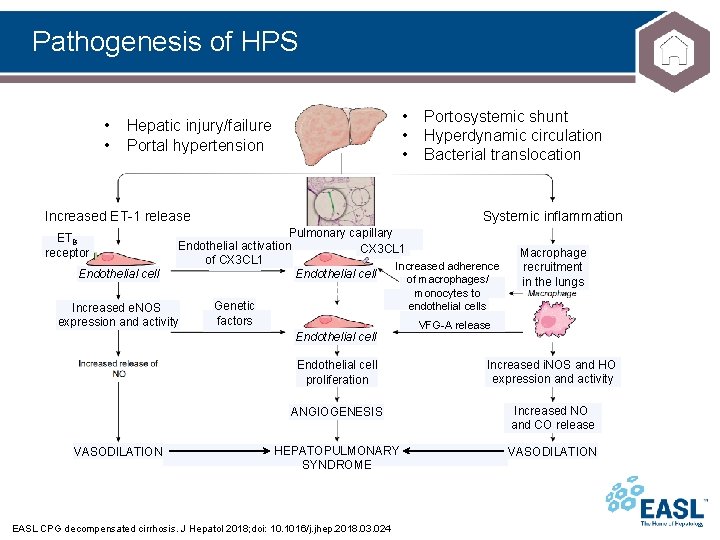
Pathogenesis of HPS • • • Hepatic injury/failure Portal hypertension Increased ET-1 release ETB receptor Endothelial cell Systemic inflammation Pulmonary capillary Endothelial activation CX 3 CL 1 of CX 3 CL 1 Increased adherence Endothelial cell of macrophages/ Increased e. NOS expression and activity monocytes to endothelial cells Genetic factors Endothelial cell VASODILATION Portosystemic shunt Hyperdynamic circulation Bacterial translocation Macrophage recruitment in the lungs VFG-A release Endothelial cell proliferation Increased i. NOS and HO expression and activity ANGIOGENESIS Increased NO and CO release HEPATOPULMONARY SYNDROME VASODILATION EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Diagnostic criteria for HPS • • • Hypoxia with partial pressure of oxygen <80 mm. Hg or alveolar–arterial oxygen gradient ≥ 15 mm. Hg in ambient air (≥ 20 mm. Hg in patients older than 65 years) Pulmonary vascular defect with positive findings on contrast-enhanced echocardiography or abnormal uptake in the brain (>6%) with radioactive lung-perfusion scanning Commonly in presence of portal hypertension, and in particular: – Hepatic portal hypertension with underlying cirrhosis – Pre-hepatic or hepatic portal hypertension in patients without underlying cirrhosis • Less commonly in presence of: – Acute liver failure, chronic hepatitis EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Diagnosis of HPS • In patients with portal hypertension and the clinical suspicion of HPS partial pressure of oxygen (Pa. O 2) in ABG should be assessed Recommendation Grade of evidence Grade of recommendation In patients with chronic liver disease, HPS should be suspected and investigated in presence of tachypnoea and polypnoea, digital clubbing and/or cyanosis II-2 1 Screening in adults: • If pulse oximetry Sp. O 2 <96% – ABG analysis should be performed • If ABG Pa. O 2 <80 mm. Hg and/or P[A-a]O 2 ≥ 15 mm. Hg* (in ambient air) – further investigations should be performed II-2 1 The use of contrast (microbubble) echocardiography to characterize HPS is recommended II-2 1 *For adults ≥ 65 years a P[A-a]O 2 ≥ 20 mm. Hg cut-off should be used EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Diagnosis of HPS • When Pa. O 2 suggests HPS, further investigations are needed to determine the underlying mechanism Recommendation Grade of evidence Grade of recommendation MAA scan should be performed to quantify the degree of shunting in patients with severe hypoxaemia and coexistent intrinsic lung disease, or to assess the prognosis in patients with HPS and very severe hypoxaemia (Pa. O 2 <50 mm. Hg) II-2 1 Neither contrast echocardiography nor MAA scan definitively differentiate discrete arteriovenous communications from diffuse precapillary and capillary dilatations or cardiac shunts • Pulmonary angiography should be performed only in patients with the severe hypoxaemia (Pa. O 2 <60 mm. Hg), poorly responsive to administration of 100% oxygen, and in whom there is a strong suspicion of arteriovenous communications that are amenable to embolization II-2 1 Trans-oesophageal contrast-enhanced echocardiography (although associated with risks) can definitively exclude intra-cardiac shunts II-2 2 EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
Management of HPS • There is no established medical therapy currently available for HPS, the only successful treatment for HPS is LT Recommendations for medical treatment Grade of evidence Grade of recommendation Long-term oxygen therapy is recommended in patients with HPS and severe hypoxaemia despite the lack of available data concerning effectiveness, tolerance, cost effectiveness, compliance and effects on survival rates of this therapy II-2 1 No recommendation can be proposed regarding the use of drugs or the placement of TIPS for the treatment of HPS I 1 Patients with HPS and Pa. O 2 <60 mm. Hg should be evaluated for LT since it is the only treatment for HPS that has been proven to be effective to date II-2 1 Severe hypoxaemia (Pa. O 2 <45– 50 mm. Hg) is associated with increased post-LT mortality • ABG analysis should be carried out every 6 months to facilitate prioritization to LT II-2 1 Recommendations for liver transplantation EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Portopulmonary hypertension • • PPHT occurs in patients with portal hypertension in the absence of other causes of arterial or venous hypertension Classification is based on mean pulmonary arterial pressure (m. PAP), and assumes high pulmonary vascular resistance (PVR) and normal pulmonary occlusion pressures – Mild: m. PAP ≥ 25 and <35 mm. Hg – Moderate: m. PAP ≥ 35 and <45 mm. Hg – Severe: m. PAP ≥ 45 mm. Hg • • Incidence between 3– 10% cirrhosis patients based on haemodynamic criteria; women are at 3 x greater risk and it is more common in autoimmune liver disease There is no clear association between the severity of liver disease or portal hypertension and the development of severe PPHT EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Monitoring and medical management of PPHT • The evidence base for pharmacological therapies in PPHT is limited Recommendation Grade of evidence Grade of recommendation Screening for PPHT should be via TDE in patients deemed potential recipients for TIPS or LT • In those with a positive screening test, right heart catheterization should be performed II-1 1 In patients with PPHT who are listed for LT, echocardiography should be repeated on the waitlist (the specific interval is unclear) III 1 β-blockers should be stopped and varices managed by endoscopic therapy in cases of proven PPHT II-3 1 Therapies approved for primary pulmonary arterial hypertension may improve exercise tolerance and haemodynamics in PPHT • However, endothelin antagonists should be used with caution because of concerns over hepatic impairment II-2 1 TIPS should not be used in patients with PPHT II-3 1 EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Liver transplantation in PPHT • Although severe PPHT has, historically, been a contraindication for LT, the advent of improved haemodynamic control (with agents such as IV prostacyclin) allows LT to be considered Recommendation Grade of evidence Grade of recommendation If m. PAP <35 mm. Hg and right ventricular function is preserved, LT should be considered • m. PAP of ≥ 45 mm. Hg should be considered an absolute contraindication to LT irrespective of therapy applied II-2 1 III 1 Therapy to lower m. PAP and improve right ventricular function should be commenced in patients with m. PAP ≥ 35 mm. Hg • Right ventricular function should be periodically evaluated II-2 1 MELD exception can be considered in patients with proven PPHT in whom targeted therapy fails to decrease m. PAP <35 mm. Hg but does facilitate normalization of PVR to <240 dyn. s/cm-5 and right ventricular function II-3 2 MELD exception should be advocated in patients with proven PPHT of moderate severity (m. PAP ≥ 35 mm. Hg) in whom targeted treatment lowers m. PAP <35 mm. Hg and PVR <400 dyn. s/cm-5 II-2 1 EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Additional recommendations

Portal hypertension gastropathy • Often presents in patients with DC – Natural history significantly influenced by the severity of liver disease and portal hypertension Recommendation Grade of evidence Grade of recommendation NSBB and iron supplementation and/or blood transfusion, when indicated, are recommended as first-line therapy for chronic haemorrhage from PHG I 1 In patients with transfusion-dependent PHG in whom NSBBs fail or are not tolerated, covered TIPS placement may be used in the absence of contraindications II-3 2 I 2 Acute PHG bleeding may be treated with somatostatin analogues or terlipressin but substantiating data are limited EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Gastric varices: classification, prevalence and risk • The Sarin classification is most commonly used for risk stratification and management of gastric varices Relative frequency Overall bleeding risk without treatment GOV type 1 OV extending below cardia into lesser curvature 70% 28% GOV type 2 OV extending below cardia into fundus 21% 55% Type Definition Gastro-oesophageal varices (GOV) Isolated gastric varices (IGV) IGV type 1 Isolated varices in the fundus 7% 78% IGV type 2 Isolated varices else in the stomach 2% 9% EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
Management of gastric varices • Gastric varices are present in about 20% of patients with cirrhosis Recommendation Grade of evidence Grade of recommendation NSBBs are suggested for primary prevention of VH from GOV type 2 or IGV type 1 III 2 Primary prevention for GOV type 1 follows the recommendations of oesophageal varices III 2 I I 1 2 II-2 1 III 2 Acute gastric VH should be treated medically, like oesophageal VH • Cyanoacrylate is the recommended endoscopic haemostatic treatment for cardiofundal varices (GOV type 2 or IGV type 1) TIPS with potential embolization efficiently controls bleeding and prevents re-bleeding in fundal VH (GOV type 2 or IGV type 1) and should be considered in appropriate candidates Selective embolization (BRTO/BATO) may also be used to treat bleeding from fundal varices associated with large gastro/splenorenal collaterals, although more data is required EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

Diagnosis of SBP • Both neutrophil count and culture results should be taken into account Recommendation Grade of evidence Grade of recommendation Patients with bacterascites (neutrophil count <250/mm 3 but positive bacterial culture) exhibiting signs of systemic inflammation or infection should be treated with antibiotics • Otherwise, the patient should undergo a second paracentesis • If the culture results come back positive again, regardless of the neutrophil count, the patient should be treated II-2 1 III 1 Spontaneous bacterial pleural empyema diagnosed by: • Positive pleural fluid culture and neutrophil count >250/mm 3 or • Negative pleural fluid culture and neutrophil count >500/mm 3 in the absence of pneumonia II-2 1 Secondary bacterial peritonitis should be suspected in case of multiple organisms on ascitic culture, very high ascitic neutrophil count and/or high ascitic protein concentration, or in those patients with an inadequate response to therapy • Patients with suspected secondary bacterial peritonitis should undergo prompt CT scanning and early considerations for surgery III 1 EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
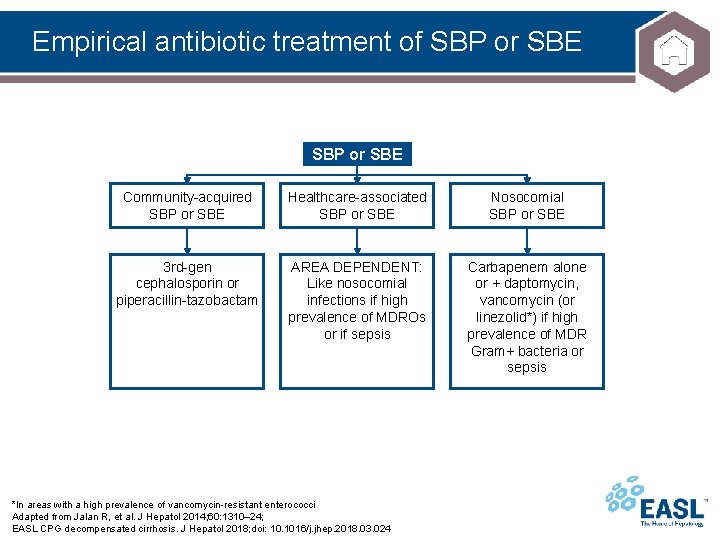
Empirical antibiotic treatment of SBP or SBE Community-acquired SBP or SBE Healthcare-associated SBP or SBE Nosocomial SBP or SBE 3 rd-gen cephalosporin or piperacillin-tazobactam AREA DEPENDENT: Like nosocomial infections if high prevalence of MDROs or if sepsis Carbapenem alone or + daptomycin, vancomycin (or linezolid*) if high prevalence of MDR Gram+ bacteria or sepsis *In areas with a high prevalence of vancomycin-resistant enterococci Adapted from Jalan R, et al. J Hepatol 2014; 60: 1310– 24; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

AKI management • Investigation and management should begin immediately Recommendation Grade of evidence Grade of recommendation Investigate AKI cause as soon as possible to prevent AKI progression • Management should begin immediately (even absent obvious cause) • Screening and treatment of infection are most important II-2 1 Diuretics and/or β-blockers as well as other drugs that could be associated with the occurrence of AKI such as vasodilators, NSAIDs and nephrotoxic drugs should be immediately stopped II-2 1 Volume replacement should be used in accordance with the cause and severity of fluid losses II-2 1 In case of no obvious cause of AKI, AKI stage >1 A or infection-induced AKI: • 20% albumin solution should be used at a dose of 1 g /kg of body weight (maximum of 100 g of albumin) for 2 consecutive days III 1 In patients with AKI and tense ascites, therapeutic paracentesis should be associated with albumin infusion even when a low volume of ascitic fluid is removed III 1 EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024

HRS outside AKI criteria • HRS-NAKI has an impaired response to vasorestrictors Recommendation Grade of evidence Grade of recommendation Vasoconstrictors and albumin are not recommended the treatment of HRS outside the criteria of AKI (HRS-NAKI)* • Terlipressin plus albumin is effective in the treatment of HRS-NAKI, but recurrence after withdrawal of treatment is the norm, and controversial data exist on the impact of the treatment on long-term clinical outcome, particularly from the perspective of LT *Formerly known as HRS type II EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024 I 1
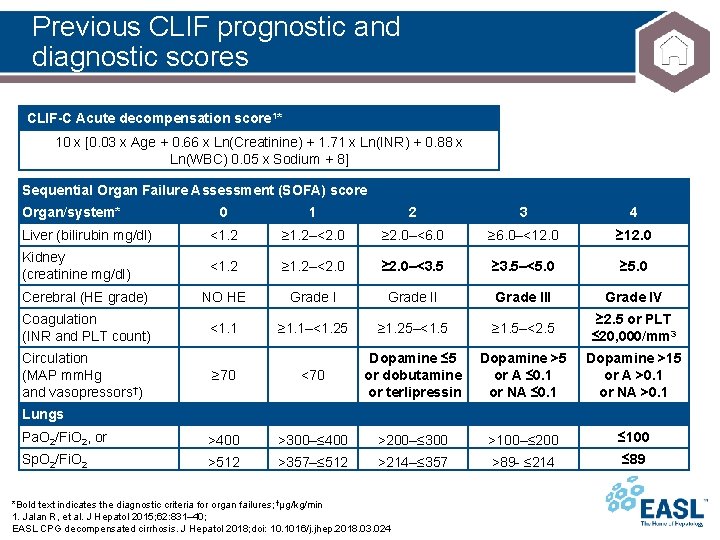
Previous CLIF prognostic and diagnostic scores CLIF-C Acute decompensation score 1* 10 x [0. 03 x Age + 0. 66 x Ln(Creatinine) + 1. 71 x Ln(INR) + 0. 88 x Ln(WBC) 0. 05 x Sodium + 8] Sequential Organ Failure Assessment (SOFA) score Organ/system* 0 1 2 3 4 Liver (bilirubin mg/dl) <1. 2 ≥ 1. 2–<2. 0 ≥ 2. 0–<6. 0 ≥ 6. 0–<12. 0 ≥ 12. 0 Kidney (creatinine mg/dl) <1. 2 ≥ 1. 2–<2. 0 ≥ 2. 0–<3. 5 ≥ 3. 5–<5. 0 ≥ 5. 0 Cerebral (HE grade) NO HE Grade III Grade IV Coagulation (INR and PLT count) <1. 1 ≥ 1. 1–<1. 25 ≥ 1. 25–<1. 5 ≥ 1. 5–<2. 5 ≥ 2. 5 or PLT ≤ 20, 000/mm 3 ≥ 70 <70 Dopamine ≤ 5 or dobutamine or terlipressin Dopamine >5 or A ≤ 0. 1 or NA ≤ 0. 1 Dopamine >15 or A >0. 1 or NA >0. 1 Pa. O 2/Fi. O 2, or >400 >300–≤ 400 >200–≤ 300 >100–≤ 200 ≤ 100 Sp. O 2/Fi. O 2 >512 >357–≤ 512 >214–≤ 357 >89 - ≤ 214 ≤ 89 Circulation (MAP mm. Hg and vasopressors†) Lungs *Bold text indicates the diagnostic criteria for organ failures; †μg/kg/min 1. Jalan R, et al. J Hepatol 2015; 62: 831– 40; EASL CPG decompensated cirrhosis. J Hepatol 2018; doi: 10. 1016/j. jhep. 2018. 03. 024
- Slides: 98