1 Chapter 8 Key Terms Oxidation Number Nomenclature

1 Chapter 8 Key Terms Oxidation Number Nomenclature Hydrate Anhydrous Binary Acid Ternary Acid Reactant Product Precipitate Catalyst Spectator Ion Replacement Reaction Decomposition Reaction Synthesis Reaction

2 Chapter 8 CHEMICAL COMPOSITION AND REACTIONS

3 Oxidation Numbers Scientists assign oxidation numbers to keep track of electrons during bonding Refers to the number of electrons an atom in a compound needs to gain or lose in order to return to its neutral state Remember that the more electronegative element will be given the negative oxidation number

4 Rules for Oxidation Numbers 1. Free (unbonded) atoms and atoms of pure elements have an oxidation number of zero 2. Oxidation number for a monatomic ion is equal to the charge 3. Sum of oxidation numbers of all atoms in a compound must be zero 4. Also applies for partial charges (covalent bonds) – H 2 O Certain elements have the same oxidation number in most of their compounds

5 Rules for Oxidation Numbers 4. Certain elements have the same oxidation number in most of their compounds a) Alkali metals always have +1 oxidation number b) Alkaline-earth metals always have +2 oxidation number c) Hydrogen has +1 oxidation number except when bonded to metals d) Oxygen is assigned -2 except when bonded with other more electronegative elements e) Halogens have oxidation number of -1 when bonded to metals If oxidation number is unknown, it can be found out by using a very simple algebraic equation

6 WHY? ? ? Scientists use oxidation numbers to help in writing chemical formulas They can determine how many of each element is needed in order to form a neutral compound Total positive and negative charges must be equal

7 Oxidation Numbers Rule 5: The oxidation numbers of all the atoms in a polyatomic ion add up to the charge of the ion OH NH 4 When these polyatomic ions are used in compounds and more than one is needed, be sure to include parentheses around the entire ion Ca(OH)2

8 Oxidation Numbers Practice Show the oxidation numbers for each element in the following compounds: NH 4 Br Na. NO 3 NH 3 Zn. O H 2 O CO 2

9 Practice Continued N 2 KMn. O 4 Al(OH)3 Mg 3(PO 4)2 Li. H Ca. H 2

10 Nomenclature Scientists give specific names to compounds to provide much information about that compound Ex. The Soda Ash vs. Sodium Carbonate IUPAC name tells what elements are present, the types of bonds, intermolecular attractions and general properties of the compound

11 Non-acidic Covalent Compounds Binary covalent compounds that are not acids (do not have H Greek Prefix Value as first element) use the Greek prefix system Least Ex. electronegative element stated 1 st CO Ending of last element changed to –ide Mono is only for the second element unless it is needed for clarity or emphasis Extra vowels omitted for clarity Mono 1 Di 2 Tri 3 Tetra 4 Penta 5 Hexa 6 Hepta 7 Octa 8 Nona 9 Deca 10

12 Practice CO 2 H 2 O PCl 3 CH 4 N 2 S 5

13 Binary Ionic Compounds Do not use the Greek prefixes, but are named according to the 2 ions involved Positive ions keep the same name (Na+1 = Sodium) Negative Positive ions gain the –ide ending (Cl-1 = Chloride) ion named 1 st, then the negative ion (sodium chloride)

14 Practice Na. Cl Ca. O Al. Br 3 Rb 2 S Potassium bromide Strontium oxide Sodium hydride Aluminum selenide

15 Review P 2 O 5 Ga. Cl 3 Si. O 2 Dinitrogen trioxide Zinc sulfide See page 199 for common polyatomic ions

16 Polyatomic Ions Same as binary ionic compounds except the –ide name is replaced with the name of the polyatomic ionic Treat the polyatomic ion as a single element (cation listed first, anion listed second) Ex. Na 2 SO 4 Polyatomic ions that involve oxygen and one other element are called oxyanions

17 Practice KOH Ba(Cl. O 3)2 Ca(Cl. O)2 Barium nitrite Ammonium chloride

18 Atoms with Multiple Oxidation States Usually Uses transition metals the Stock System/Roman Numeral System Cation listed first, then anion A Roman numeral follows the cation to signify the oxidation number of the cation Ex. Hg 2 I 2 Mercury (I) iodide

19 Practice Hg. I 2 Fe. Cl 3 Sn. O 2 Pb(SO 4)2 Copper Lead (I) bromide (II) phosphate Mercury Cobalt (II) bromate (II) oxalate

20 Hydrates Compounds that hold water molecules in their crystalline structure Called the “Water of Hydration” Indicated in the formula - Na 2 CO 3 • 3 H 2 O “Hydrate” is added after the name of the compound with the Greek prefix describing how many water molecules Anhydrous – compound that does not contain water of hydration in its crystalline structure Identifies the dehydrated form of the compound

21 Binary Acids When they exist as a gas, they are named the same as nonacidic covalent compounds HCl is called Hydrogen chloride When these compounds are dissolved in water, they form acids Prefix hydro-, suffix –ic added to the anion, and the word acid HCl dissolved in water is now named Hydrochloric acid HBr H 2 S

22 Ternary Acids Composed Name of Hydrogen, Oxygen and another nonmetal is derived from the anion in the compound If the anions name ends in –ate, the ending changes to –ic If the anions name ends in –ite, the ending changes to –ous Word acid is added to the end of the compound HNO 3 H 2 SO 4 H 2 SO 3

23 Chemical Equations They describe the reaction Substances present before reaction Substances present after reaction Solid, Show liquid, gas the composition of the substance What type and how many of each element is present (molecular formulas) Must account for all the atoms involved in the reaction “What goes in must come out. ”

24 Chemical Equations Ca(HCO 3)2 + Ca(OH)2 H 2 O + Ca. CO 3 2 Ca atoms 1 Ca atom 4 H atoms 2 C atoms 1 C atom 8 O atoms 4 O atoms Balanced chemical equations Equations change Ca(HCO 3)2 that account for all atoms, and the mass does not + Ca(OH)2 2 H 2 O + 2 Ca. CO 3

25 Parts of an Equation Ca(HCO 3)2 + Ca(OH)2 Reactants: substances that are present before the reaction Products: 2 H 2 O + 2 Ca. CO 3 substances that emerge from the reaction Subscripts and coefficients

26 Special Symbols Double arrows show that the reaction goes forward and backward The physical states of those compounds is shown Solid (s), liquid (l), gas (g) Aqueous (aq) means substance is dissolved in water
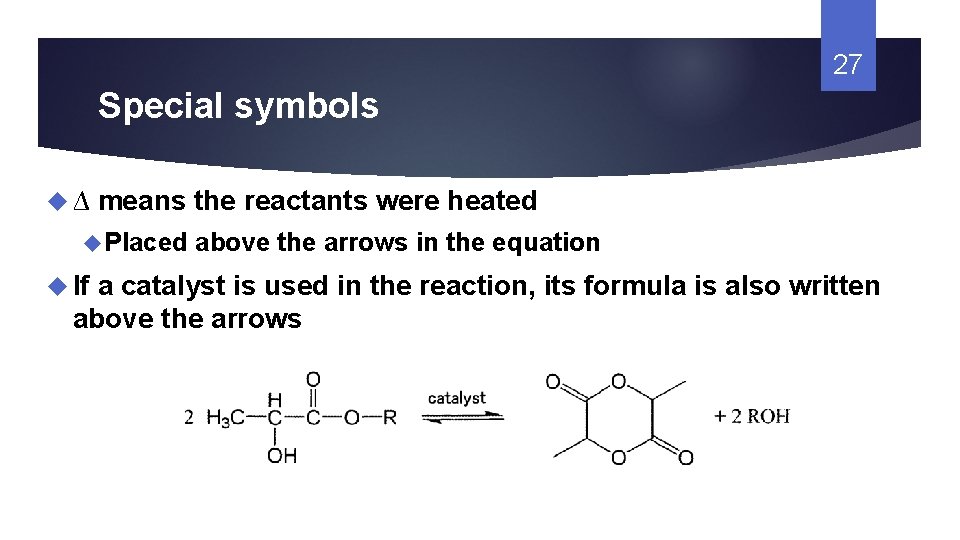
27 Special symbols ∆ means the reactants were heated Placed If above the arrows in the equation a catalyst is used in the reaction, its formula is also written above the arrows

28 Balancing Equations Adjust coefficients to show that atoms and mass are conserved General guidelines when balancing equations 1. Write the correct formulas for all reactants and products Nitrogen monoxide + oxygen NO + O 2 nitrogen dioxide NO 2 2. Count atoms of each kind to see if equation is already balanced N=1 O=3 O=2

29 Balancing Equations 3. Adjust coefficients until there are equal numbers of atoms on both sides of the arrow 4. Always check to be sure the coefficients are all whole numbers and that they are in the simplest ratio possible Balancing Chemical Equations

30 Types of Reactions Combination Combine Reactions two or more substances(reactants) into a single product Often called “synthesis reactions” Represented Ex. by the general equation A + B When Magnesium reacts with Oxygen 2 Mg + O 2 2 Mg. O AB

31 Types of Reactions Decomposition When This a substance breaks apart into two or more substances reaction usually requires input of energy General H 2 CO 3 Reactions equation: AB CO 2 + H 2 O A+B

32 Types of Reactions Single Replacement Reactions When an active element replaces a less active element in a compound Also called “displacement reaction” or “substitution reaction” General equation: A + BZ Mg + 2 HCl Mg. Cl 2 + H 2 B + AZ

33 Types of Reactions Double When Replacement Reactions two compounds switch partners with each other Most occur in an aqueous solution and are often characterized by a precipitate General equation: AX + BZ AZ + BX HCl + KOH HOH + KCl

34 Reactions Ionic equation Represents Soluble all the particles present before and after the reaction ionic compounds are shown as separate ions Insoluble ionic compounds are not Pb 2+ (aq) + 2 NO 3 - (aq) + 2 K+ (aq) + Cr. O 42 - (aq) Pb. Cr. O 4 (s) + 2 K+ + 2 NO 3 - (aq) Spectator Appear Ions in the reactants and products, but do not precipitate or join other ions

35 Reactions Net ionic equations Shows only the ions that actually react; spectator ions are left out Pb 2+ (aq) + 2 NO 3 - (aq) + 2 K+ (aq) + Cr. O 42 - (aq) Pb. Cr. O 4 (s) + 2 K+ + 2 NO 3 - (aq) Pb 2+ (aq) + Cr. O 42 - (aq) Pb. Cr. O 4 (s)
- Slides: 35