1 Cancer Registry Reporting Use Case November 4

|1| Cancer Registry Reporting Use Case November 4 th, 2020 HL 7® FHIR® is a registered trademark owned by Health Level Seven International® Approved for Public Release; Distribution Unlimited. Case Number 19 -2023

minimal Common Oncology Data Elements A FHIR-based core set of common data elements for cancer that is standardized, computable and clinically applicable in every electronic patient record with a cancer diagnosis A standard health record for oncology The minimal set of data elements applicable to all cancers, and collected for: Patients Providers CANCER Research Payers Standardized information exchange Use-case driven and targeted use Oncology data element domains: patient, disease, treatment, outcomes, genomics, lab/vital m. CODE Release 1 STU 1: http: //hl 7. org/fhir/us/mcode/ Vendors © 2020 The MITRE Corporation. All rights reserved. Approved for public release. Distribution unlimited 19 -03297 -19 Government/ Regulatory

A New HL 7 FHIR Accelerator A member-driven community accelerating interoperable data modeling and implementation around the FHIR and m. CODE HL 7 standards, leading to step-change improvements in cancer care and research http: //hl 7. org/Code. X
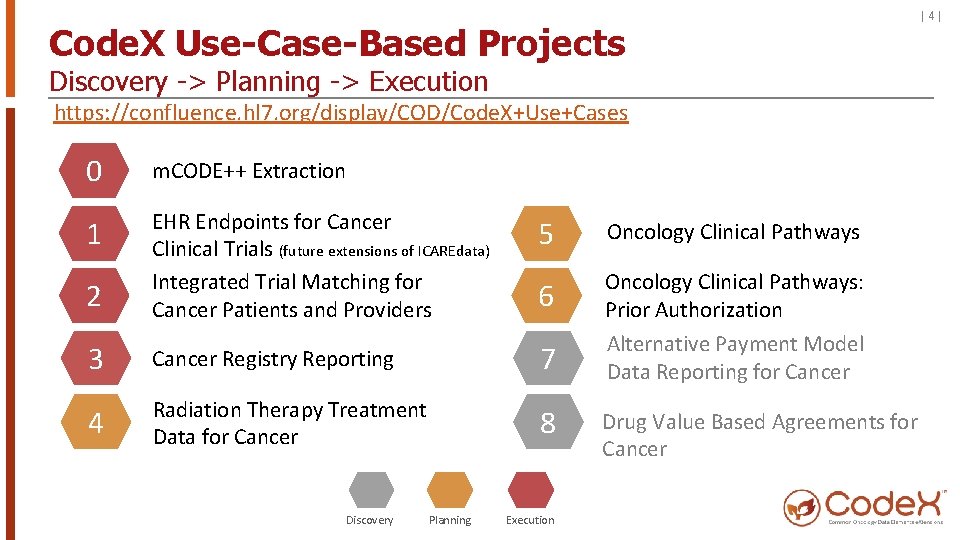
Code. X Use-Case-Based Projects Discovery -> Planning -> Execution https: //confluence. hl 7. org/display/COD/Code. X+Use+Cases 0 m. CODE++ Extraction 1 5 Oncology Clinical Pathways 2 EHR Endpoints for Cancer Clinical Trials (future extensions of ICAREdata) Integrated Trial Matching for Cancer Patients and Providers 6 3 Cancer Registry Reporting 7 Oncology Clinical Pathways: Prior Authorization Alternative Payment Model Data Reporting for Cancer 4 Radiation Therapy Treatment Data for Cancer 8 Discovery Planning Execution Drug Value Based Agreements for Cancer |4|

|5| 3 Cancer Registry Reporting Problem • • Clinical data is stored in disparate systems in multiple data formats Variability in data collection processes imposes a high burden on data reporters and negatively impacts understanding of patient care • Solution Heterogeneity of data collection makes it difficult to aggregate and share data for use in clinical research and standards of care • A low-burden, standardized reporting of cancer data from cancer centers to registries that are aggregating data for different reasons Desired Impact • As a patient begins and continues cancer care, outcomes are tracked, and effectiveness of care is determined in real time and reported in a low burden and interoperable manner • Reduce reporting burden and enhance insights into clinical practice
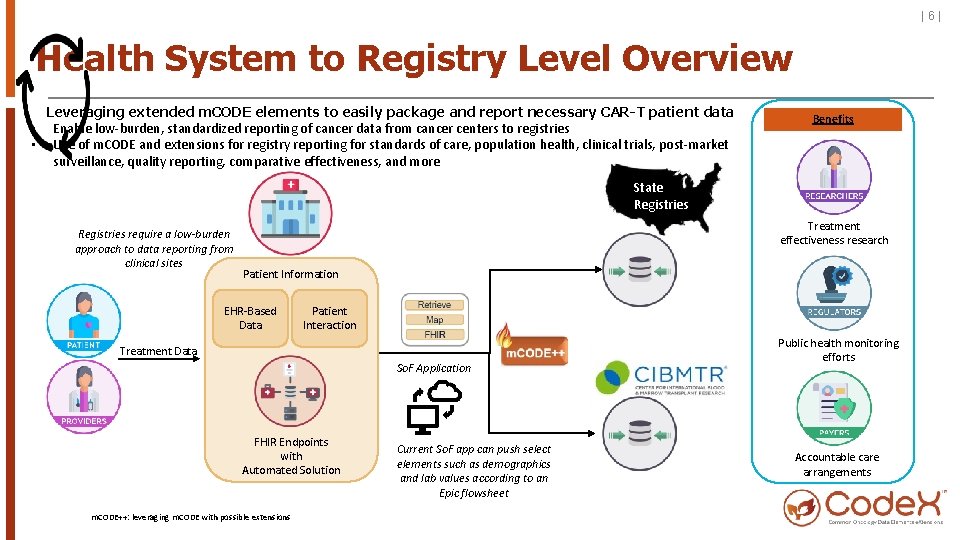
|6| Health System to Registry Level Overview Leveraging extended m. CODE elements to easily package and report necessary CAR-T patient data • Enable low-burden, standardized reporting of cancer data from cancer centers to registries • Use of m. CODE and extensions for registry reporting for standards of care, population health, clinical trials, post-market surveillance, quality reporting, comparative effectiveness, and more Benefits State Registries require a low-burden approach to data reporting from clinical sites Treatment effectiveness research Patient Information EHR-Based Data Patient Interaction Treatment Data So. F Application FHIR Endpoints with Automated Solution m. CODE++: leveraging m. CODE with possible extensions Current So. F app can push select elements such as demographics and lab values according to an Epic flowsheet Public health monitoring efforts Accountable care arrangements
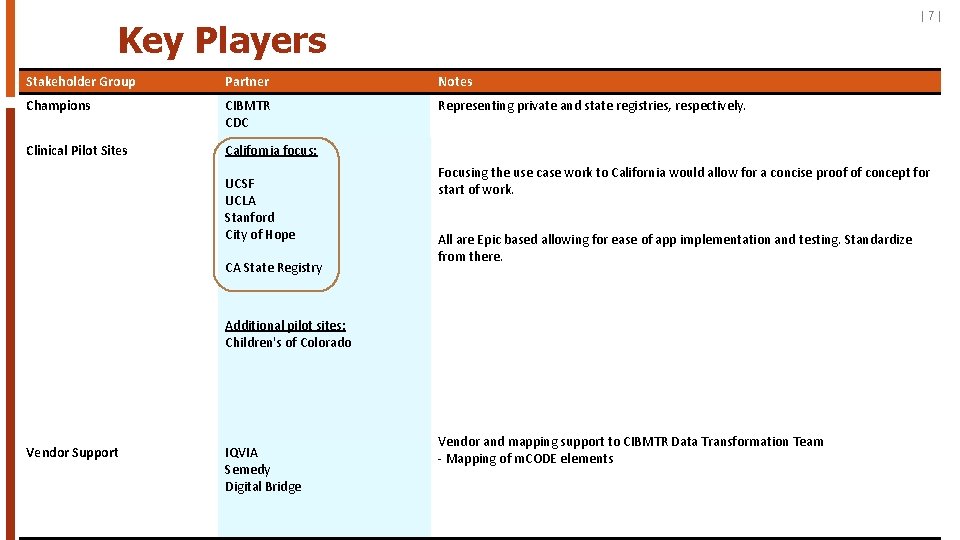
|7| Key Players Stakeholder Group Partner Notes Champions CIBMTR CDC Representing private and state registries, respectively. Clinical Pilot Sites California focus: UCSF UCLA Stanford City of Hope CA State Registry Focusing the use case work to California would allow for a concise proof of concept for start of work. All are Epic based allowing for ease of app implementation and testing. Standardize from there. Additional pilot sites: Children's of Colorado Vendor Support IQVIA Semedy Digital Bridge Vendor and mapping support to CIBMTR Data Transformation Team - Mapping of m. CODE elements
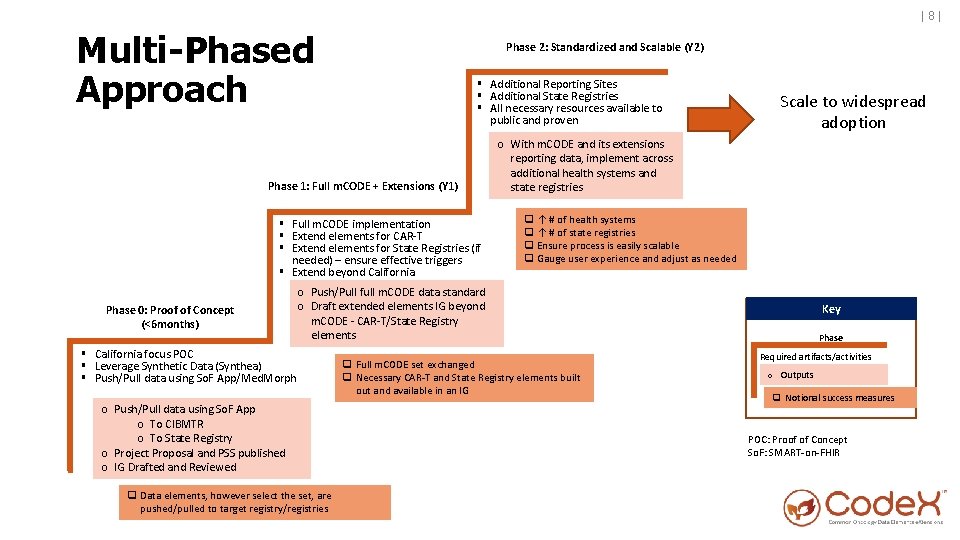
|8| Multi-Phased Approach Phase 2: Standardized and Scalable (Y 2) § Additional Reporting Sites § Additional State Registries § All necessary resources available to public and proven Phase 1: Full m. CODE + Extensions (Y 1) § Full m. CODE implementation § Extend elements for CAR-T § Extend elements for State Registries (if needed) – ensure effective triggers § Extend beyond California Phase 0: Proof of Concept (<6 months) Scale to widespread adoption o With m. CODE and its extensions reporting data, implement across additional health systems and state registries q ↑ # of health systems q ↑ # of state registries q Ensure process is easily scalable q Gauge user experience and adjust as needed o Push/Pull full m. CODE data standard o Draft extended elements IG beyond m. CODE - CAR-T/State Registry elements § California focus POC § Leverage Synthetic Data (Synthea) § Push/Pull data using So. F App/Med. Morph o Push/Pull data using So. F App o To CIBMTR o To State Registry o Project Proposal and PSS published o IG Drafted and Reviewed q Data elements, however select the set, are pushed/pulled to target registry/registries q Full m. CODE set exchanged q Necessary CAR-T and State Registry elements built out and available in an IG Key Phase Required artifacts/activities o Outputs q Notional success measures POC: Proof of Concept So. F: SMART-on-FHIR

|9| 3 Cancer Registry Reporting (Update as of October 2020) Collaborators Centers for Disease Control and Prevention, California State Cancer Registry, MITRE, University of California - San Francisco, Stanford Project Updates • Phase 0 Working Group kickoff meeting held on October 27 th • Phase 0 scope of work: subset of current m. CODE-compliant data elements will be sent via FHIR to both a private and state registry • Onboarding University of California – Los Angeles, and Children’s Hospital of Colorado • Discussing project opportunities with American Society of Clinical Oncology/Cancer. Lin. Q, California State Cancer Registry, University of California - San Francisco, Stanford, Digital Bridge, and CDC’s Med. Morph • Have created a synthetic patient data module for CAR-T, actively reviewing with clinicians https: //confluence. hl 7. org/display/COD/m. CODE+Test+Data

Organization of Work Confluence Public Site: https: //confluence. hl 7. org/display/COD/Cancer+Registry+Reporting We also have a private site for internal organization, progress tracking, resources Cadence of work: - Kickoff was on October 27 th - Biweekly All Workgroup Meetings - Monthly Leadership Call - Convening 1) Terminology and 2) Technical Architecture subgroups - Community of Practice https: //confluence. hl 7. org/display/COD/m. CODE+Community+of+Practice © 2020 The MITRE Corporation. All rights reserved. Approved for public release. Distribution unlimited 19 -03297 -19

| 11 | Impact • Smarter data for the fight against cancer that is instantly and easily reportable to registries • Approach is reproducible across all reporting sites and to all registries • Burden reduced on data managers/reporters, and unique therapies like CAR-T are better understood

| 12 | Leveraging the m. CODE™ standard (minimal Common Oncology Data Elements), Code. X will expand around this core to encompass additional use cases, accelerating opportunities to create a learning health system based on interoperable data and improved patient care. Learn more www. hl 7. org/codex/
- Slides: 12