1 BTW Turn on the recorder 3 Note

1 BTW: Turn on the recorder

3 Note to myself for Lec. 1: Web site is required reading (at least twice a week) Problem book Web lectures Email questions, Q&A database, office hours Evening vs. morning lectures Recitation sign-up Note exam dates and times (drop an exam); note final date Transparency Exam topics, nature

What is expected of you as a student If you can: then you will: 1) Repeat what you have heard exactly as it was presented, know all the terms and the definitions we or the texts have provided Probably fail 2) Repeat what you have heard in your own words, showing you have some understanding of the concepts and not just the words Probably pass with up to a B- 3) Apply what you have learned to a new situation you have never seen before thus proving that you have understood the concept and not just learned how to describe it. B to A 4) Synthesize a new application yourself or extend what you have learned to a new situation of your own design A
5 Physics
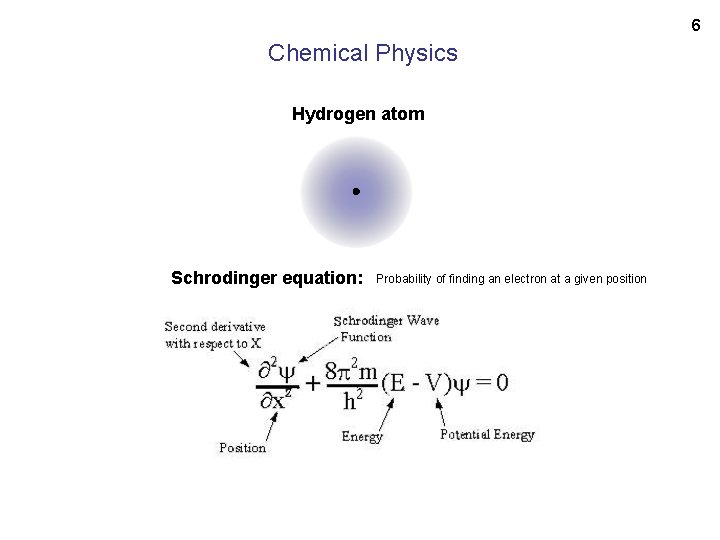
6 Chemical Physics Hydrogen atom Schrodinger equation: Probability of finding an electron at a given position
7 Chemistry http: //www. wou. edu/las/physci/ch 336/lecture/lect 10. htm

8 Biology Predicting the amount of energy released in a chemical reaction ΔG = ΔGo + RTln(Q) ΔGo = - RTln(Keq) [products] Q = [reactants]

Chemistry and Math for this course • • • basic atomic structure and bonding ions salts molecular weight stoichiometry chemical equilibria, equilibrium constants p. H etc. ? and: • • exponents logarithms algebra no calculus 9

Characteristics of living things 1) Structure = complex 2) Metabolism = chemical interaction with the environment 3) Reproduction = duplication of the complex, metabolizing structure 10

Artificial rubber plant vs. a real one: 1. complexity 11 Artificial: polypropylene polyester 5 dye molecules_____ 7 different distinguishable molecules Real: 20, 000 different distinguishable molecules And each one is doing a job.

Characteristics of living things 1) Structure = complex 2) Metabolism = chemical interaction with the environment 3) Reproduction = duplication of the complex, metabolizing structure 12
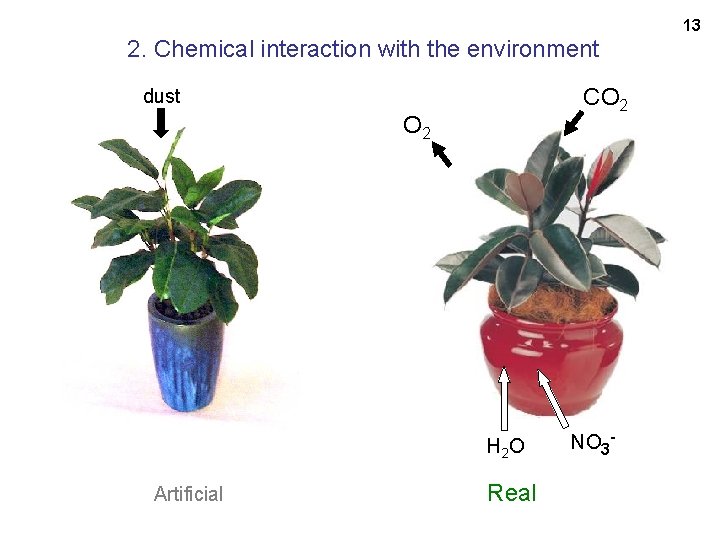
13 2. Chemical interaction with the environment CO 2 dust O 2 H 2 O Artificial Real NO 3 -

Characteristics of living things 1) Structure = complex 2) Metabolism = chemical interaction with the environment 3) Reproduction = duplication of the complex, metabolizing structure 14

15 3. Reproduction Cannot reproduce itself Can reproduce itself

16 Consider 2 approaches to studying biology: 1. Focusing on differences to learn about nature Darwin’s finches
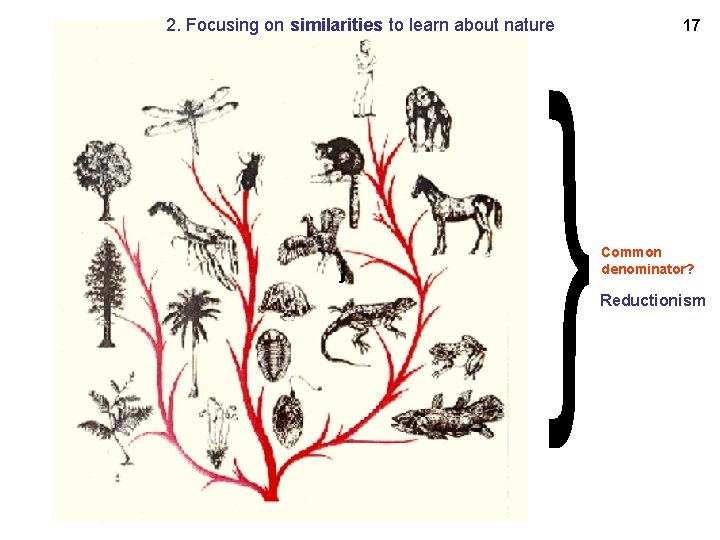
2. Focusing on similarities to learn about nature 17 Common denominator? Reductionism
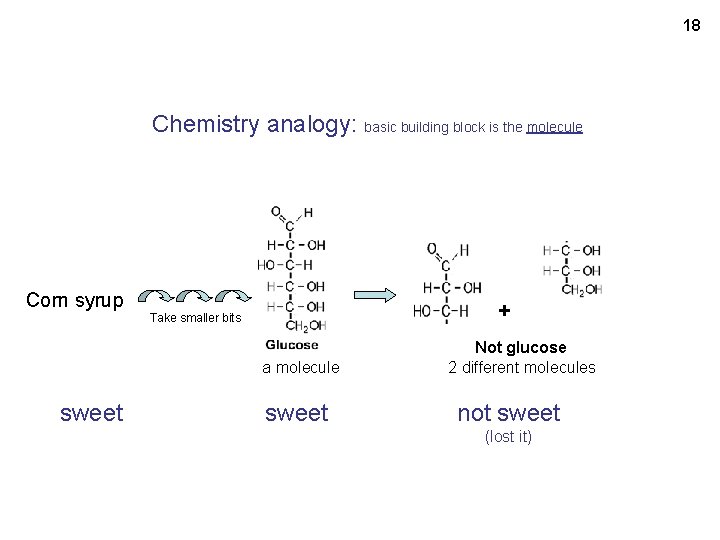
18 Chemistry analogy: basic building block is the molecule Corn syrup + Take smaller bits a molecule sweet Not glucose 2 different molecules not sweet (lost it)
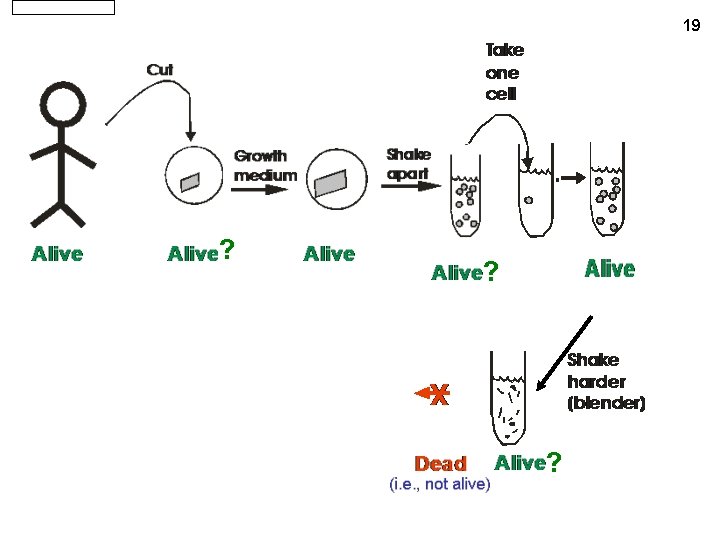

Cell Theory All living things are made up of cells (or their by-products), and all cells come from other cells by growth and development. 20
21 ‘ Outside Inside ‘
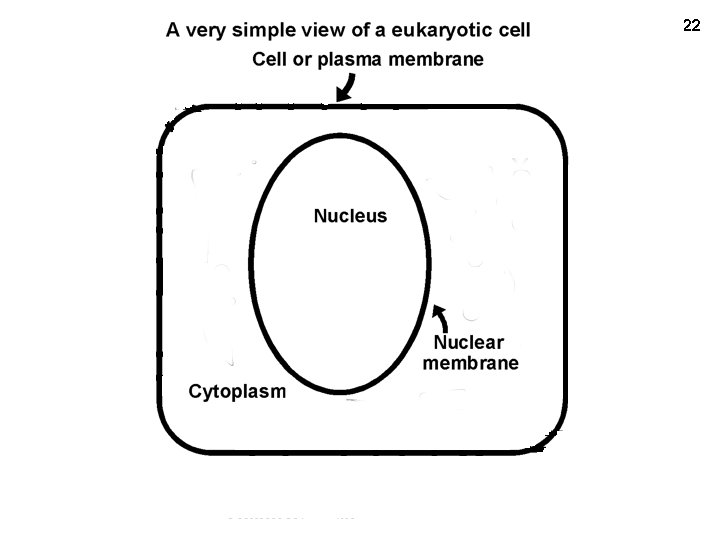
Mem+nuc 22
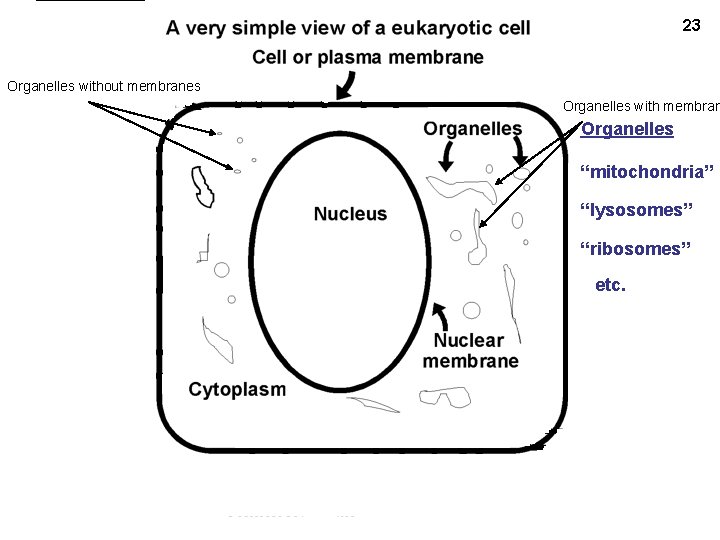
23 Organelles without membranes Organelles with membran Organelles “mitochondria” “lysosomes” “ribosomes” etc.
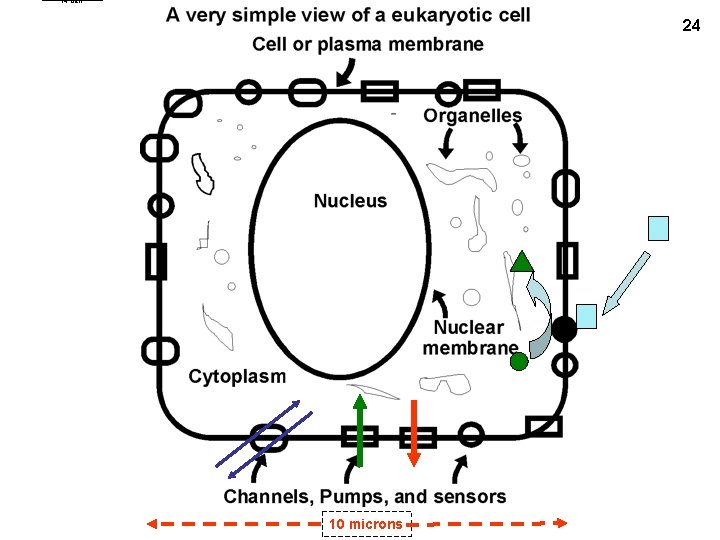
A cell 24 10 microns

Sizes • Skin cell ~ 10 micrometers (microns, um) in diameter – Millimeter (mm) = 1/1000 of a meter: e. g. , head of a pin – Micron = 10 -6 meters (1 millionth of a meter, 1/1000 of a millimeter): e. g. , cells – Nanometer (nm) = 10 -9 meters (1 billionth of a meter, 1/1000 of a micron): e. g. , diameter of molecules – Angstrom (A) = 1/10 of a nanometer: e. g, distance between 2 atoms in a molecule • Smallest cells ~ 1 micron in diameter (so volume = ~1/1000 of skin cell) 25

bactcell 0 26 A bacterial cell A bacterium
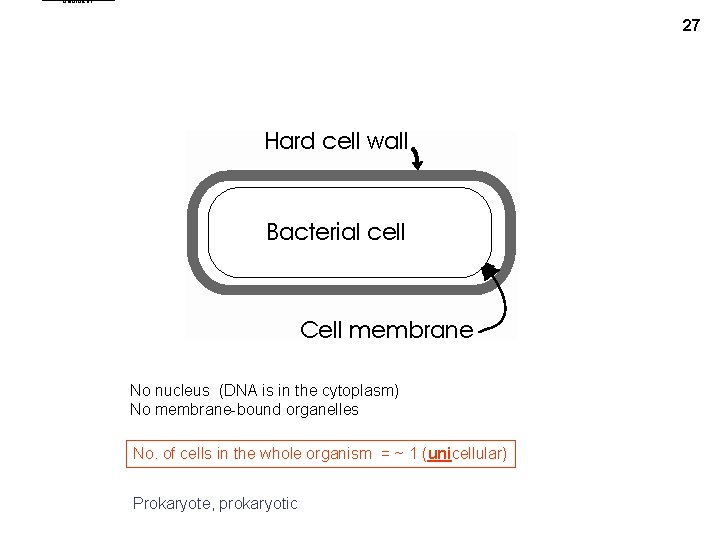
bactcell 1 27 No nucleus (DNA is in the cytoplasm) No membrane-bound organelles No. of cells in the whole organism = ~ 1 (unicellular) Prokaryote, prokaryotic
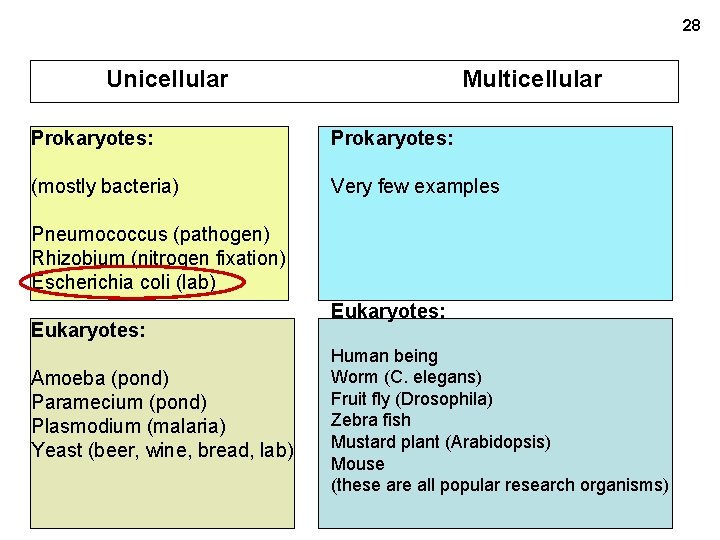
28 Unicellular Multicellular Prokaryotes: (mostly bacteria) Very few examples Pneumococcus (pathogen) Rhizobium (nitrogen fixation) Escherichia coli (lab) Eukaryotes: Amoeba (pond) Paramecium (pond) Plasmodium (malaria) Yeast (beer, wine, bread, lab) Eukaryotes: Human being Worm (C. elegans) Fruit fly (Drosophila) Zebra fish Mustard plant (Arabidopsis) Mouse (these are all popular research organisms)
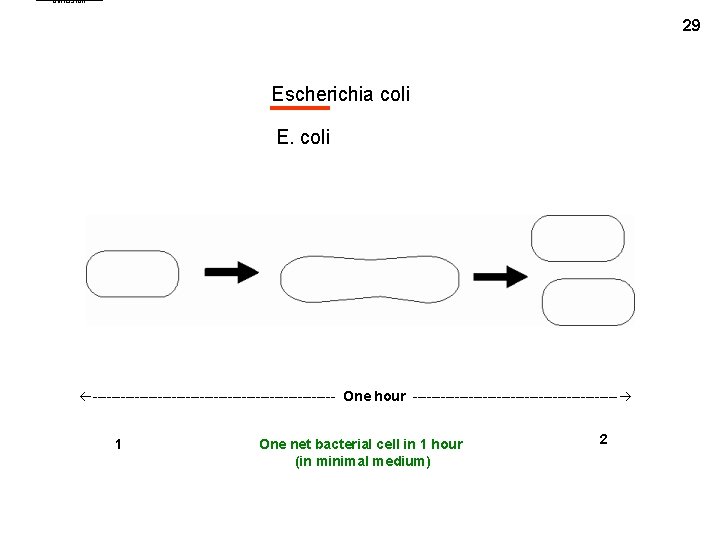
binfission 29 Escherichia coli E. coli -------------------------- One hour ---------------------- 1 One net bacterial cell in 1 hour (in minimal medium) 2
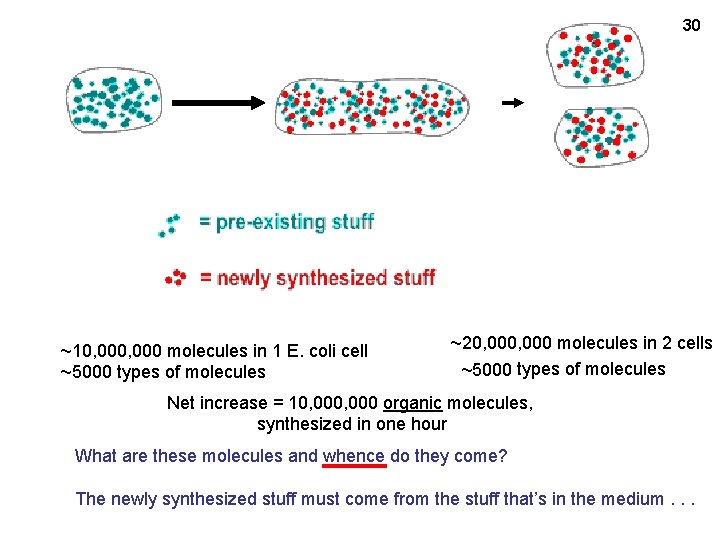
30 ~10, 000 molecules in 1 E. coli cell ~5000 types of molecules ~20, 000 molecules in 2 cells ~5000 types of molecules Net increase = 10, 000 organic molecules, synthesized in one hour What are these molecules and whence do they come? The newly synthesized stuff must come from the stuff that’s in the medium. . .

A minimal medium for E. coli C 6 H 12 O 6 glucose, a sugar KH 2 PO 4 potassium phosphate Mg. SO 4 magnesium sulfate NH 4 Cl ammonium chloride H 2 O water +trace elements (e. g. , Zn, Fe, Cu, Se, … ) 31
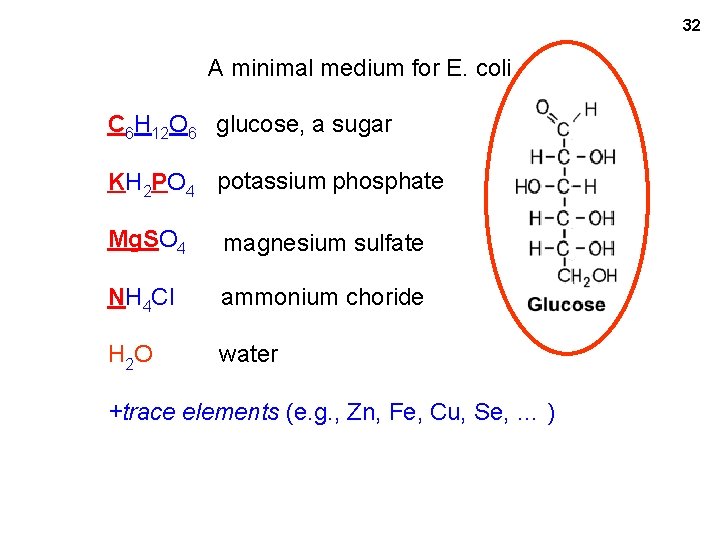
32 MM with glucose A minimal medium for E. coli C 6 H 12 O 6 glucose, a sugar KH 2 PO 4 potassium phosphate Mg. SO 4 magnesium sulfate NH 4 Cl ammonium choride H 2 O water +trace elements (e. g. , Zn, Fe, Cu, Se, … )
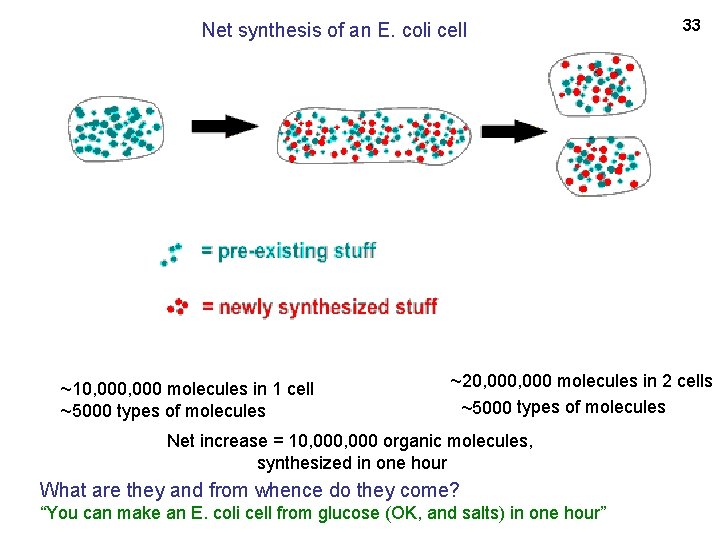
Net synthesis of an E. coli cell ~10, 000 molecules in 1 cell ~5000 types of molecules 33 ~20, 000 molecules in 2 cells ~5000 types of molecules Net increase = 10, 000 organic molecules, synthesized in one hour What are they and from whence do they come? “You can make an E. coli cell from glucose (OK, and salts) in one hour”
34 Preview • 1. What is an E. coli cell? – Polysaccharides, – Lipids, – Nucleic Acids, – Proteins, – Small molecules Large molecules Organic chemicals Small molecules • 2. How do we get those chemicals (in minimal medium)? -- From glucose, -- via biosynthetic chemical reactions (= metabolism). • 3. Where does the energy for this process come from? -- From glucose, via energy metabolism. • 4. Where does E. coli get the information for doing all this? -- it's hard-wired in its DNA.
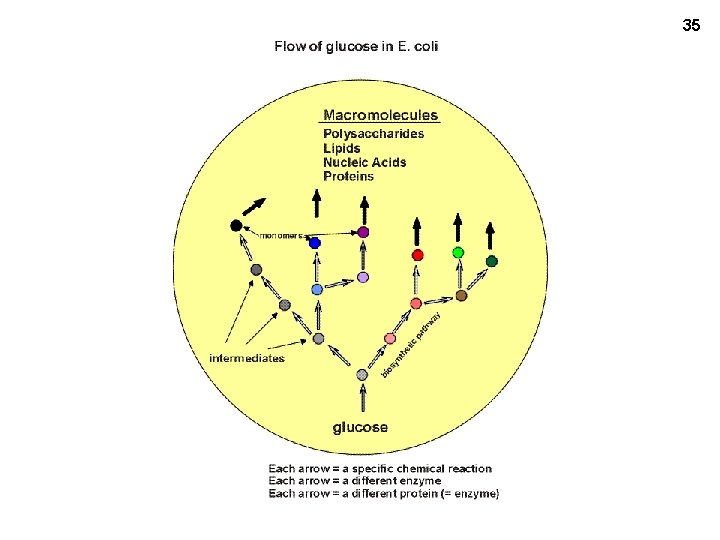
35
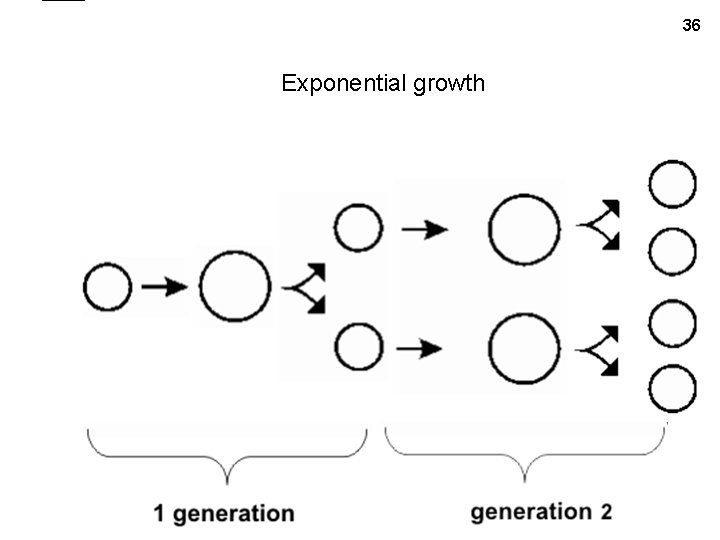
36 Exponential growth

• • So, starting with one cell, after 1 generation , get 2 cells, after 2 gens. , 4 cells, after 37 3 gens, 8 cells, etc. Looks like 2 is a key number: 21=2, 22 = 4, 23 = 8, … and so in general, N = 1 x 2 g • • • And if we start with 100 cells, then have 200, 400, 800, etc, so N = 100 x 2 g : Or in general: N = No x 2 g And to express growth in terms of real time: g = t/t. D where t. D = the doubling time, or generation time. • So the number of cells as a function of time is : N = No 2 t/t. D • Or: if we let k= 1/t. D, then N = No 2 kt • • But 2 is not a common base, so we can also write: N = No 10 k’t , but here k’ = log(2)/t. D rather than 1/t. D • Or we can use the natural log, e: N = Noek”t where k” = ln(2)/t. D • And if we take the log of both sides, we get (base 10 case): (“log” = log base 10) log(N/No) = k’t (k’ = log(2)/t. D = 0. 3/t. D) and ln(N/No) = k”t (k” = ln(2)/t. D = 0. 69/t. D) See exponential growth handout
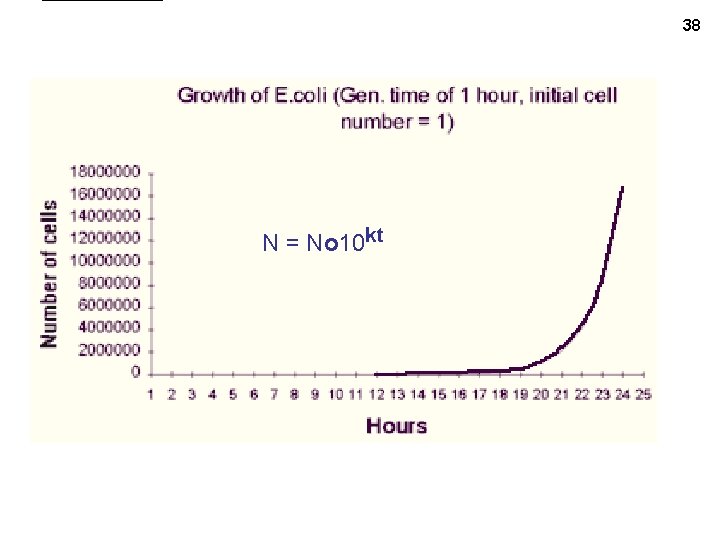
38 N = No 10 kt
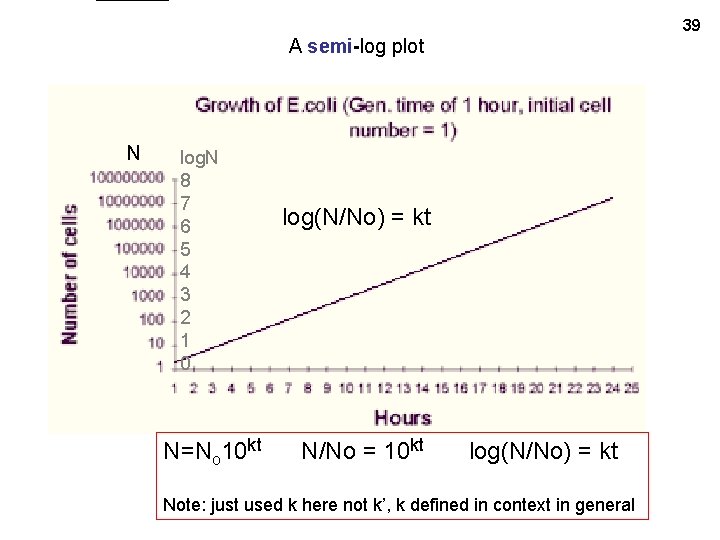
39 A semi-log plot N log. N 8 7 6 5 4 3 2 1 0 N=No 10 kt log(N/No) = kt N/No = 10 kt log(N/No) = kt Note: just used k here not k’, k defined in context in general

Got this far
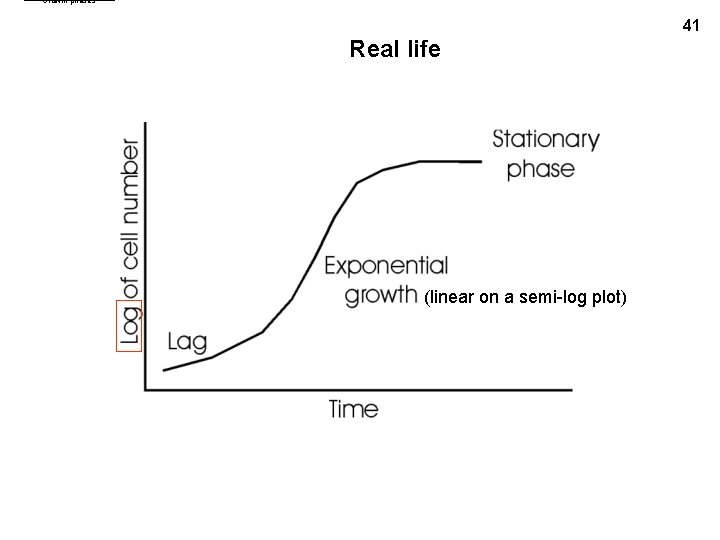
Growth phases 41 Real life (linear on a semi-log plot)

42 Use calculus if you know it, it’s more natural: d. N/dt = k. N Separating variables: d. N/N = kdt Integrating between time zero when N = No and time t, when N = N, d. N/N = kdt, we get: ln. N - ln No = kt - 0, or ln(N/No) = kt, or N = Noekt, which is exactly what we derived above. But is this k the same k as before? We can now calculate this constant k by considering the case of the time interval over which No has exactly doubled; in that case: N/No = 2 and t = t. D, so: N = Noekt 2 = ekt. D To solve for k, take the natural logarithm of both sides: ln 2=kt. D, or k=ln 2/t. D, so the constant comes out exactly as before as well. See exponential growth handout
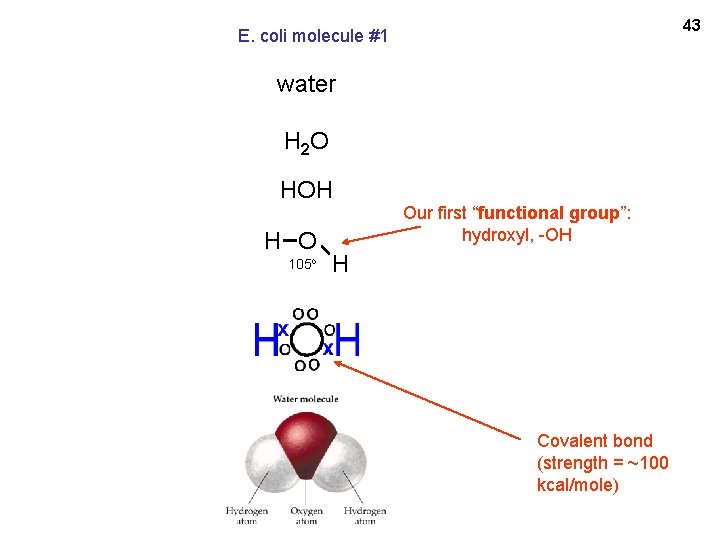
43 E. coli molecule #1 water H 2 O HOH H O 105 o Our first “functional group”: hydroxyl, -OH H Covalent bond (strength = ~100 kcal/mole)
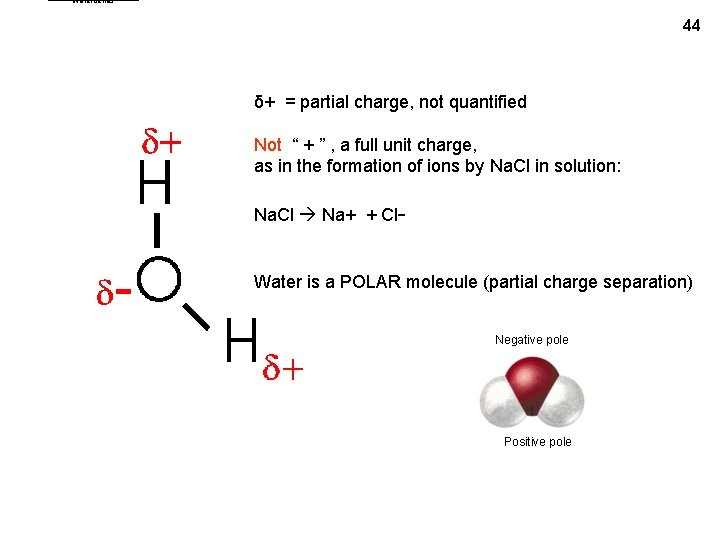
Waterdeltas 44 δ+ = partial charge, not quantified Not “ + ” , a full unit charge, as in the formation of ions by Na. Cl in solution: Na. Cl Na+ + Cl- Water is a POLAR molecule (partial charge separation) Negative pole Positive pole
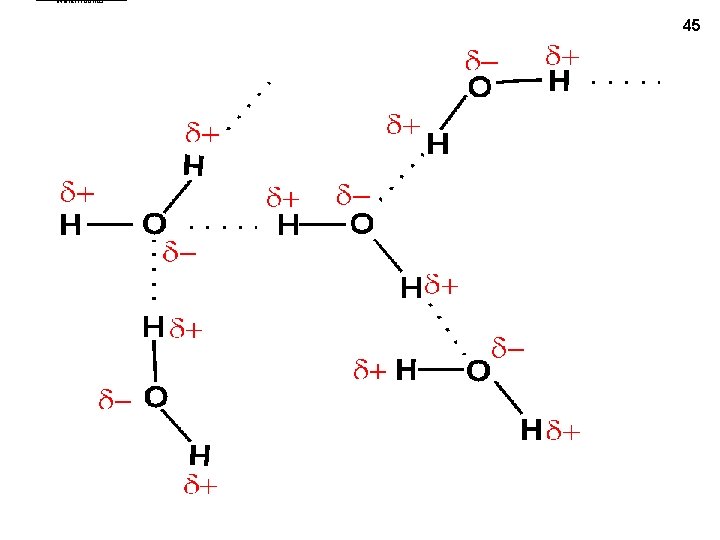
water. Hbonds 45
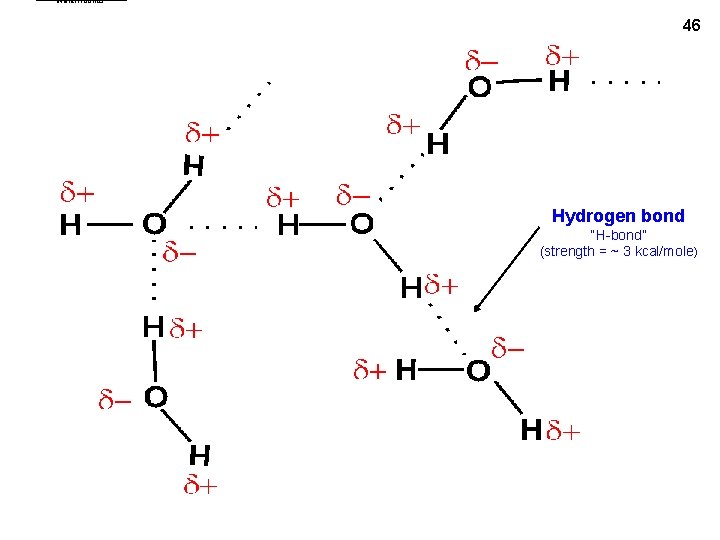
water. Hbonds 46 Hydrogen bond “H-bond” (strength = ~ 3 kcal/mole)
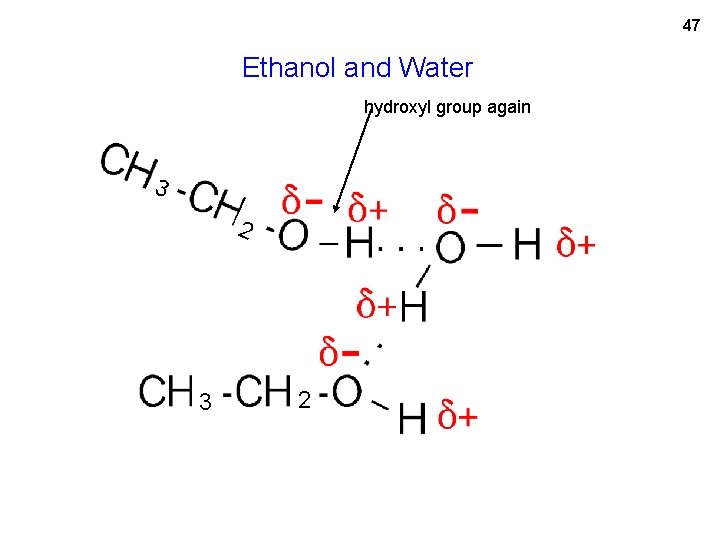
47 Ethanol and Water hydroxyl group again 3 2
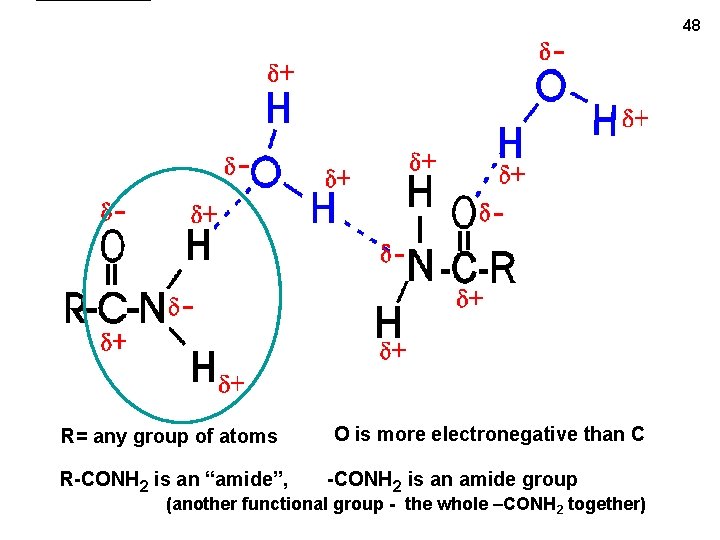
48 R= any group of atoms R-CONH 2 is an “amide”, O is more electronegative than C -CONH 2 is an amide group (another functional group - the whole –CONH 2 together)
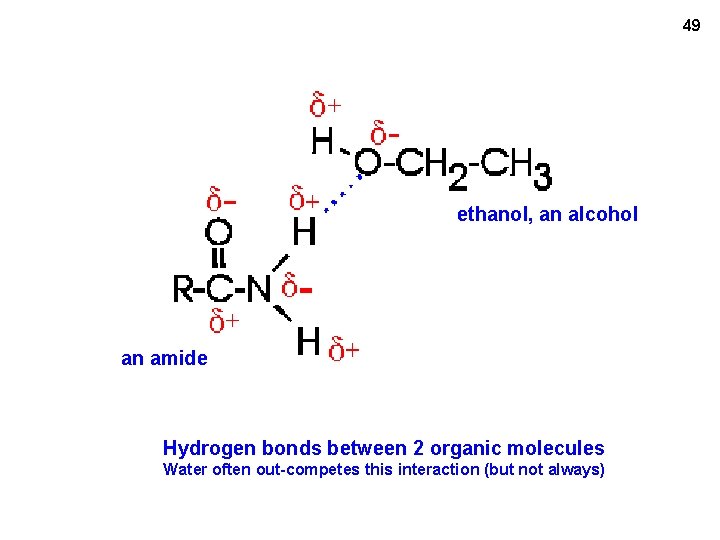
49 ethanol, an alcohol an amide Hydrogen bonds between 2 organic molecules Water often out-competes this interaction (but not always)
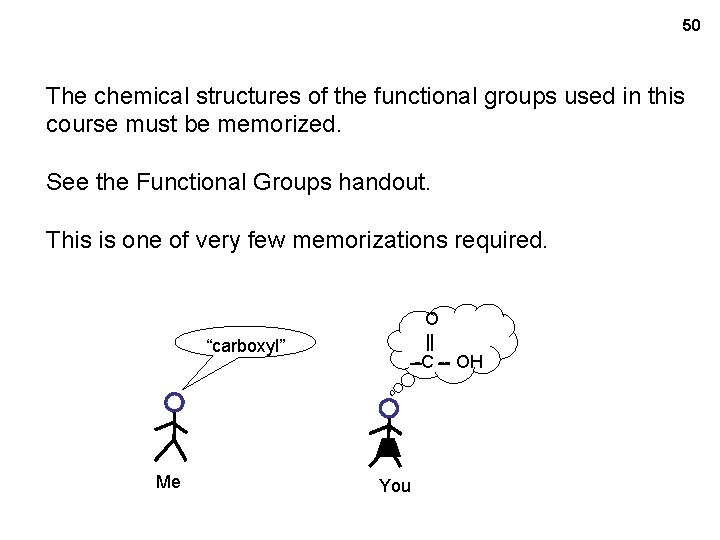
50 The chemical structures of the functional groups used in this course must be memorized. See the Functional Groups handout. This is one of very few memorizations required. O || -C -- OH “carboxyl” Me You
- Slides: 50