1 BIOGENETICALLY INSPIRED TOTAL SYNTHESIS OF LYCOPODIUM ALKALOIDS

1 BIOGENETICALLY INSPIRED TOTAL SYNTHESIS OF LYCOPODIUM ALKALOIDS, (+)-FLABELLIDINE AND (–)LYCODINE Masayuki Azuma, Tetsuya Yoshikawa, Noriyuki Kogure, Mariko Kitajima and Hiromitsu Takayama*

2 Introduction Retrosynthesis of the nature product Procedure of synthesis conclusion
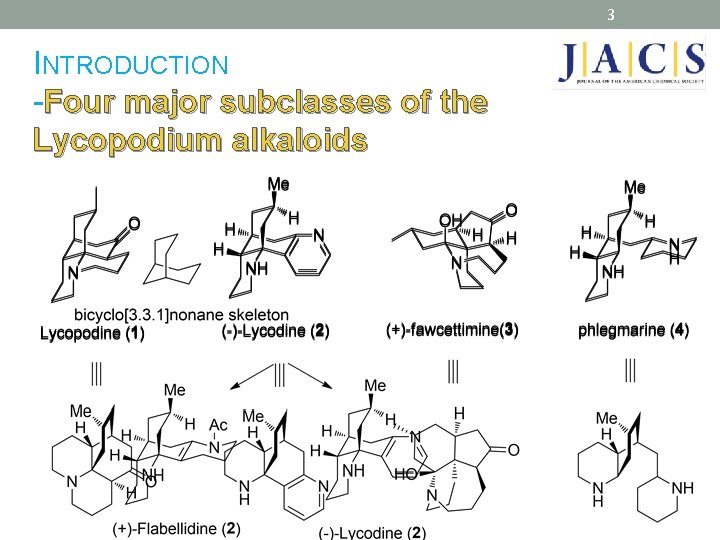
3 INTRODUCTION -Four major subclasses of the Lycopodium alkaloids
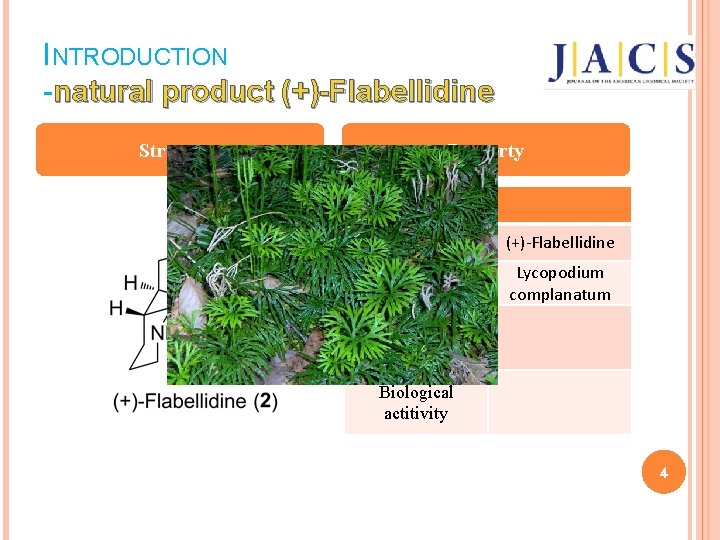
INTRODUCTION -natural product (+)-Flabellidine Structure Property term Name (+)-Flabellidine Isolated from Lycopodium complanatum Isolated by Biological actitivity 4

INTRODUCTION -natural product (-)-Lycodine Structure Property term Name (-)-Lycodine Isolated from Lycopodium annotinum Isolated by Anet and Eves Biological actitivity 5
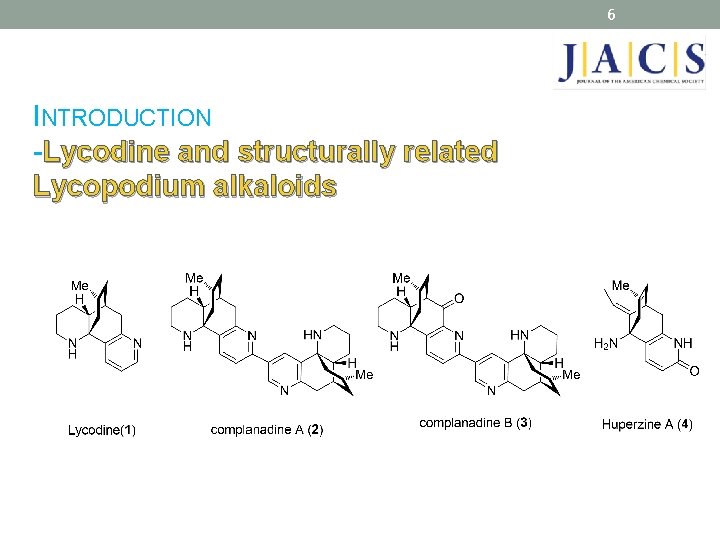
6 INTRODUCTION -Lycodine and structurally related Lycopodium alkaloids
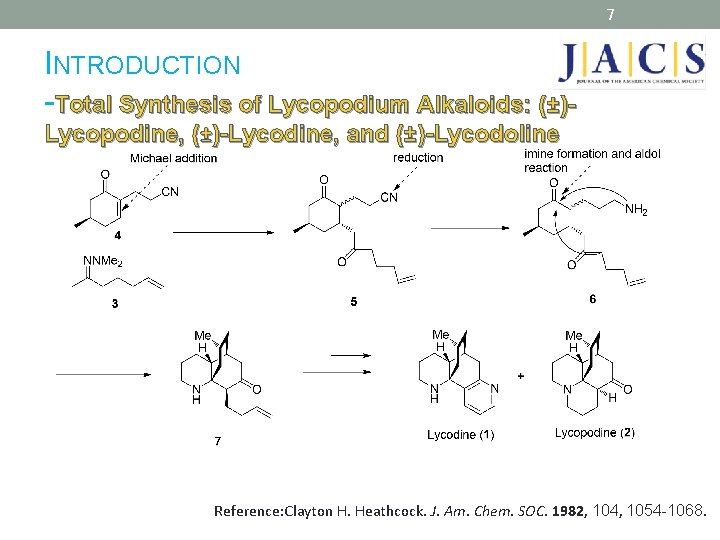
7 INTRODUCTION -Total Synthesis of Lycopodium Alkaloids: (±)Lycopodine, (±)-Lycodine, and (±)-Lycodoline Reference: Clayton H. Heathcock. J. Am. Chem. SOC. 1982, 104, 1054 -1068.
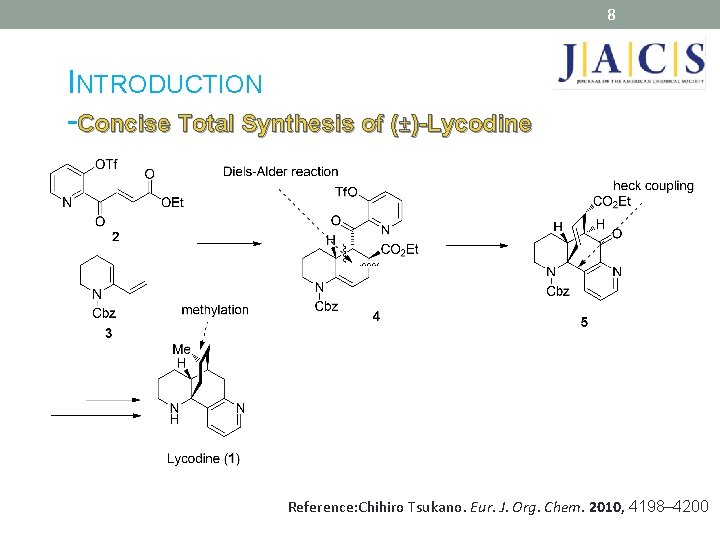
8 INTRODUCTION -Concise Total Synthesis of (±)-Lycodine Reference: Chihiro Tsukano. Eur. J. Org. Chem. 2010, 4198– 4200
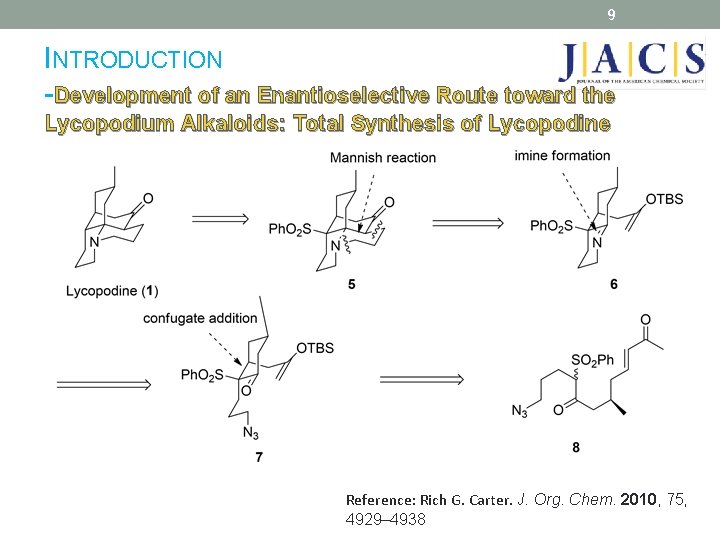
9 INTRODUCTION -Development of an Enantioselective Route toward the Lycopodium Alkaloids: Total Synthesis of Lycopodine Reference: Rich G. Carter. J. Org. Chem. 2010, 75, 4929– 4938

10 Introduction Retrosynthesis of the nature product Procedure of synthesis conclusion

11 Proposed biosynthetic pathway by spender group
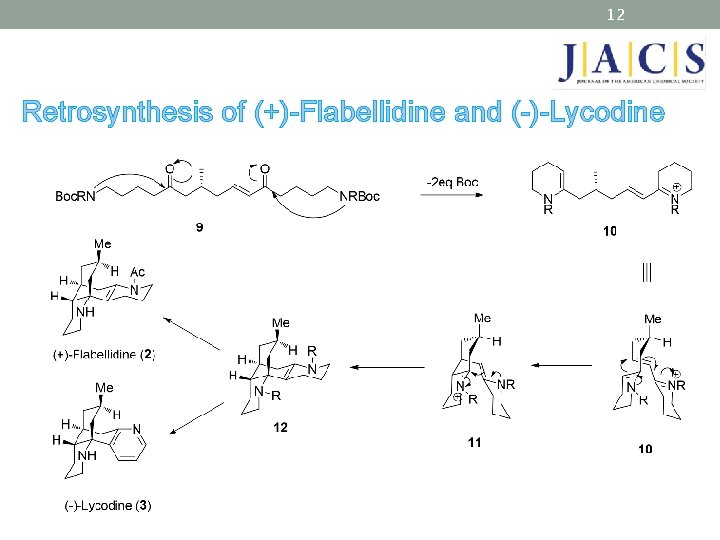
12 Retrosynthesis of (+)-Flabellidine and (-)-Lycodine

13 Introduction Retrosynthesis of the nature product Procedure of synthesis conclusion

14 PROCEDURE OF SYNTHESIS -Synthesis of Linear Substrate 21

15 PROCEDURE OF SYNTHESIS -Total synthesis of (+)-Flabellidine and (-)-Lycodine

16 Introduction Retrosynthesis of the nature product Procedure of synthesis conclusion

17 CONCLUSION the yield of the (+)-flabellidinen is 20. 7% (11 step) the yield of the (–)-lycodine is 14. 6% (11 step) The first asymmetric total synthesis of (+)-flabellidine tetracyclic lycodine skeleton which was inspired by the biosynthetic consideration

18 CONCLUSION Publish date author J. Am. Chem. Soc. XXXX, XXX−XXX Hiromitsu Takayama* Tetrahedron Letters No. 43, pp 4125 4128 Clayton H. Heathcock Eur. J. Org. Chem. 2010, 4198– 4200 Chihiro Tsukano J. AM. CHEM. SOC. 2008, 13778– 13789 Amos B. Smith III* J. AM. CHEM. SOC. Richmond 2008, Sarpong* 7222– 7223 J. Am. Chem. Soc. 2013, 3243− 3247 Tohru Fukuyama step yield production 11 20. 7% (+)-flabellidine 11 14. 6% (–)-lycodine 5 22% (±)-lycodine 5 20. 5% (±)-lycopodine 15 2. 8% (±)-Lycodine 27 2. 2% (+)-lyconadin A 28 2. 2% (-)-lyconadin B 18 10% 13 0. 71% (+)-lyconadin A 14 0. 49% (±)-lyconadin B 11 5. 13% (±)-lyconadin C (±)-Lyconadin A

19 CONCLUSION Publish date author Angew. Chem. Int. Ed. 2014, 3922 – 3925 Mingji Dai Angew. Chem. Int. Ed. 2012, 51, 491 – 495 Xiaoguang Lei* Angew. Chem. Int. Ed. 2011, 50, 8025 – 8028 Hiromitsu Takayama* Angew. Chem. Int. Ed. 2013, 52, 11373 – 11376 J. AM. CHEM. SOC. 2010, 132, 14338– 14339 Yong-Qiang Tu, * Ju¨rgen Ramharter, * step yield production 8 5. 87% lyconadins A 10 4. 8% lyconadins C 12 8% (+)-Fawcettimine 12 16% (+)-Fawcettidine 12 16% (-)-8 -Deoxyserratinine 19 16. 4% Huperzine-Q 12 5% (-)-Lycojaponicumin C, 10 2. 0% (-)-8 -Deoxyserratinine 10 1. 8% (+)-Fawcettimine 10 1. 6% (+)-Fawcettidine 8 15% (+)-Lycoflexine

20 REFERENCE 1. J. AM. CHEM. SOC. 2008, 130, 13778– 13789 2. J. AM. CHEM. SOC. 2008, 130, 7222– 7223 3. J. AM. CHEM. SOC. 2013, 135, 3243– 3247 4. Angew. Chem. Int. Ed. 2012, 51, 491 – 495 5. Angew. Chem. Int. Ed. 2013, 52, 11373 – 11376 6. Angew. Chem. Int. Ed. 2011, 50, 8025 – 8028 7. J. Org. Chem. 2010, 75, 4929– 4938 8. Angew. Chem. Int. Ed. 2014, 53, 3922 – 3925 9. J. Am. Chem. SOC. 1982, 104, 1054 -1068 10. Eur. J. Org. Chem. 2010, 4198– 4200
THE END 21

PROBLEM 22
- Slides: 22