1 Atoms John Dalton 1766 1844 established a

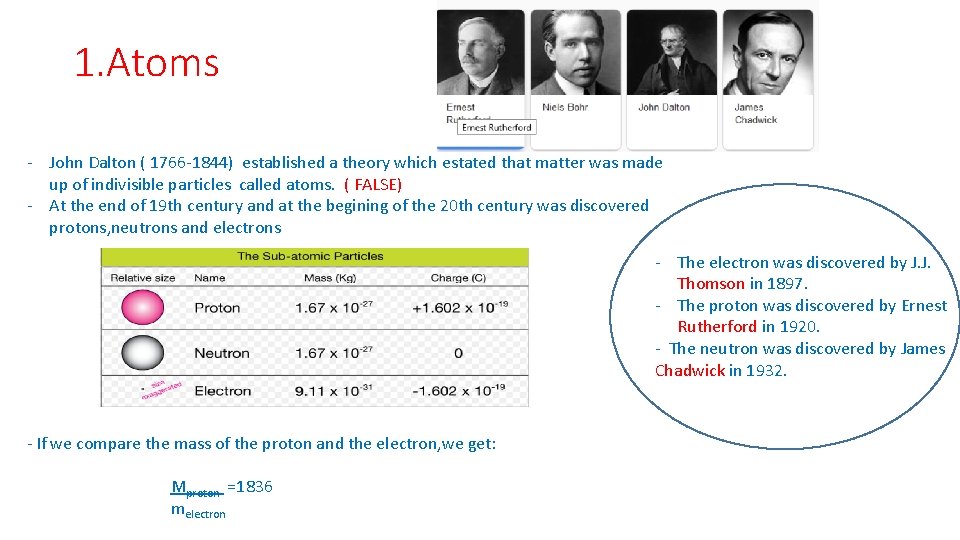
1. Atoms - John Dalton ( 1766 -1844) established a theory which estated that matter was made up of indivisible particles called atoms. ( FALSE) - At the end of 19 th century and at the begining of the 20 th century was discovered protons, neutrons and electrons - The electron was discovered by J. J. Thomson in 1897. - The proton was discovered by Ernest Rutherford in 1920. - The neutron was discovered by James Chadwick in 1932. - If we compare the mass of the proton and the electron, we get: Mproton =1836 melectron

1. Atoms - A unit of atomic mass has been defined by the iupac ( international unión of pure and applied chemistry) as being equivalent to 1. 66 10 -27 kg. 1 u=1. 66 10 -27 kg. - The unit of electric charge in the international system of units is the coulomb ( C) - We now know that atoms also contains other smaller particles called quarks, which form protons and neutrons. QUARKS

2. Atomic models 2. 1 Thomson´s atomic model INCORRECT https: //www. youtube. com/watch? v=X 2 uvu. SThtu. I
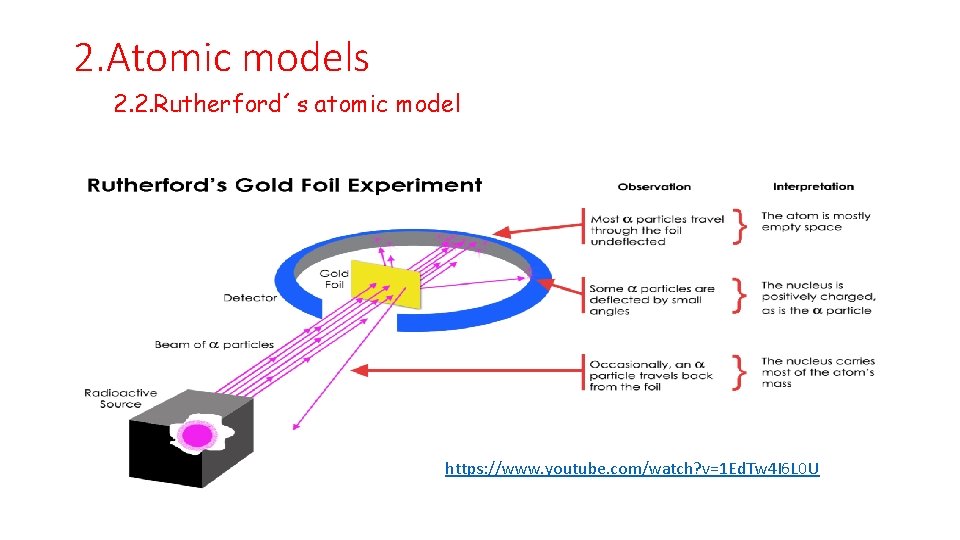
2. Atomic models 2. 2. Rutherford´s atomic model https: //www. youtube. com/watch? v=1 Ed. Tw 4 I 6 L 0 U

2. Atomic models 2. 2. Rutherford´s atomic model https: //www. youtube. com/watch? v=B-k_k. Mw. B 1 z. M
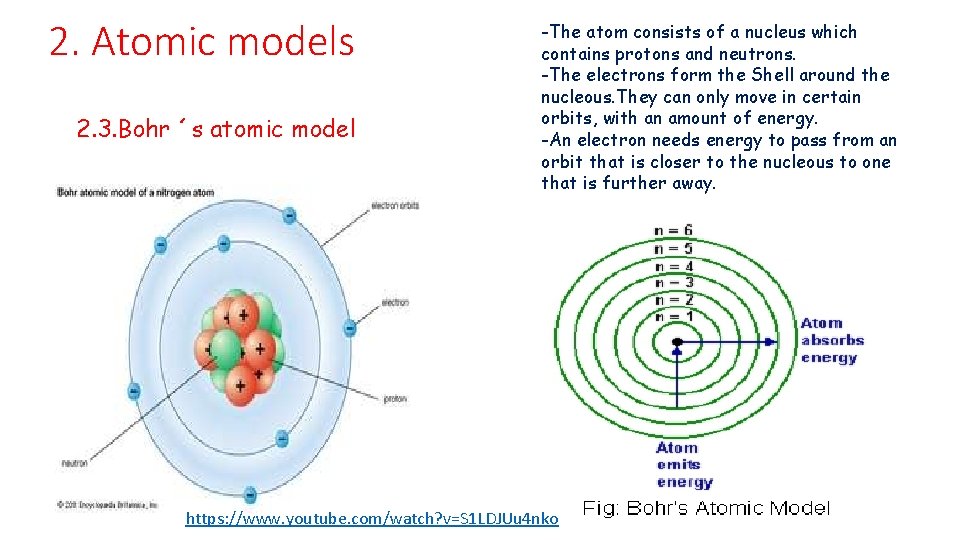
2. Atomic models 2. 3. Bohr ´s atomic model -The atom consists of a nucleus which contains protons and neutrons. -The electrons form the Shell around the nucleous. They can only move in certain orbits, with an amount of energy. -An electron needs energy to pass from an orbit that is closer to the nucleous to one that is further away. https: //www. youtube. com/watch? v=S 1 LDJUu 4 nko

2. Atomic models 2. 3. Bohr ´s atomic model. The quantises atom • Atoms are quantised • We also know how many electrons there can be in each layer • • In the first layer there can be up to 2 electrons. In the second layer there can be up to 8 electrons In the third layer there can be up to 18 electrons In the fourth layer there can be up to 32 electrons
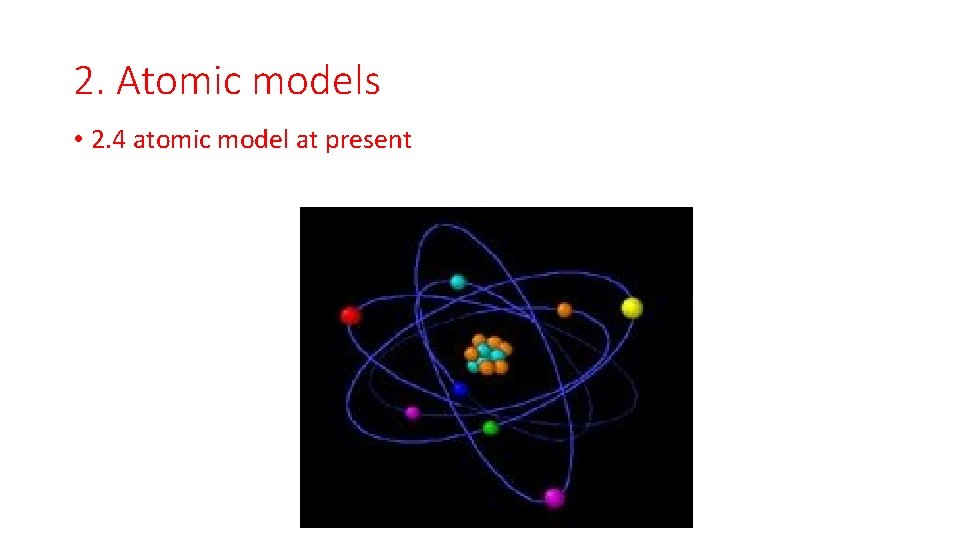
2. Atomic models • 2. 4 atomic model at present
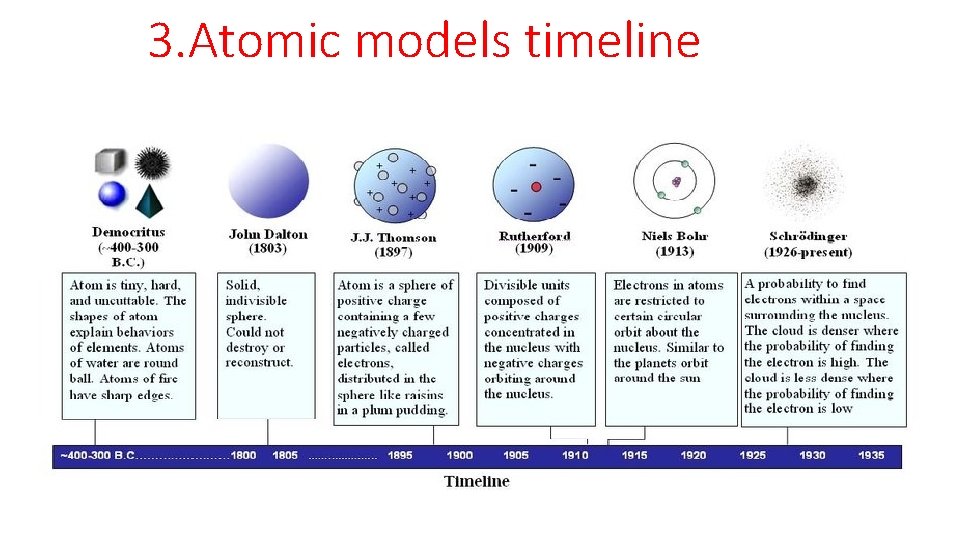
3. Atomic models timeline

4. atoms, isotopes and ions 4. 1. atoms • An atom is the smallest recognizable part of an element containing protons (positively charged particles), neutrons (neutral particles), and electrons (negatively charged particles). • Atoms are electrically neutral because they are composed of an equal number of protons and electrons • We represented atoms with a simbols and two numbers: • A is the mass number: number of the protons plus the number of neutrons. • Z is the atomic number: number of protons. • X is the symbol represents the first letter of the element´s name in latin. https: //www. youtube. com/watch? v=_S 7 ov 25 y 3_M

4. atoms, isotopes and ions 4. 2. isotopes https: //www. youtube. com/watch? v=Eas. Eso 5 -tj 8

4. atoms, isotopes and ions 4. 3 the atomic mass of chemical elements • The atomic mass of an isotope is the sum of the mass of the particles that form it • The atomic mass of a chemical element is the average mass of one of the element´s atoms, taking into account all its isotopes.

4. atoms, isotopes and ions 4. 3 the atomic mass of chemical elements

4. atoms, isotopes and ions 4. 4 ions • When an atom loses electrons it gains a positive charge and becomes a positive ion or a catión • When an atom gains electrons, it acquires a negative charge and becomes a negative ion or an anion.
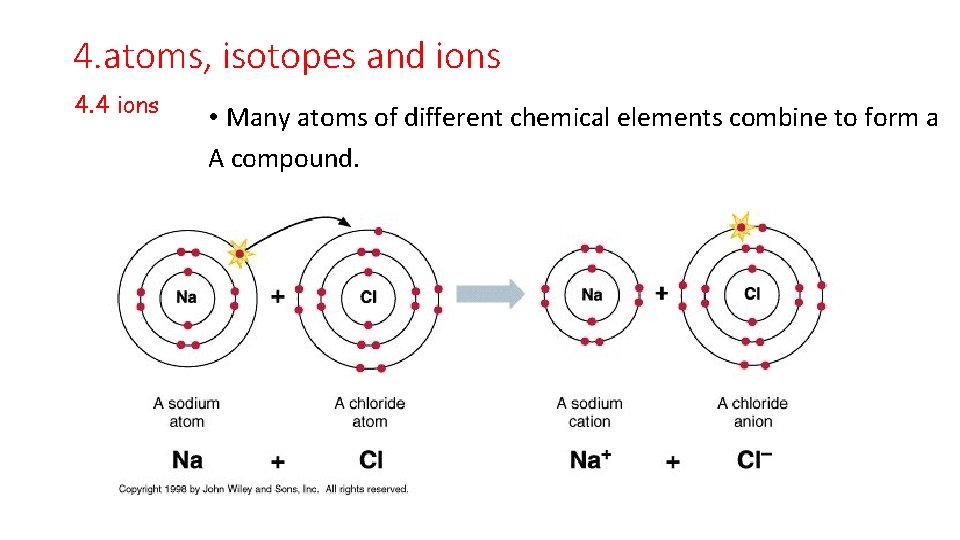
4. atoms, isotopes and ions 4. 4 ions • Many atoms of different chemical elements combine to form a A compound.

5. Radiactivity 5. 1. Radioactive emissions • Radioactivity refers to the particles which are emitted from nucleus as a result of nuclear instability.
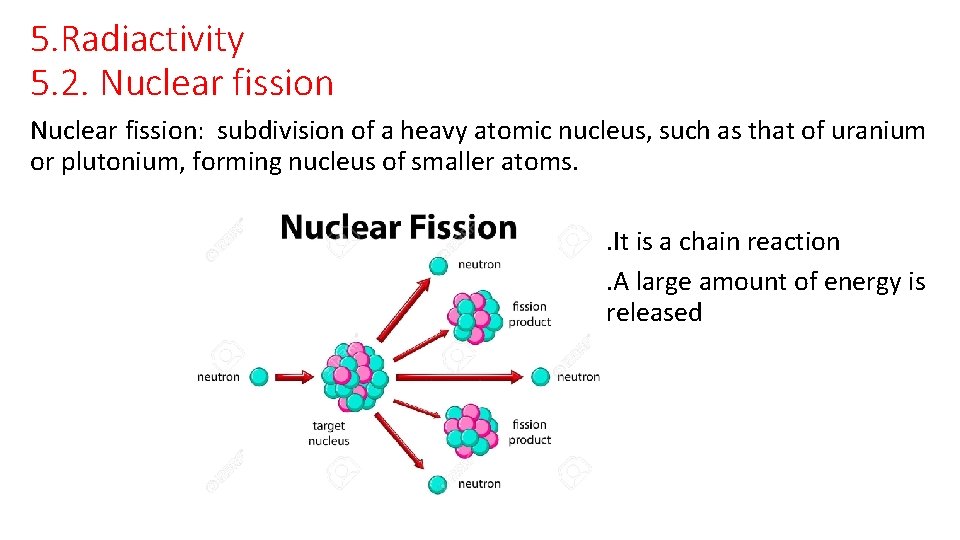
5. Radiactivity 5. 2. Nuclear fission: subdivision of a heavy atomic nucleus, such as that of uranium or plutonium, forming nucleus of smaller atoms. . It is a chain reaction. A large amount of energy is released
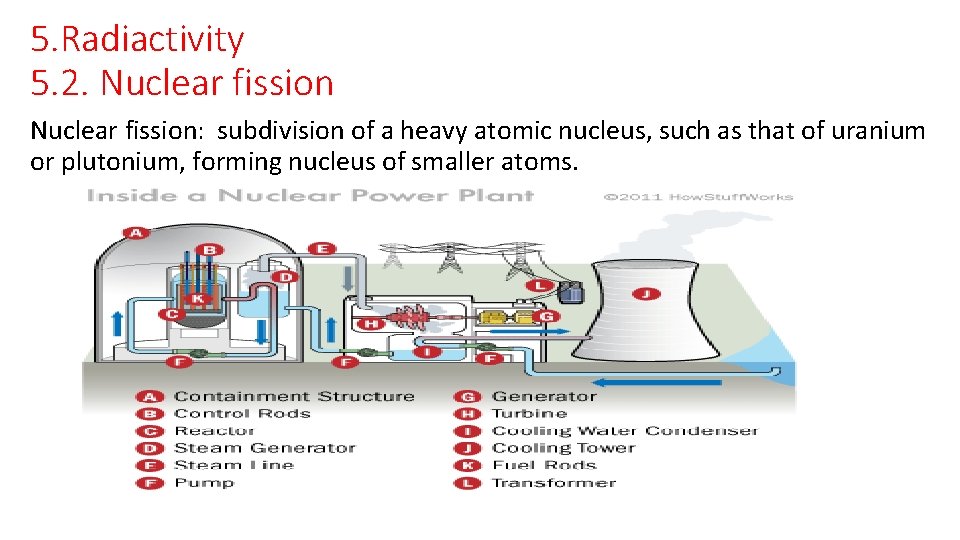
5. Radiactivity 5. 2. Nuclear fission: subdivision of a heavy atomic nucleus, such as that of uranium or plutonium, forming nucleus of smaller atoms.
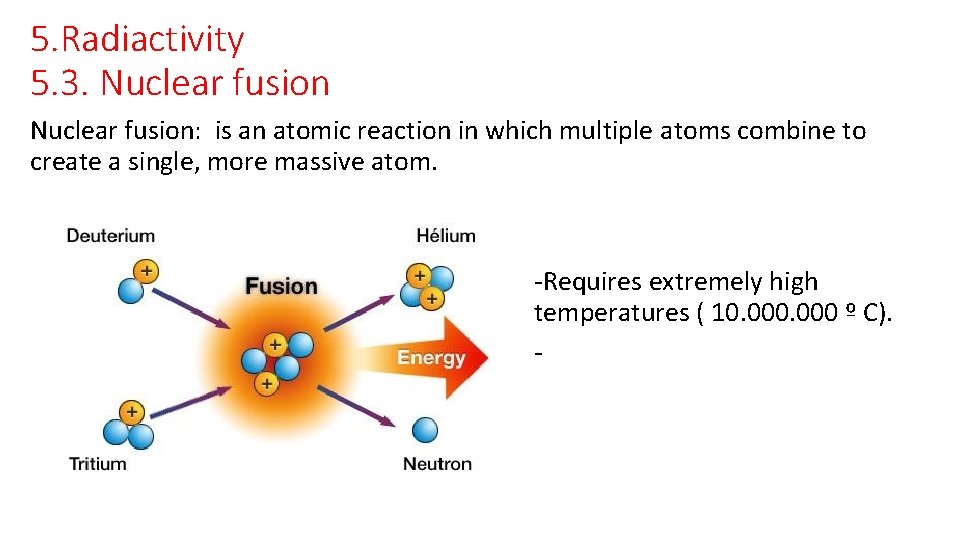
5. Radiactivity 5. 3. Nuclear fusion: is an atomic reaction in which multiple atoms combine to create a single, more massive atom. -Requires extremely high temperatures ( 10. 000 º C). -
5. Applications of Radiactivity 5. 1. In Medicine: Radioisotopes have found extensive use in diagnosis and therapy, and this has given rise to a rapidly growing field called nuclear medicine. These radioactive isotopes have proven particularly effective as tracers in certain diagnostic procedures. Such radioisotopes as cobalt-60 and cesium-137 are widely used to treat cancer.

5. Applications of Radiactivity 5. 2. In industry: It is a power generation based on the release of the fission energy of uranium (see nuclear fission; nuclear reactor: Nuclear fission reactors). Other applications include the use of radioisotopes to measure (and control) the thickness or density of metal and plastic sheets, to stimulate the cross-linking of polymers, to induce mutations in plants in order to develop hardier specie…

5. Applications of Radiactivity 5. 3. In science: A special application of this type of radioactivity age method, carbon-14 dating, has proved especially useful to physical anthropologists and archaeologists. It has helped them to better determine the chronological sequence of past events by enabling them to date more accurately fossils and artifacts from 500 to 50, 000 years old.

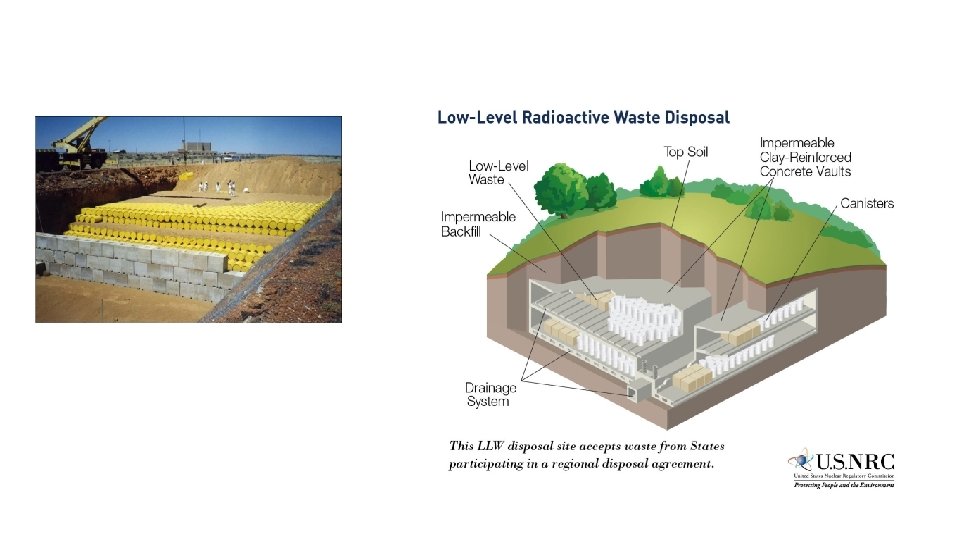
6. Applications of Radiactivity There are many industries like mining, defense, medicine, scientific research, nuclear power generation which produce by-products that include radioactive waste. The radioactive waste can remain radioactive for few months, years or even hundreds of years and the level of radioactivity can vary. -Low-level waste (LLW) includes radioactively contaminated protective clothing, tools, filters, rags, medical tubes, and many other items -Waste incidental to reprocessing (WIR) refers to certain waste byproducts that result from reprocessing spent nuclear fuel, which the U. S. Department of Energy (DOE) has distinguished from high-level waste (described below) -High-level waste (HLW) is "irradiated" or used nuclear reactor fuel -Uranium mill tailings are the residues remaining after the processing of natural ore to extract uranium and thorium
- Slides: 25