1 anomeric carbon 2 3 Relationship between Haworth
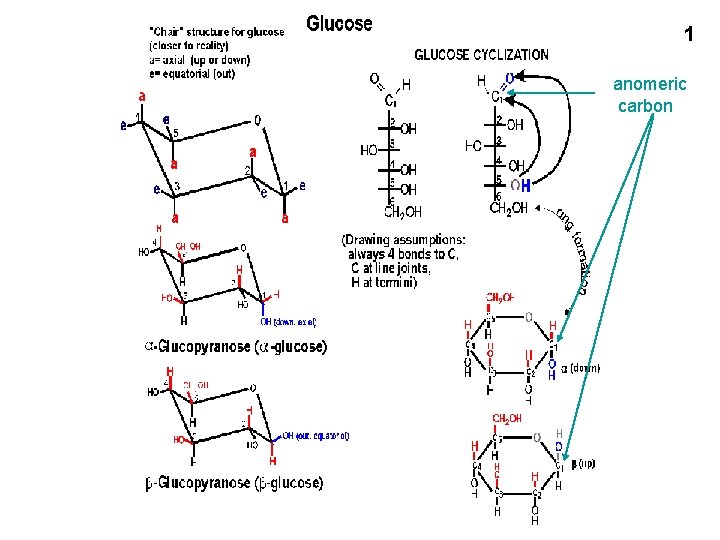
1 anomeric carbon
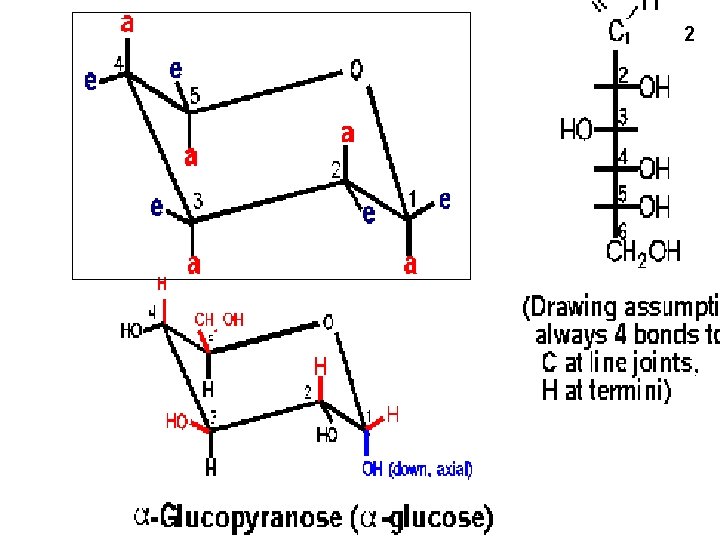
2
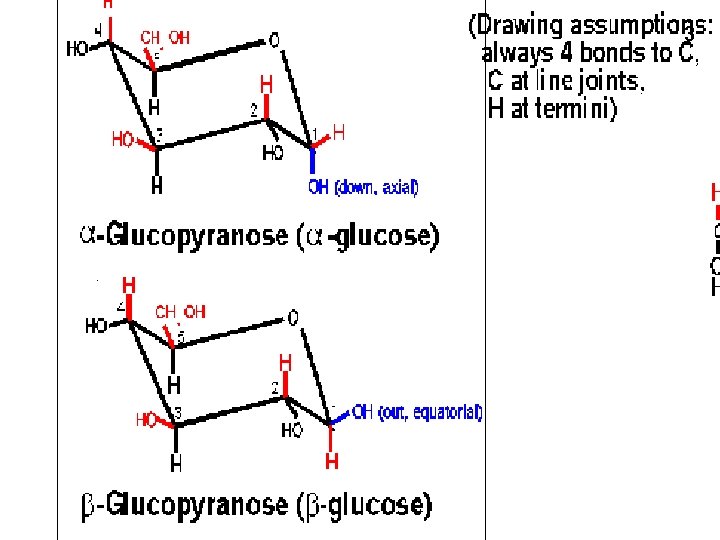
3
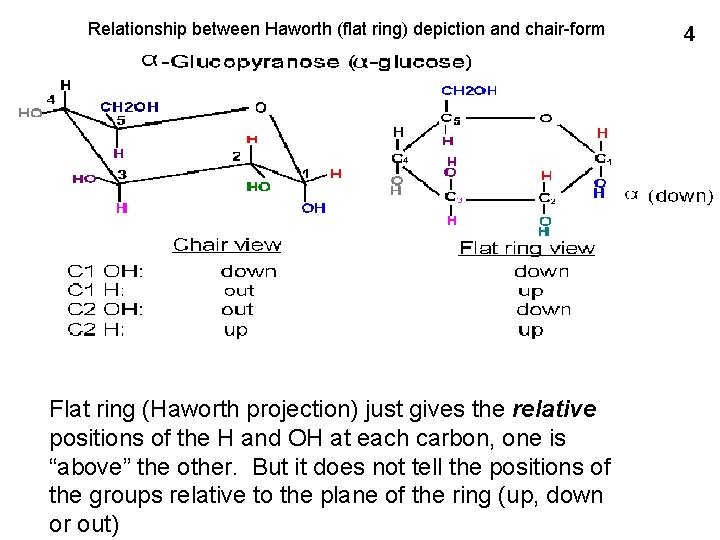
Relationship between Haworth (flat ring) depiction and chair-form Flat ring (Haworth projection) just gives the relative positions of the H and OH at each carbon, one is “above” the other. But it does not tell the positions of the groups relative to the plane of the ring (up, down or out) 4
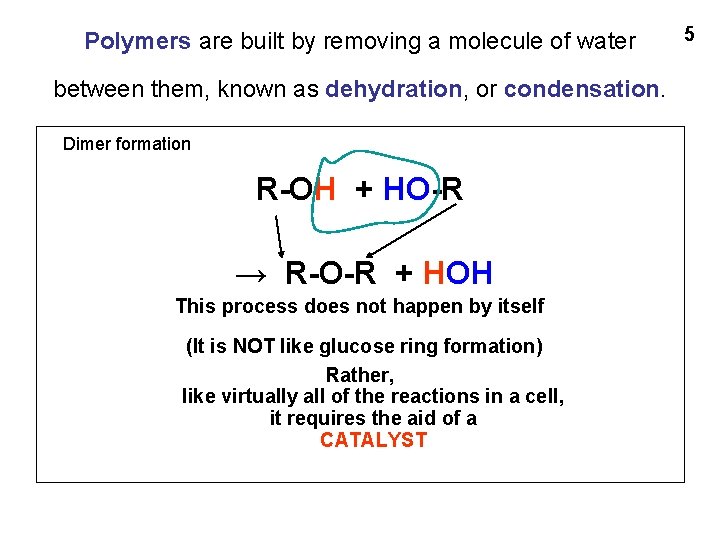
Polymers are built by removing a molecule of water between them, known as dehydration, or condensation. Dimer formation R-OH + HO-R → R-O-R + HOH This process does not happen by itself (It is NOT like glucose ring formation) Rather, like virtually all of the reactions in a cell, it requires the aid of a CATALYST 5

AND: Polymers are broken down by the reverse process, 6 ADDING a molecule of water between them, known as DIMER HYDROLYSIS R-O-R + HOH→ R-OH + HO-R This process does not happen by itself Rather, like virtually all of the reaction in a cell, it requires the aid of a CATALYST
7 Building a polymer from glucose
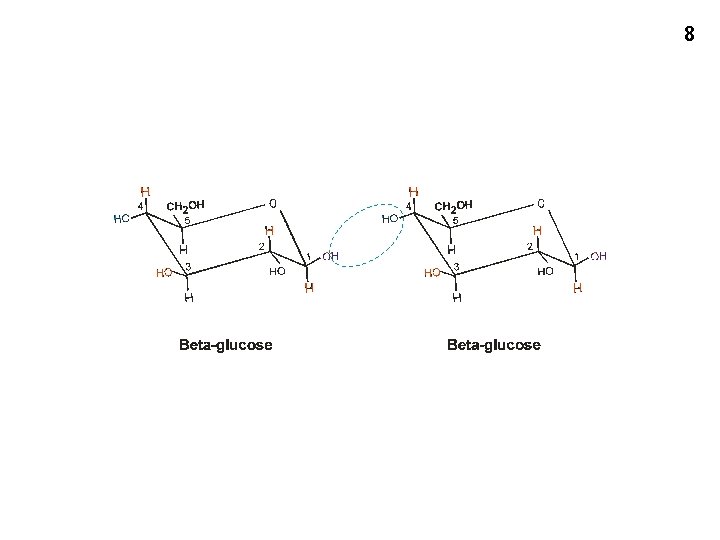
8
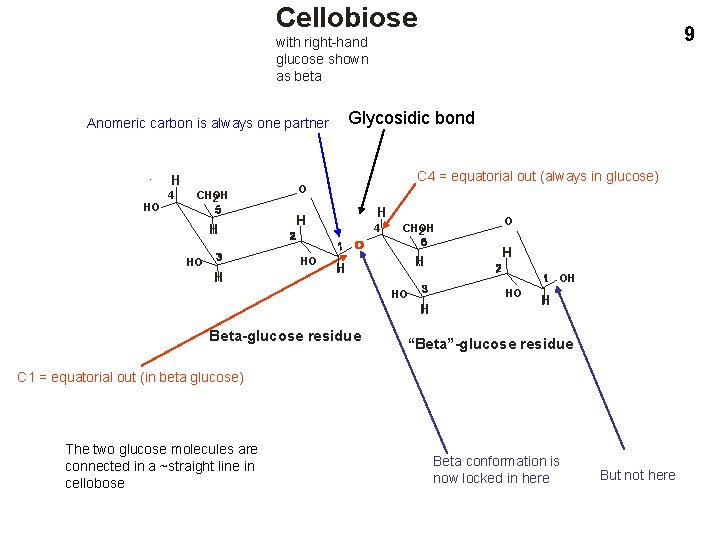
Cellobiose 9 with right-hand glucose shown as beta Glycosidic bond Anomeric carbon is always one partner H HO 4 CHOH 2 H H HO HO C 4 = equatorial out (always in glucose) O 4 CHOH 2 H H H OH HO HO H Beta-glucose residue O H “Beta”-glucose residue C 1 = equatorial out (in beta glucose) The two glucose molecules are connected in a ~straight line in cellobose Beta conformation is now locked in here But not here
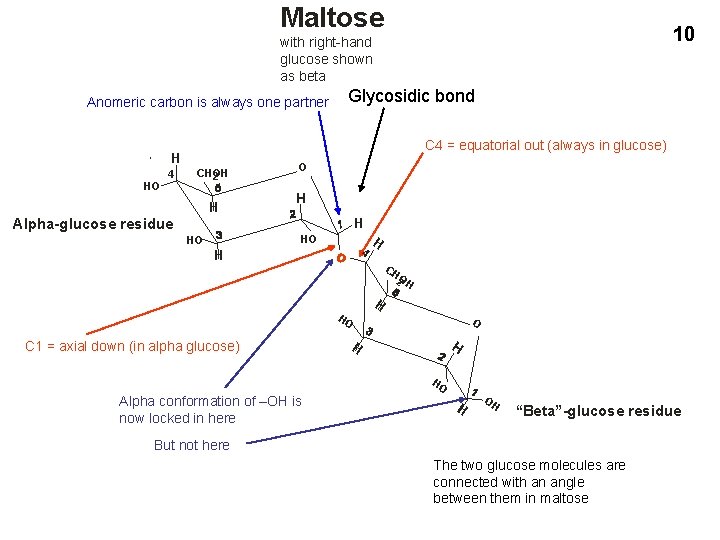
Maltose 10 with right-hand glucose shown as beta Anomeric carbon is always one partner C 4 = equatorial out (always in glucose) H HO 4 Glycosidic bond CHOH 2 H O H H Alpha-glucose residue HO HO 4 H H CH 2 OH H HO C 1 = axial down (in alpha glucose) O H H HO Alpha conformation of –OH is now locked in here H OH “Beta”-glucose residue But not here The two glucose molecules are connected with an angle between them in maltose
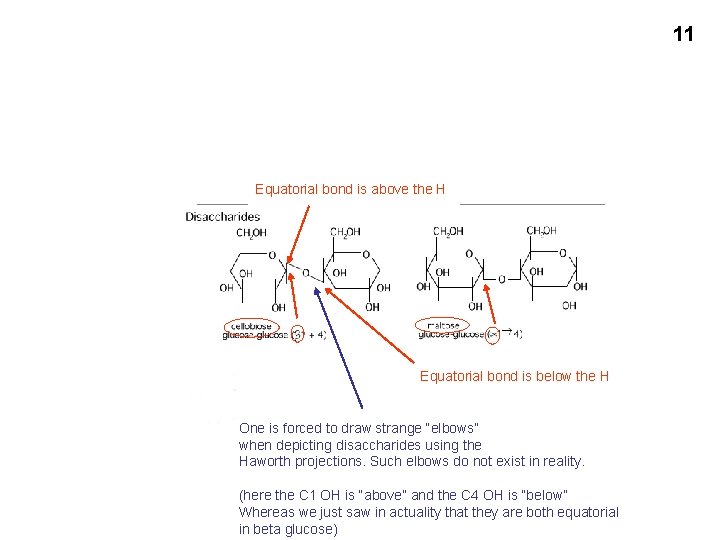
11 Equatorial bond is above the H Equatorial bond is below the H One is forced to draw strange “elbows” when depicting disaccharides using the Haworth projections. Such elbows do not exist in reality. (here the C 1 OH is “above” and the C 4 OH is “below” Whereas we just saw in actuality that they are both equatorial in beta glucose)
12 down t ou H H Cellulose Starch or glycogen chain Tinker toys
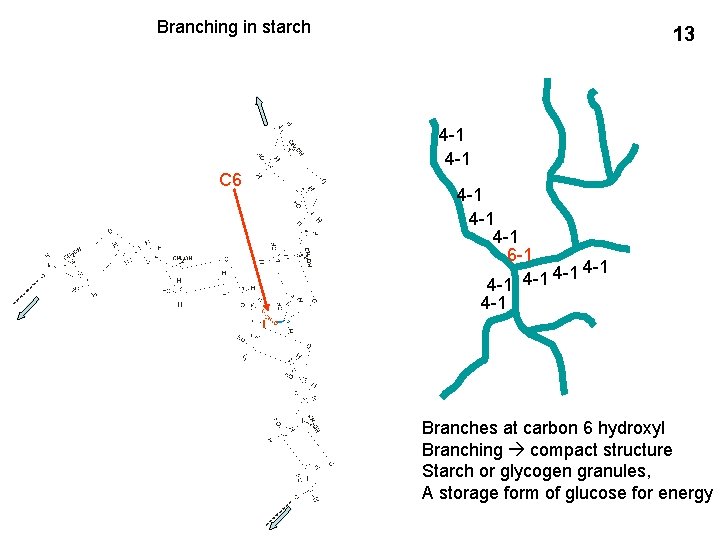
Branching in starch 13 4 -1 C 6 4 -1 4 -1 6 -1 4 -1 4 -1 4 -1 Branches at carbon 6 hydroxyl Branching compact structure Starch or glycogen granules, A storage form of glucose for energy
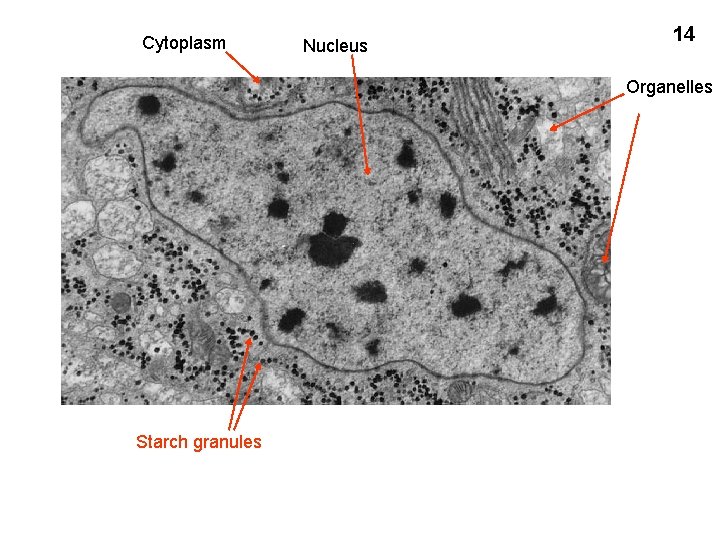
Cytoplasm Nucleus 14 Organelles Starch granules
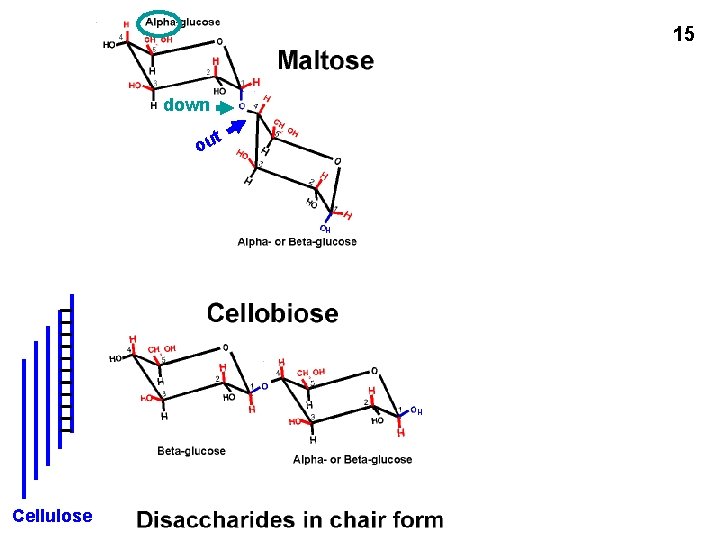
15 down t ou H H Cellulose or glycogen chain
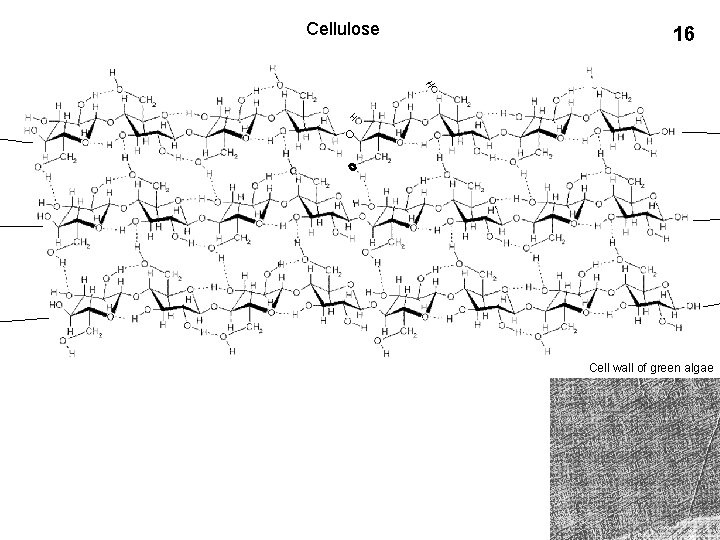
Cellulose 16 Cell wall of green algae
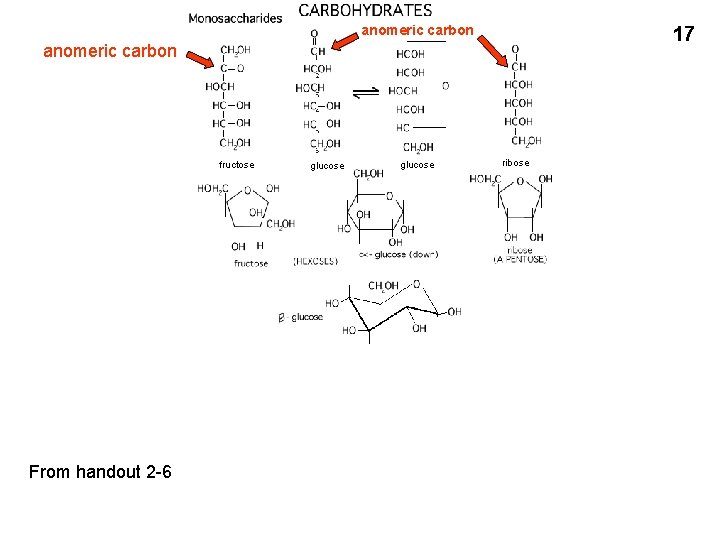
anomeric carbon 17 anomeric carbon fructose From handout 2 -6 glucose ribose
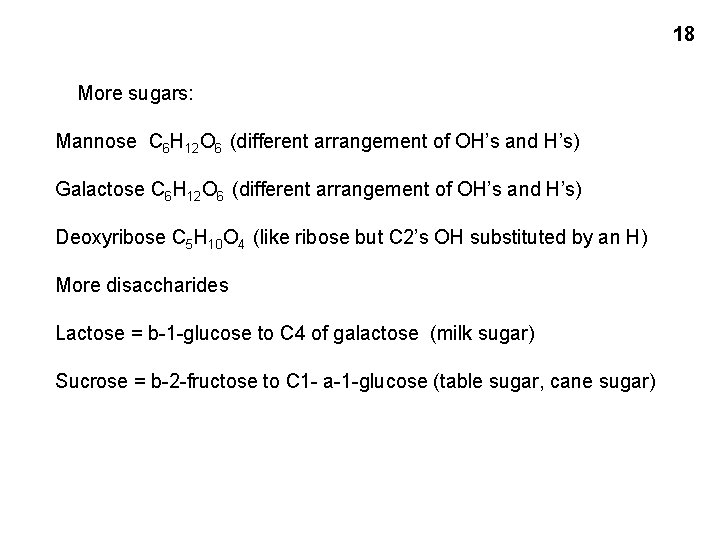
18 More sugars: Mannose C 6 H 12 O 6 (different arrangement of OH’s and H’s) Galactose C 6 H 12 O 6 (different arrangement of OH’s and H’s) Deoxyribose C 5 H 10 O 4 (like ribose but C 2’s OH substituted by an H) More disaccharides Lactose = b-1 -glucose to C 4 of galactose (milk sugar) Sucrose = b-2 -fructose to C 1 - a-1 -glucose (table sugar, cane sugar)
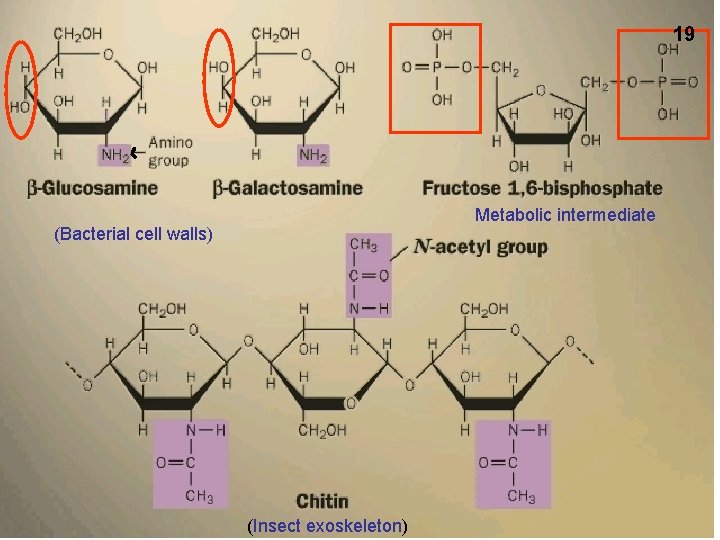
19 Metabolic intermediate (Bacterial cell walls) (Insect exoskeleton)

Lipids • Soluble in organic solvents (like octane, a hydrocarbon) • Heterogeneous class of structures • Not very polymer-like (in terms of covalently bonded structures) 20
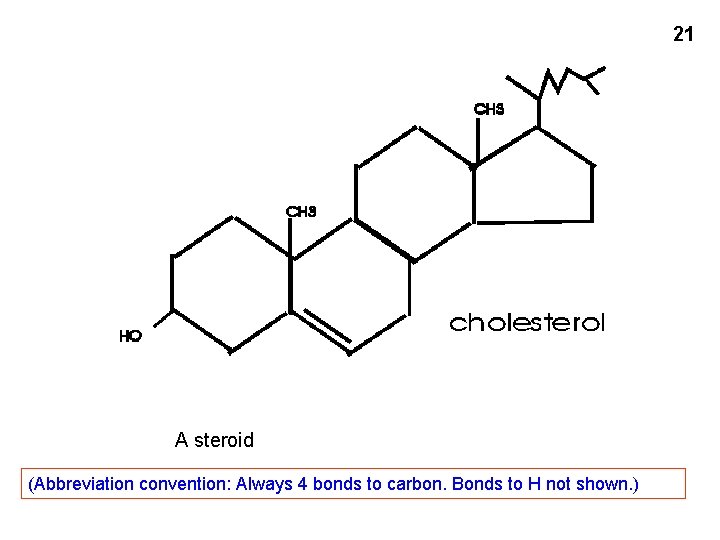
21 A steroid (Abbreviation convention: Always 4 bonds to carbon. Bonds to H not shown. )
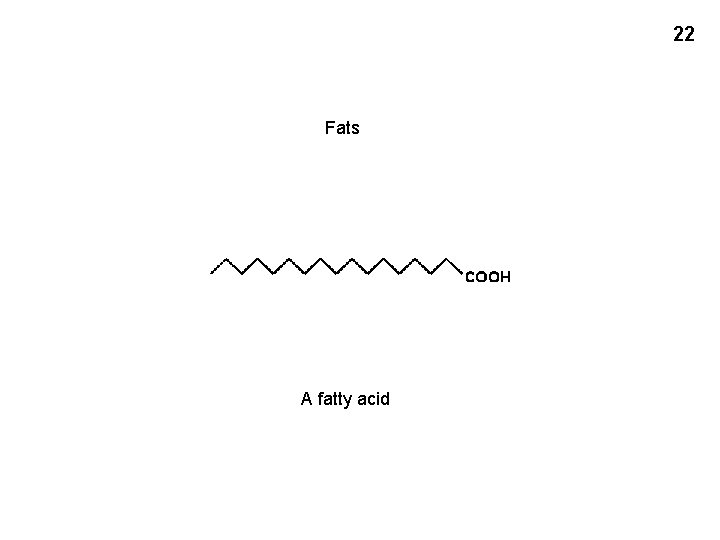
22 Fats A fatty acid
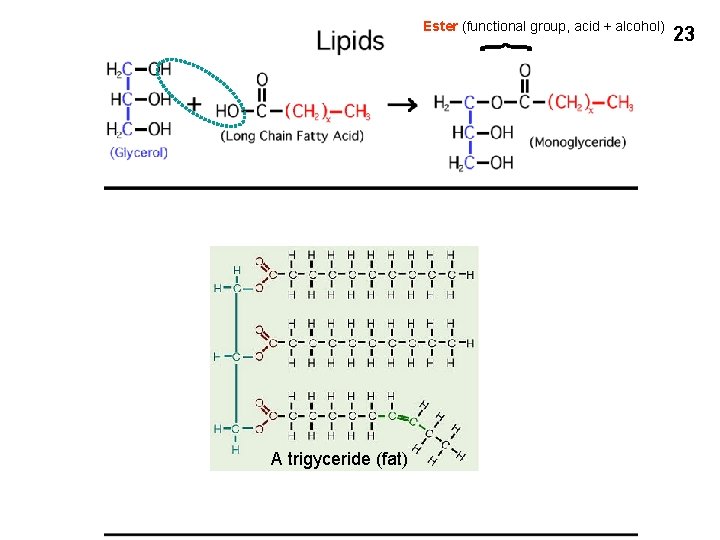
Ester (functional group, acid + alcohol) A trigyceride (fat) 23
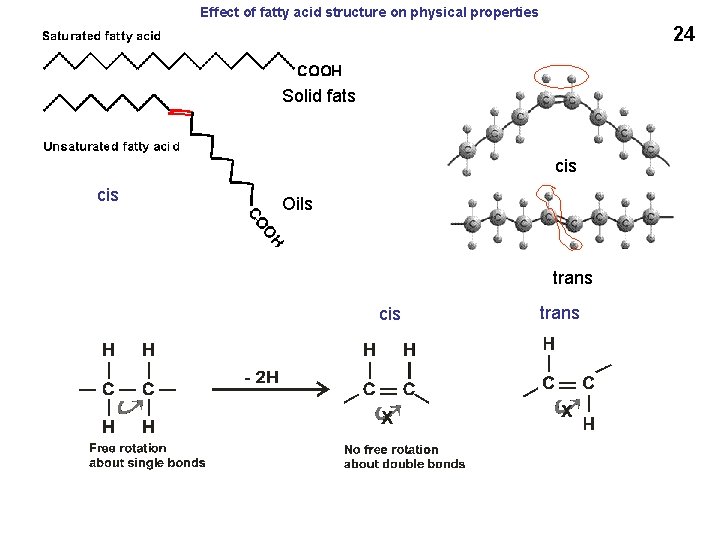
Effect of fatty acid structure on physical properties 24 Solid fats cis Oils trans cis trans
25 Adipocyte (fat storage cell) Nucleus Fat globule
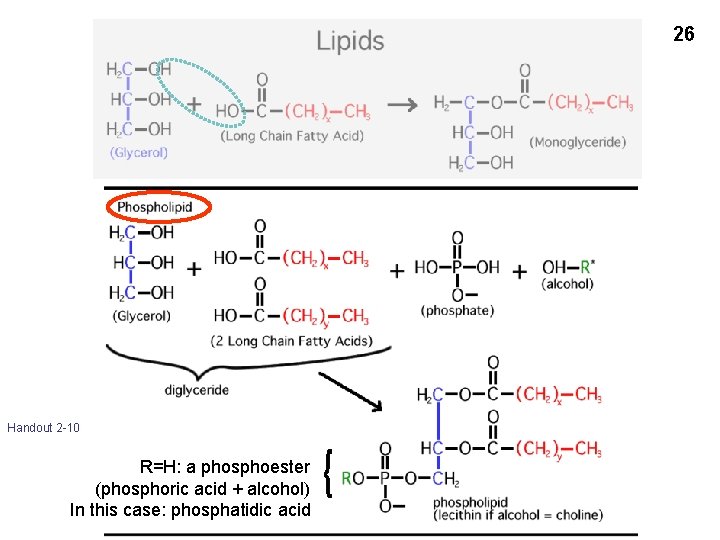
26 Handout 2 -10 R=H: a phosphoester (phosphoric acid + alcohol) In this case: phosphatidic acid
![27 [HO] Handout 2 -10 27 [HO] Handout 2 -10](http://slidetodoc.com/presentation_image_h/917b64a9a54e72dcb898f41a9e7d4c24/image-27.jpg)
27 [HO] Handout 2 -10
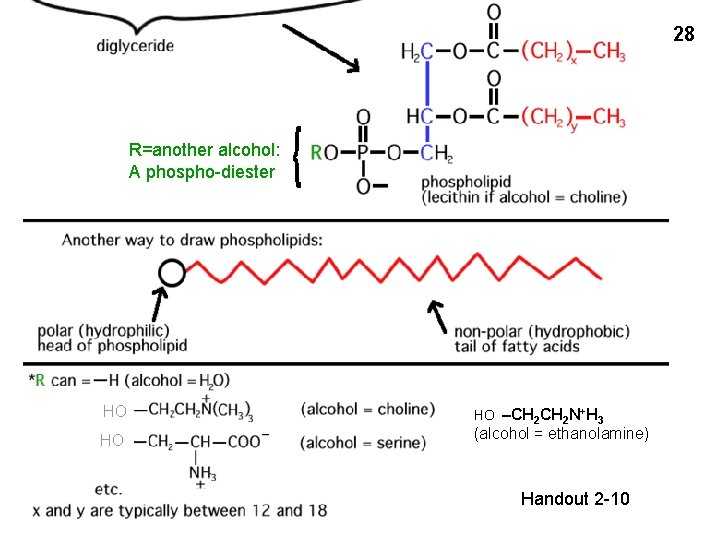
28 R=another alcohol: A phospho-diester HO HO HO –CH 2 N+H 3 (alcohol = ethanolamine) Handout 2 -10
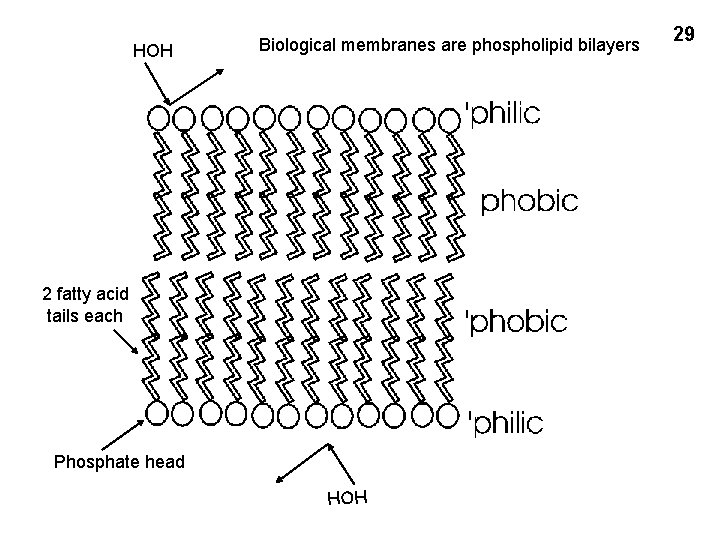
HOH Biological membranes are phospholipid bilayers 2 fatty acid tails each Phosphate head 29 HOH

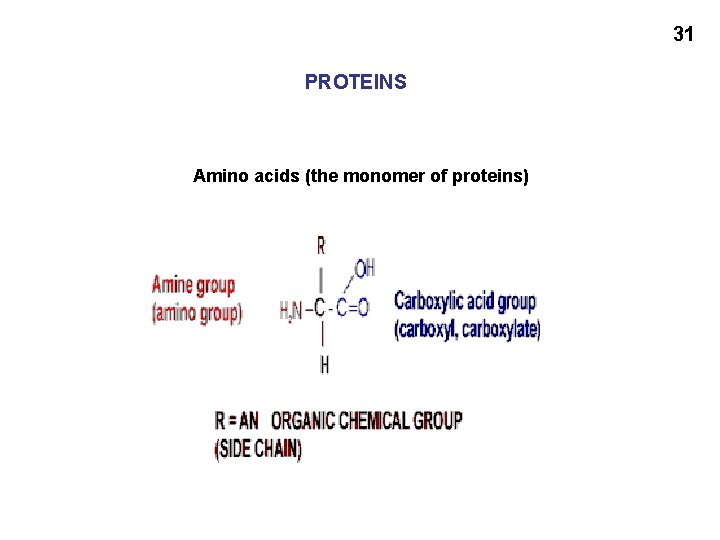
30 Incidentally, note the functional groups we have met so far: Hydroxyl Amine Amide Carboxyl Carbonyl Aldehyde Ketone Ester: Carboxylic acid ester Phosphoester And: Glycosidic bonds C=C double bonds (cis and trans)
31 PROTEINS Amino acids (the monomer of proteins)
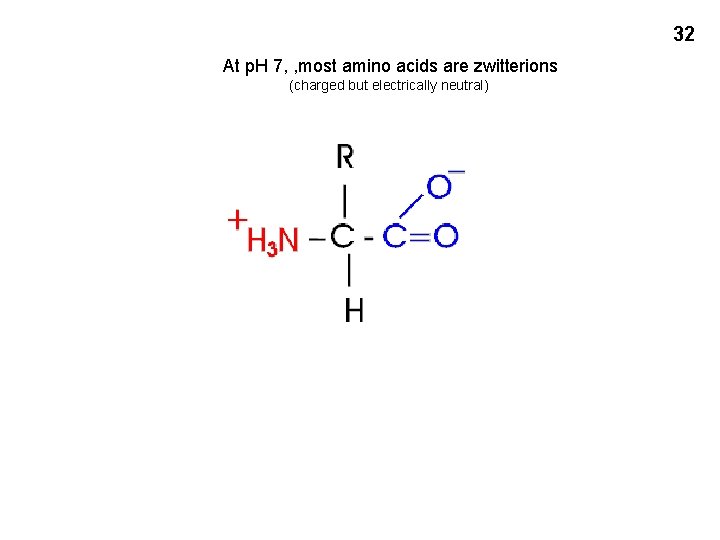
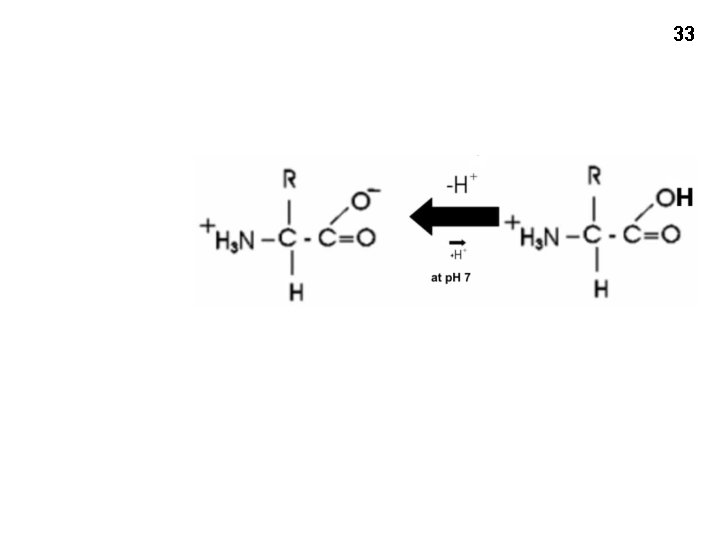
32 At p. H 7, , most amino acids are zwitterions (charged but electrically neutral)
33
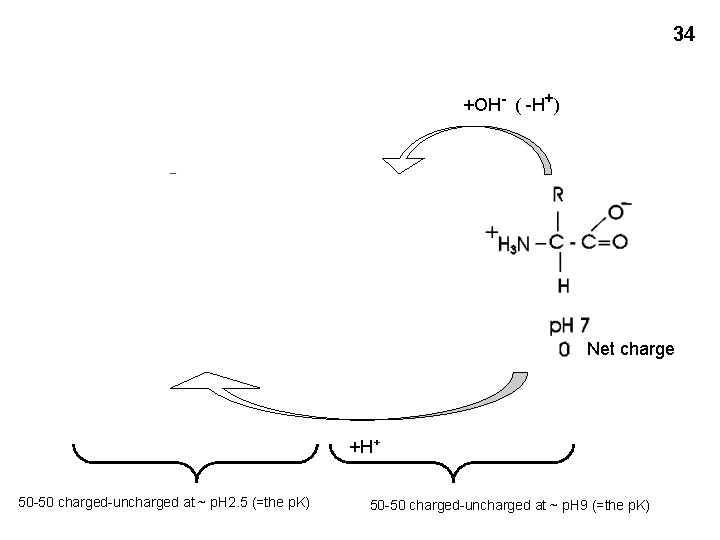
34 +OH- ( -H+) Net charge +H+ 50 -50 charged-uncharged at ~ p. H 2. 5 (=the p. K) 50 -50 charged-uncharged at ~ p. H 9 (=the p. K)
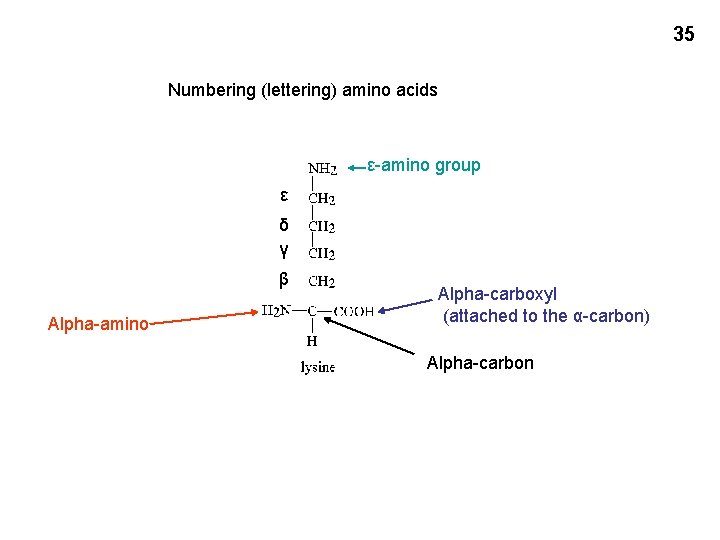
35 Numbering (lettering) amino acids ε-amino group ε δ γ β Alpha-amino Alpha-carboxyl (attached to the α-carbon) Alpha-carbon
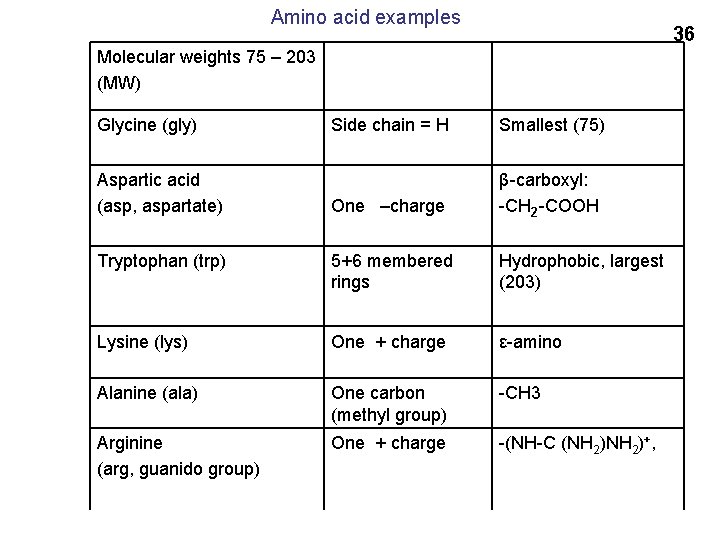
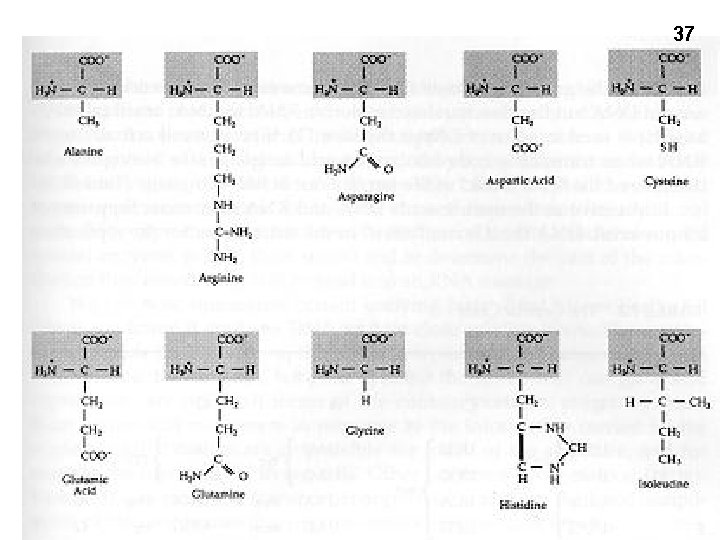
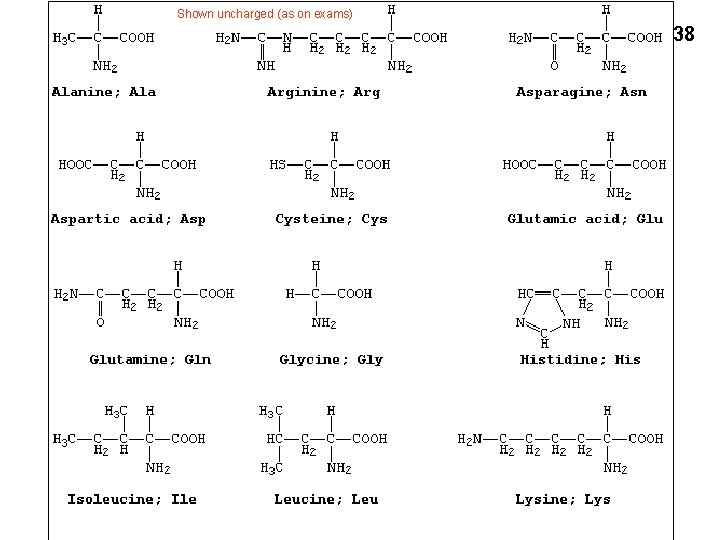
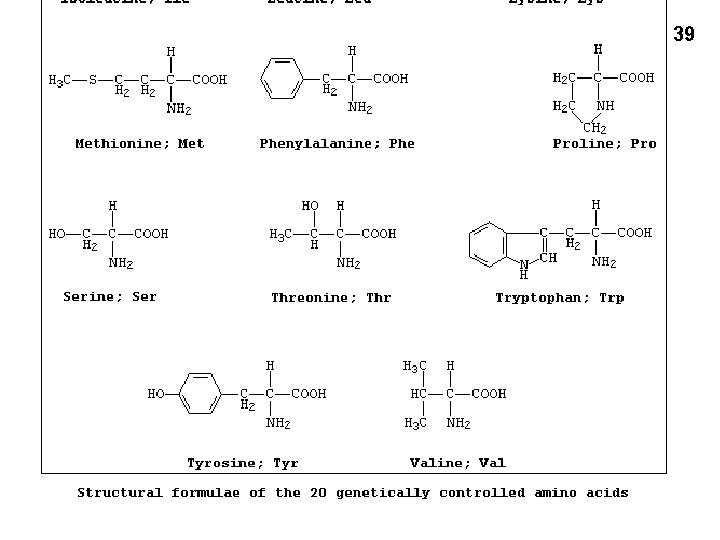
Amino acid examples 36 Molecular weights 75 – 203 (MW) Glycine (gly) Side chain = H Smallest (75) One –charge β-carboxyl: -CH 2 -COOH Tryptophan (trp) 5+6 membered rings Hydrophobic, largest (203) Lysine (lys) One + charge ε-amino Alanine (ala) One carbon (methyl group) -CH 3 Arginine (arg, guanido group) One + charge -(NH-C (NH 2)+, Aspartic acid (asp, aspartate)
37
Shown uncharged (as on exams) 38
39
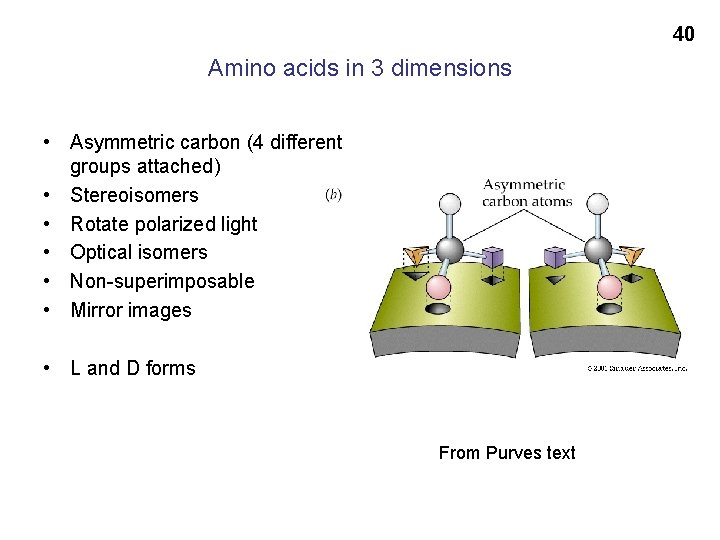
40 Amino acids in 3 dimensions • Asymmetric carbon (4 different groups attached) • Stereoisomers • Rotate polarized light • Optical isomers • Non-superimposable • Mirror images • L and D forms From Purves text
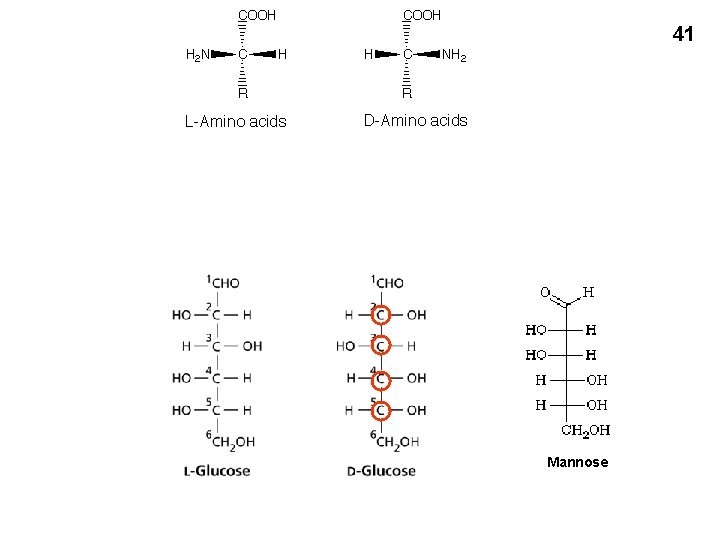
41 Mannose
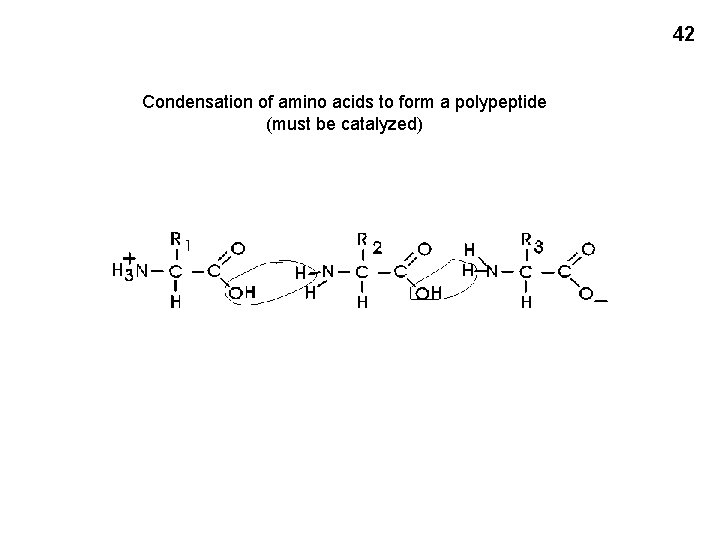
42 Condensation of amino acids to form a polypeptide (must be catalyzed)
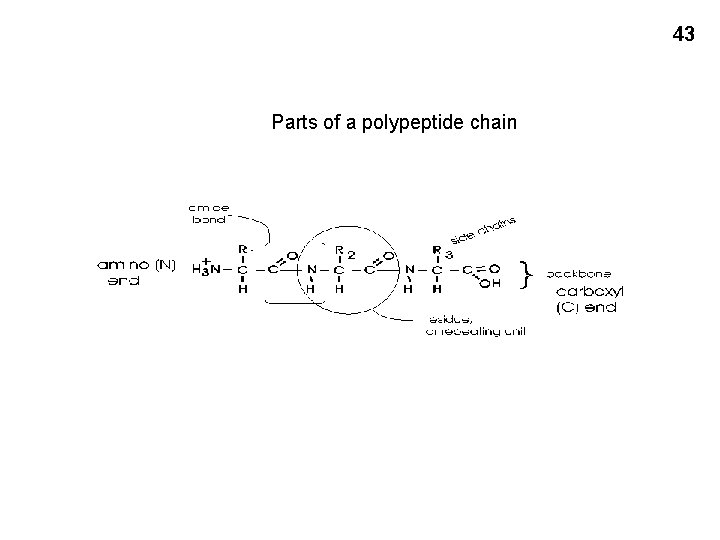
43 Parts of a polypeptide chain
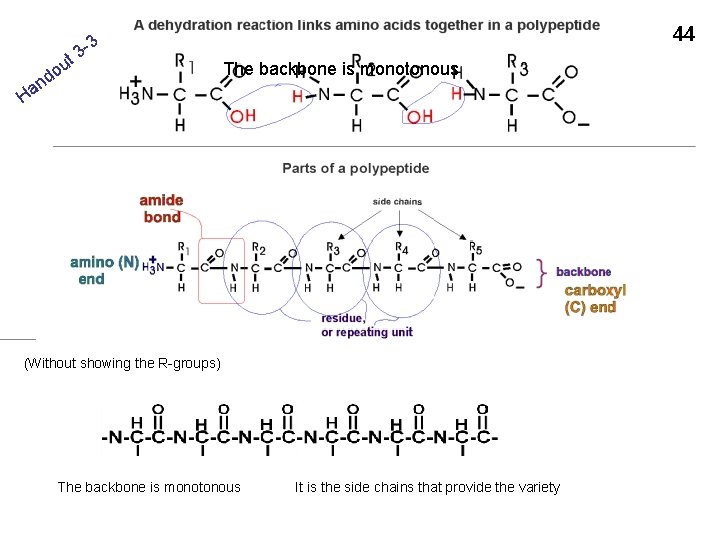
t n a H u do 3 3 - 44 The backbone is monotonous (Without showing the R-groups) The backbone is monotonous It is the side chains that provide the variety

45 “Polypeptides” vs. “proteins” • Polypeptide = amino acids connected in a linear chain (polymer) • Protein = a polypeptide or several associated polypeptides (discussed later) • Often used synonymously • Peptide (as opposed to polypeptide) is smaller, even 2 AAs (dipeptide)
- Slides: 45