1 ANEMIA 3 Anemia is a frequent laboratory

1


ANEMIA 3 Anemia is a frequent laboratory abnormality in children. 20 % of children in the United States and 80 % of children in developing countries will be anemic at some point by the age of 18 years

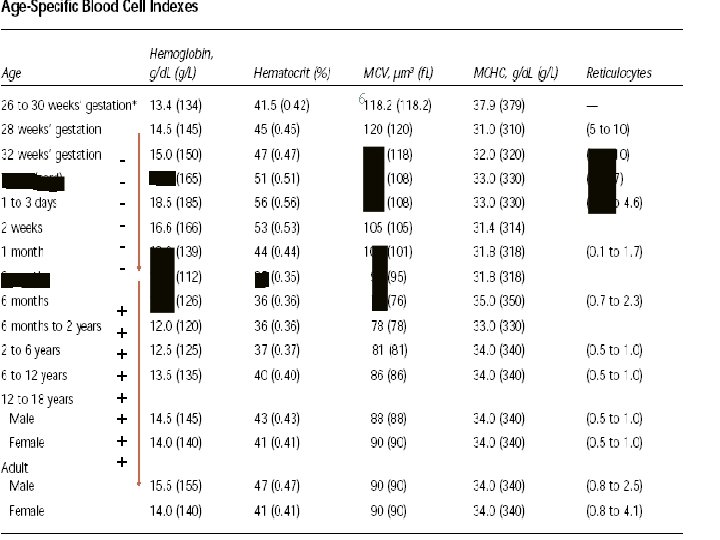
ANEMIA 4 Anemia is defined as the reduction in RBC mass or blood HB concentration resulting in a decrease in the oxygen binding capacity of the blood. It is a HB concentration < 2 SD below the mean for the child’s age. Hb <10. 5 is anemia

ANEMIA 5 HB varies according to age groups. It differs with ethnicity (black children tend to have 0. 5 g less HB than white and Asian children) A sex difference in HB about 0. 5 -1. 0 g/dl appears around puberty. .

Physiology of Hemoglobin Production 7 Erythropoietin is the primary hormone regulator of red blood cell (RBC) production In the fetus, erythropoietin comes from the monocyte/macrophage system of the liver. Postnatally, erythropoietin is produced in the peritubular cells of the kidneys.

Physiology of Hemoglobin Production 8 Normal RBCs survive an average of 120 days. The hemoglobin molecule is a heme-protein complex of two pairs of similar polypeptide chains There are six types of hemoglobin in humans: -the embryonic: Gower-I, Gower-II, Portland, fetal hemoglobin (Hb. F) and -normal adult hemoglobin (Hb. A and Hb. A 2). Hb F has a higher affinity for oxygen than adult hemoglobin Hb F rapidly decrease to trace levels and is ultimately replaced by the age of 6 -12 months by adult Hb. A and Hb. A 2.

PHYSIOLOGICAL ANEMIA 9 PHYSIOLOGIC ANEMIA OF INFANCY occurs at 8 -12 wk in full term babies and 6 -8 wk in premature babies. hemoglobin concentration is as low as 9– 11 g/dl in term and, in premature Minimal hemoglobin levels of 7– 9 g/d. L

PHYSIOLOGIC ANEMIA OF INFANCY 10 Why? at birth, the hemoglobin-oxygen saturation increases from 50 to 95% or more. the increase in tissue oxygen delivery downregulates erythropoietin (EPO) production and, erythropoiesis is suppressed. (still the response to EPO IS NORMAL)

PHYSIOLOGIC ANEMIA OF INFANCY 11 Dilution factors, rapid growth of the baby Shortened RBC SPAN 90 In this physiologic anemia there is no hematologic problem, and no therapy is required If there are : Symptoms , family hx of disease , very low drop in Hb = need investigation , unlikley to be physiological anemia

12 CLASSIFICATION OF ANEMIA

Classification of anemia by pathophysiologic mechanism 13 I. Failure of erythrocyte production A. Bone marrow failure B. Impaired erythropoietin production C. Disorders of erythroid maturation/ineffective erythropoiesis (anemia due to elemints , as vit. b 12 and folate) , there is rbc production but abnormal RBC so destroid , anemic pt (some pic of hemolysis) , II. Increased RBC loss or destruction

14 I. Failure of erythrocyte production

A. Bone marrow failure 15 1. Aplastic anemia (congenital or acquired) pancytopenia 2. Pure red cell aplasia. Just rbc line a. Diamond-Blackfan anemia (congenital) b. Transient erythroblastopenia of childhood (acquired) 3. Marrow replacement a. Malignancies b. Osteopetrosis c. Myelofibrosis

B. Impaired erythropoietin production 16 1. Chronic renal disease most common 2. Hypothyroidism, hypopituitarism 3. Chronic inflammation 4. Protein malnutrition

C. Disorders of erythroid maturation/ineffective erythropoiesis 17 1. Abnormalities of cytoplasmic maturation a. Iron deficiency b. Thalassemia syndromes c. Lead poisoning d. Sideroblastic anemia Maturation disorder : 1 - cytoplasm : the end result is microcytic rbc 2 - nuclear : the end result is macrocytic

C. Disorders of erythroid maturation/ineffective erythropoiesis 18 2. Abnormalities of nuclear maturation a. Vitamin B 12 deficiency b. Folic acid deficiency

II. Increased RBC loss or destruction 19 1. Hemoglobinopathies (including structural and synthetic mutants) 2. Red cell membrane defects (spherocytosis , elliptocytosis) 3. Red cell metabolic defects (G 6 PD, pyruvate d) 4. Antibody-mediated 5. Mechanical injury to the erythrocyte 6. Oxidant-induced injury to the erythrocyte 7. Infectious agent-induced red cell injury 8. Paroxysmal nocturnal hemoglobinuria

Classification of anemia based on red cell size 20 I. Microcytic anemia (MCV < 70 f. L + years of age) II. Normocytic anemia III. Macrocytic anemia (MCV > 84 f. L + 0. 6 × yrs of age) <72 is micro >95 -100 is macro

Microcytic anemia 21 1. Iron deficiency 2. Drug/toxin-mediated (including lead poisoning) 3. Thalassemia syndromes 4. Sideroblastic anemias

Normocytic anemia 22 1. Congenital hemolytic anemias a. Hemoglobinopathies b. Red cell enzyme defects c. Red cell membrane defects 2. Acquired hemolytic anemias. Hemolysis =normo a. Antibody-mediated b. Microangiopathic hemolytic anemias c. Anemia secondary to acute infection 3. Acute blood loss 4. Chronic renal disease 5. Splenic sequestration The most imp. DDX of microcytic. They may have ass. iron deficiency and microcytic in addition to normo

Macrocytic anemia 23 1. With megaloblastic bone marrow (disturbance of DNA synthesis) a. Vitamin B 12 deficiency b. Folic acid deficiency c. Drugs (eg, methotrexate, certain anticonvulsants)

Megaloblastic Changes 24 Megaloblastic anemias share common characteristics. -Erythrocytes are larger and have higher nuclearto-cytoplasmic ratios -Neutrophils hypersegmented, and megakaryocytes are abnormal. -On the molecular level, the maturation of nuclei is delayed, while cytoplasmic development is normal. Must do bone marrow , just hypersegmented neutophile is appear on perphral blood film Megaloplastic : vit. 12 , folate Macrocytic : just rbcs , more DDx of it

Macrocytic anemia 25 2. Without megaloblastic bone marrow a. Aplastic anemia b. Diamond-Blackfan anemia c. Hypothyroidism/hypopituitarism. d. Liver diseasee. e. Bone marrow infiltration. f. Congenital dyserythropoietic anemia

Approach to Patient Newborn age : usualy hemolytic anemia 26 (ABO incomp. , Rh incomp) Age: 12 y : age of iron deficiency IDA develops after doubling of birth wt. Thalassemia and sickle cell anemia. . seen > 6 months Alle are recessive Sex: G 6 PD (more in male ) except spherocytosis Race: G 6 PD in mediteranean , FAMILY Hx (thalasemia , sickle , spherocytosis, G 6 pd ) Neonatal history: hyperbilirubinemia (G 6 PD, spherocytosis, ABO , Rh incompatability) In X-linked : take hx from maternal side

27 #Nutrition : Most commonly iron deficiency : 1 - financialsocial issue 2 - habit of nutrition : 1 - milk exclusivly (enough up to 4 m ) 2 - ﻣﺎﺑﻴﺎﻛﻞ ﻏﻴﺮ ﺷﺒﺲ ﻭﻋﺼﻴﺮ #hemolysis : 1 - juanice 2 - dark urine if massive 3 - family hx 4 - oxidant (fava bean) , dugs(sulfadrugs as trisul) 5 - on lab : normocytic anemia , high retic. count , high LDH , low haptoglubin , schistocyt on blood film # chronic illness ( chronic kidney disease , liver failure , malabsorbtion )

Approach to Patient 28 The patient's past medical history : hyperbilirubinemia , diet, medications, acute or chronic infections, endocrinopathies, easy bruising or blood loss DRUGS: phynetoin. . megaloblastic anemia chloramphinicol : aplastic anemia Infections: Parvo aplastic anemia , EBV autoimmune anemia Malignancy : leuckemia : wloss , rec. infection. Thrombocytopenia (skin rash) , joint pain , bone pain Always ask about it

Physical examination 29 Physical examination: The physical examination is important but will be unremarkable in most children with anemia. Pitecheal rash / purpura : HUS , BM infiltration. Hyperpigmentation: Fnconi Anemia Jaundice / hep B , hep C … APLASTIC ANEMIA Frontal bossing, prominent maxilla Triphalyngeal thumb…Black Fan Diamond TAR absent radius, thrombocytopenia Splenomegaly… spherocytosis, thalassemia Pale : Hb <7

Physical examination 30 Irritability. pallor (usually not seen until hemoglobin levels are less than 7 g per d. L. glossitis, a systolic murmur , growth delay and nail bed changes Children with acute anemia often present more dramatically with jaundice, tachypnea, tachycardia, splenomegaly , hematuria and congestive heart failure. and hypotention Hemodynamic status :

31 Labs & Investigations

Lab 32 CBC: Hb level, WBC count, PLATELET count…is it only low Hb or pancytopenia? Red cell indices: MCV mean corpuscular volume microcytic, normocytic and macrocytic(megalo or not ) RETIC COUNT to distinguish a (decreased RBC production) from a destructive process (increased RBC destruction). corrected reticulocyte count A corrected reticulocyte count above 2% suggests increased RBC production. The marrow normally responds with increased reticulocyte production. Corrected Retic Count=Retic% X (Patient's HCT/Normal HCT) High retic in hemolysis --------In Ineffective erythrobiosis , bone marraw try to overcome so may find high retic. count , do corrected retic. Count >> non hemolysis case will down to normal if still high >> hemolysis --------Low Hb and low retic in bone marrow failure (aplastic anemia )
Lab 33 BLOOD SMEAR/FILM : red cell morphology , Basophilic stippling : aggregated ribosomes; can be seen in thalassemia, iron deficiency and lead poisoning Howell-Jolly bodies : are nuclear remnants seen in asplenia, pernicious anemia and severe iron deficiency Heinz's bodies : are denatured aggregated hemoglobin and can be seen in thalassemia, asplenia and chronic liver disease, G 6 PD. Spherocytes: spherocytosis, hemolytic anemia Target cells: Thalassemia, post splenectomy, iron def. liver dis Fragmented RBCs…. Hemolytic anemia, microangiopathic hemolysis Intravascular hemolysis Blast cell EBV : on blood film : atypical lymphocytes Spherocytosis seen in all hemolytic anemia so don’t diagnose spherocytosis depending only on blood film , you have to do osmotic fragile test

LABs 34 RDW: RBC distribution width (RDW) an index of the variation in red cell size. normal RDW (11. 5%-14. 5%) indicates that a uniform population of RBCs exists. This is seen in healthy patients as well as those with β-thalassemia trait, although the cells are microcytic. elevated RDW indicates a varying population of RBCs ( anisocytosis). Happens with iron deficiency. In cases of hemoglubinopathies or bone marrow problem the RDW is normal (uniform) In case of elemints deficiencies (iron , folate , b 12): high

LABs 35 Other tests include : serum iron level, total iron binding capacity (TIBC) , lead level and serum ferritin direct Coombs' test, lactate dehydrogenase (LDH), haptoglobin and bilirubin (immunmediated ) the vitamin B 12, folate Hb electrophoresis enzyme panel to diagnose enzymopathies, osmotic fragility to diagnose hereditary spherocytosis bone marrow aspiration may be indicated (if malignancy or aplastic anemia). Pancytopenia or when unkown cause or malignancy

36 Iron Deficiency Anemia

IRON DEFICIENCY ANEMIA 37 IDA is the most common hematologic disease of infancy and childhood. The body of a newborn infant contains about 0. 5 g of iron, the adult content is estimated at 5 g. Iron is absorbed in the proximal small intestine, mediated by duodenal proteins. absorption of dietary iron is assumed to be about 10%, a diet containing 8– 10 mg of iron daily is necessary for optimal nutrition. Iron is absorbed two to three times more efficiently from human milk than from cow's milk, partly because of differences in calcium content.

IRON DEFICIENCY ANEMIA 38 During the first years of life , the diet should include foods as infant cereals or formulas that have been fortified with iron; Infants breast-fed exclusively should receive iron supplementation from 4 mo of age. ﺍﻻﻡ ﻳﺎ ﻫﻮ ﻳﺎ Adolescents are also susceptible to iron deficiency because of high requirements due to the growth spurt, dietary deficiencies, and menstrual blood loss.

IDA : Etiology 39 The dietary pattern observed in infants with iron-deficiency anemia is consumption of large amounts of cow's milk and of foods not supplemented with iron. The iron stores are usually sufficient for blood formation in the first 6– 9 mo of life in term infants In term infants, anemia caused by inadequate dietary iron is unusual before 6 mo and usually occurs at 9– 24 mo of age. In low-birth weight infants or those with perinatal blood loss, stored iron may be depleted earlier Blood loss must be considered : lesion of the gastrointestinal tract , hookworm infestation, and Chronic diarrhea in early childhood

Cow milk blood loss 40 chronic intestinal blood loss induced by exposure to a heat- labile protein in whole cow's milk. can be prevented either by reducing the quantity of whole cow's milk , by using heated or evaporated milk, or by feeding a milk substitute. It is not related to enzymatic abnormalities in the mucosa, such as lactase deficiency, nor to typical “milk allergy. ”

IDA : Clinical Manifestations 41 Pallor , headache, generalized weakness Tachycardia and cardiac dilation systolic murmurs. irritability and anorexia characteristic of advanced cases affects attention span, alertness, and learning of both infants and adolescents

LAB diagnosis 42 CBC, red cell indices, RDW blood film, retic count ESR Iron, ferritin, transferrin saturation , TIBC level The serum transferrin receptor (Tf. R) level HB electrophoresis Bone marrow Aspirate (no need if y sure about ID) transferrin receptor saturation: <30 iron deficency (best indication ) Ferritin is an acute phase reactant so may be normal in iron defic. If there is infection

Hematological sequence in IDA 43 First, the tissue iron stores (hemosiderin) disappear Decreased serum ferritin, increased RDW Then serum iron level decreases Iron-binding capacity of the serum (serum transferrin) increases and transferrin saturation falls below normal , then Then free erythrocyte protoporphyrins (FEP) accumulates , then (RBCs) become smaller than normal and their hemoglobin content decreases (Low MCV)

Differential Diagnosis 44 differential from other hypochromic microcytic anemia. α– and β–thalassemia ( RDW is elevated in iron deficiency. ) Thalassemia trait has elevated levels of hemoglobin A 2 and/or increased fetal hemoglobin concentration. Serum iron, total iron-binding capacity (transferrin), and ferritin are normal.

Differential Diagnosis 45 IDA from The anemia of chronic disease (ACD) and infection usually normocytic both the serum iron level and iron-binding capacity (transferrin) are reduced but serum ferritin levels are normal or elevated. serum transferrin receptor (Tf. R) level is not affected by inflammation.

Differential Diagnosis 46 Lead poisoning and iron-deficiency anemia the RBCs are morphologically similar but coarse basophilic stippling in lead poisoning Elevations of blood lead level.

RDW 47 Low MCV Normal MCV High MCV Normal RDW thalassemia Chronic Aplastic anemia HIGH RDW IDA Folate/ b 12 deficiency disease Liver disease

Treatment 48 Treatment with Iron supplement is both therapeutic and diagnostic. Simple ferrous salts (sulfate, gluconate, fumarate) The therapeutic dose is total of 4 -6 mg/kg daily Therapy duration for 3 -6 months Intolerance to oral iron is uncommon in young children, although older children and adolescents sometimes have gastrointestinal complaints.

49 patient's diet, and the consumption of milk should be limited to a reasonable quantity, preferably 500 m. L (1 pint)/24 hr or less. The amount of iron-rich foods is increased

Responses to Iron Therapy in Iron-Deficiency Anemia 50 12– 24 hr Replacement of intracellular iron enzymes; subjective improvement; decreased irritability; increased Appetite 36– 48 hr Initial bone marrow response; erythroid hyperplasia 48– 72 hr Reticulocytosis, peaking at 5– 7 days 4– 30 days Increase in hemoglobin level 1– 3 mo Repletion of stores

Blood Transfusion for IDA 51 a good rapid hematologic response can be achieved in typical iron deficiency, blood transfusion is indicated only when the anemia is very severe with hemoglobin values less than 4 g/d. L , when the patient is clincally symptomatic or critically ill. This procedure may be dangerous because of the associated hypervolemia and cardiac dilatation If symptomatic = transufuse ﺍﻧﺴﻰ ﺍﻟﺮﻗﻢ In case of hemolysis = transfuse when Hb <7

52 THANK YOU
- Slides: 52