1 Air and Oxygen Learning Outcomes Understand that









- Slides: 12

1 Air and Oxygen – Learning Outcomes Understand that air is a mixture of gases. State the composition of air. Show that approximately one fifth of air is oxygen. Show that there is CO 2 and water vapour in air. Demonstrate and describe what happens when a wooden splint is burned in air. Demonstrate and describe what happens when a piece of magnesium is burned in air.

2 Air and Oxygen – Learning Outcomes Prepare a sample of oxygen by decomposing H 2 O 2 using Mn. O 2 as a catalyst. Give the word equation for preparing oxygen. HL: Give the chemical equation for preparing oxygen. Investigate the ability of oxygen to support combustion in a wooden splint and a candle. State two uses of oxygen. Burn carbon and magnesium in oxygen, and test the products using moist litmus paper.

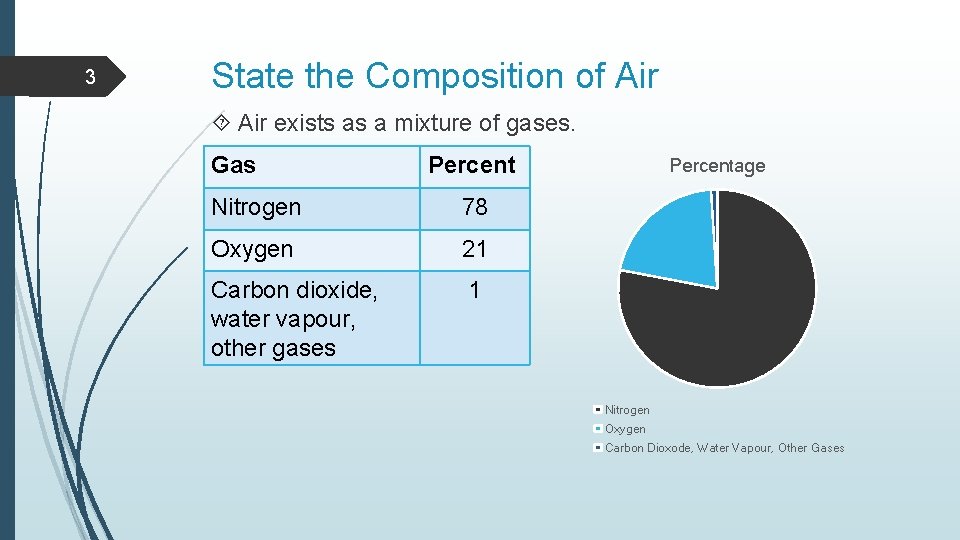
3 State the Composition of Air exists as a mixture of gases. Gas Percent Nitrogen 78 Oxygen 21 Carbon dioxide, water vapour, other gases 1 Percentage Nitrogen Oxygen Carbon Dioxode, Water Vapour, Other Gases

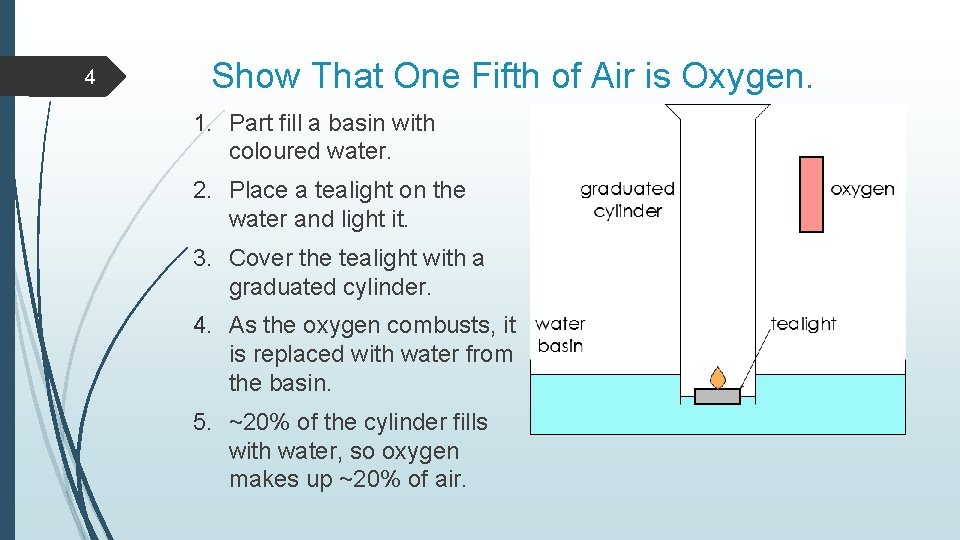
4 Show That One Fifth of Air is Oxygen. 1. Part fill a basin with coloured water. 2. Place a tealight on the water and light it. 3. Cover the tealight with a graduated cylinder. 4. As the oxygen combusts, it is replaced with water from the basin. 5. ~20% of the cylinder fills with water, so oxygen makes up ~20% of air.

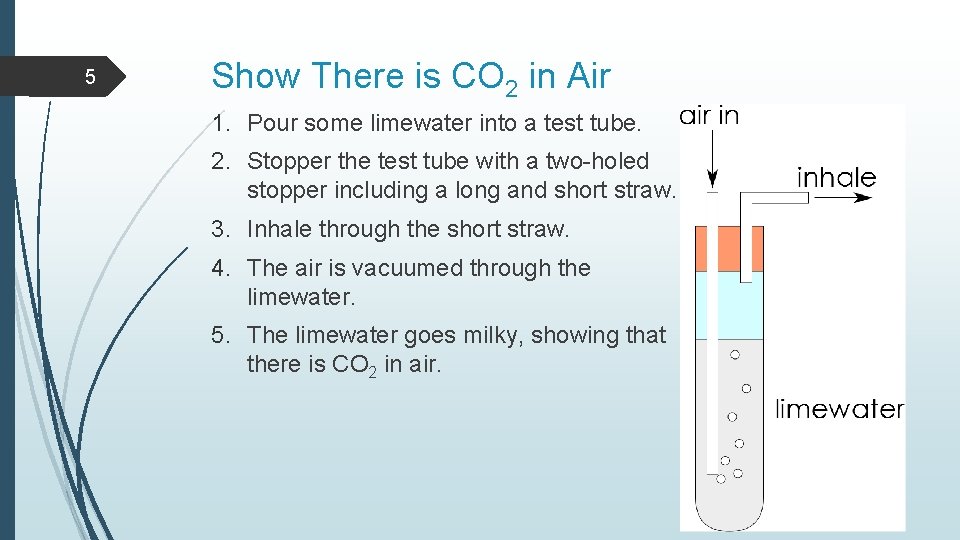
5 Show There is CO 2 in Air 1. Pour some limewater into a test tube. 2. Stopper the test tube with a two-holed stopper including a long and short straw. 3. Inhale through the short straw. 4. The air is vacuumed through the limewater. 5. The limewater goes milky, showing that there is CO 2 in air.

6 Show There is Water Vapour in Air 1. Fill a test tube with ice and water. 2. A liquid forms on the outside of the test tube. 3. Test the liquid using anhydrous copper sulfate. 4. The copper sulfate turns blue, indicating the liquid formed was water. 5. Thus there is water vapour in air.

7 Prepare a Sample of Oxygen 1. Connect the output of Buchner flask containing manganese dioxide to a beehive shelf in a water basin. 2. Place a gas jar over the beehive shelf. 3. Drop hydrogen peroxide into the Buchner flask. 4. Replace gas jar as needed when it fills with oxygen.

8 Give the Equation for Preparing Oxygen

9 Burn a Wooden Splint in Air 1. Light a wooden splint in air. Investigate the Ability of Oxygen to Relight a Glowing Wooden Splint 1. Light a wooden splint and blow it out. 2. Hold the glowing splint in a container of oxygen. 3. The glowing splint relights.

10 Burn Carbon in Oxygen and Test the Product 1. Heat a piece of carbon over a Bunsen burner until it starts to glow. 2. Quickly put it into a jar of oxygen. 3. The carbon ignites. 4. Add some water to the jar. 5. Test the water using blue litmus paper. 6. The paper turns red, indicating the gas (carbon dioxide) is acidic.

11 Burn Magnesium in Oxygen and Test the Product 1. Heat a piece of magnesium over a Bunsen burner until it starts to glow. 2. Quickly put it into a jar of oxygen. 3. The magnesium burns with a blight flame. 4. Add some water to the jar. 5. Test the water using red litmus paper. 6. The paper turns blue, indicating the magnesium oxide is basic.

by Daniel Scott – public domain Used in welding to increase combustion. Used in medicine to help breathing. by Hmwith – public domain State Two Uses of Oxygen 12