1 7 Dimensional Analysis Dimensional Analysis FactorLabel Method

1. 7 – Dimensional Analysis

Dimensional Analysis (Factor-Label Method) • 1. Dimensional Analysis – An approach to changing units making use of conversion factors. • • 2. Conversion Factor – An equality such as “ 12 inches = 1 foot”. If this conversion factor is expressed as a fraction, then it has the value of 1: 1 foot = 1 12 inches • Multiplying a quantity by 1 does not change the value of the quantity. If the conversion factor is exact, it will not affect significance. If the conversion factor is rounded (when it converts between English and Metric unit systems), then it must be considered when determining sig figs.

Conversion Factors Found on Equation Sheet • • • Conversion Factors: 1 cm 3 = 1 m. L (exact) 1 L = 1. 06 qt 1 qt = 946 m. L 1 lb = 453. 6 g 1 kg = 2. 20 lb 1 inch = 2. 54 cm (exact) 1 m = 39. 37 in 1 mile = 1. 609 km • • 1 mile = 5, 280 ft 1 hour = 3600 sec 1 gal = 4 qts. 1 yd = 3 ft. 1 ft = 12 in 1 L = 1000 m. L 1 kg = 1000 g 1 m = 100 cm

Dimensional Analysis (Factor-Label Method) • HELPFUL HINTS: • When solving these problems, use the conversion factors given as a means to convert units. The unit you are trying to eliminate should always go on the opposite side. • Always start with your given. • If you set up the conversion factor first, with units on opposite sides, and then put in the numbers, should lead to the correct answer
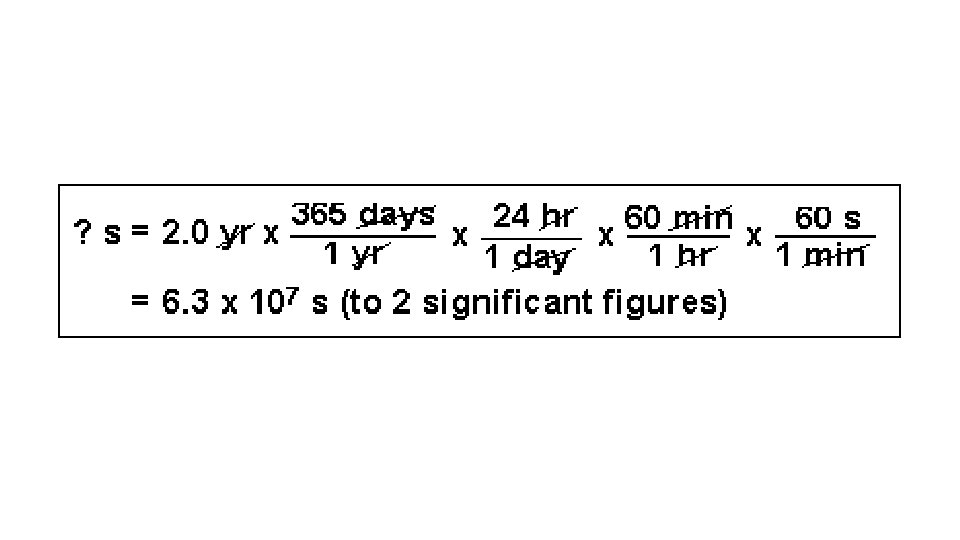

Sample problems: • 1. A distance of 320. inches is equal to how many feet? • 320. in x 1 ft = 26. 7 ft • 12 in • ** The answer has 3 sig figs because the given had 3 sig figs.

Sample problems: • 2. 17. 4 hours is equal to how many minutes? 17. 4 hours x 60 min = 1044 min = 1040 min 1 hr ** The answer has 3 sig figs because the given had 3 sig figs

Sample problems: • 3. 1, 480, 000 inches are equal to how many miles? = 23. 4 mi

• 4. If you are traveling at 35 miles per hour, what is your speed in feet per second? • • 51 ft/sec • 5. Your car has a fuel efficiency rating of 36. 2 miles per gallon. Convert this to kilometers per liter. • • 15. 4 km/L

Temperature • Temperature: measure of the average kinetic energy of particles in a material

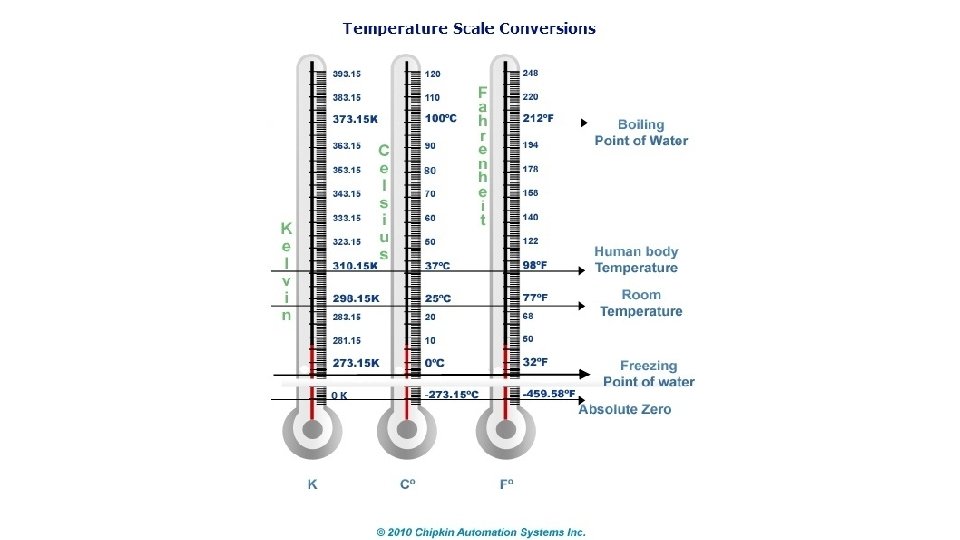
Temperature scales • a. The Celsius temperature scale (°C) is defined on a couple of the properties of water. In degrees Celsius, give the • Freezing point of water: 0°C (= 273 K = 32°F) • Boiling point of water: 100°C (= 373 K = 212°F)
Countries that don’t use Celsius.

Temperature scales • b. Kelvin: The Kelvin scale has the same size degree as the Celsius scale, but starts at absolute zero. Thus, it has no negative values. • i. Absolute Zero: coldest theoretical temperature where all particle motion stops. • 0°K = -273. 15°C = -459. 67°F

Temperature scales • • c. Fahrenheit: English Unit of Temperature; will only be used for reference.
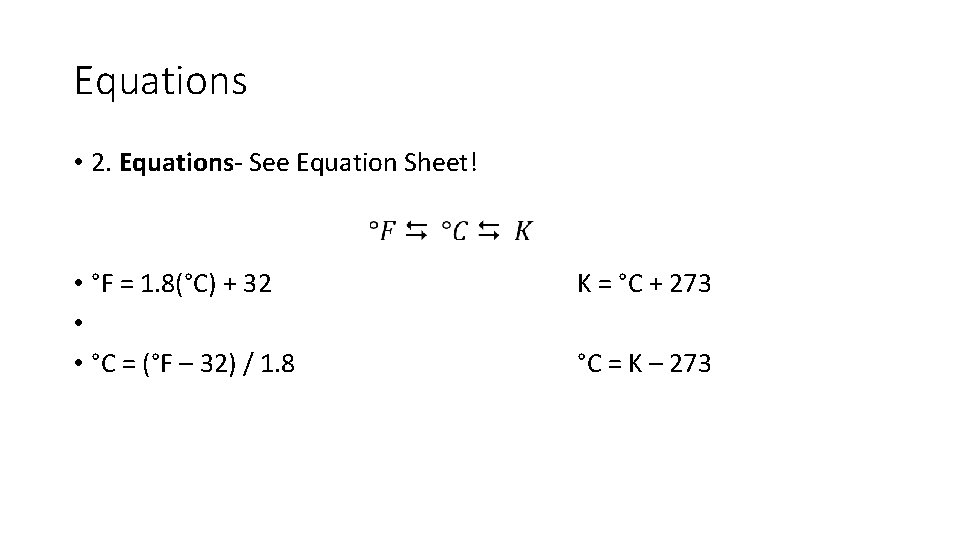
Equations • 2. Equations- See Equation Sheet! • °F = 1. 8(°C) + 32 • • °C = (°F – 32) / 1. 8 K = °C + 273 °C = K – 273

• Sample Problems: Significant Figures are NOT important with temperature; use the whole number • The boiling point of liquid nitrogen is -196°C. What would this be in °F and °K? • K = °C + 273 = -196+273 = 77 K • °F = 1. 8 X °C +32 = 1. 8(-196) + 32 = -320. °F

• . Normal body temperature is 98. 6°F. Convert this to °C and °K. • °C = (°F – 32)/1. 8 = (98. 6 – 32) /1. 8 = 37. 0°C • K = °C + 273 = 37. 0 + 273 = 310. 0 K
- Slides: 18