1 2 Hydrocarbons Alkanes Alkanes bonds are still

(1. 2) Hydrocarbons Alkanes

Alkanes § bonds are still relatively weak yet a single bond is harder to break than a multiple bond § are non-polar and have weak intermolecular force, therefore relatively low boiling points § boiling point increases with an increase in number of atoms (increasing intermolecular force)
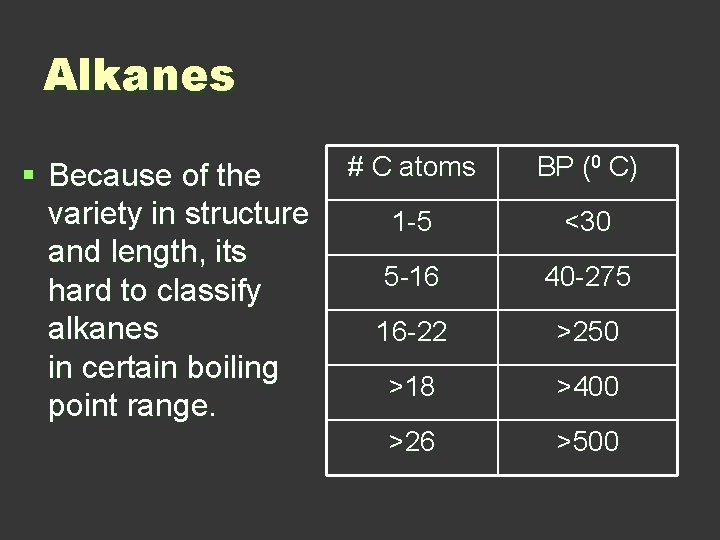
Alkanes § Because of the variety in structure and length, its hard to classify alkanes in certain boiling point range. # C atoms BP (0 C) 1 -5 <30 5 -16 40 -275 16 -22 >250 >18 >400 >26 >500
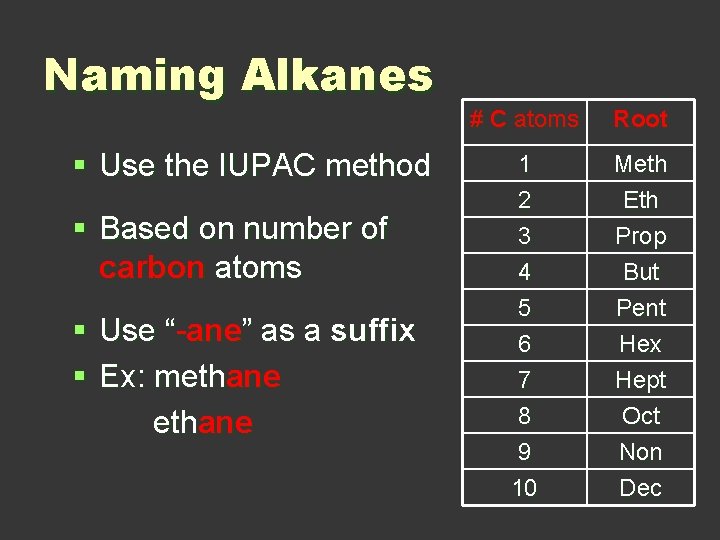
Naming Alkanes § Use the IUPAC method § Based on number of carbon atoms § Use “-ane” as a suffix § Ex: methane # C atoms Root 1 2 3 Meth Eth Prop 4 5 6 7 8 9 10 But Pent Hex Hept Oct Non Dec

Naming Alkanes Use general formula for alkanes Cn. H 2 n+2 where n= # C atoms ex. Propane (C 3 H 8) Heptane (7 C atoms) (C 7 H 16)

Alkanes As a group of compounds alkanes form a homologous series. Each molecule differs from the next by the structural unit CH 2 ex. ethane, propane, butane
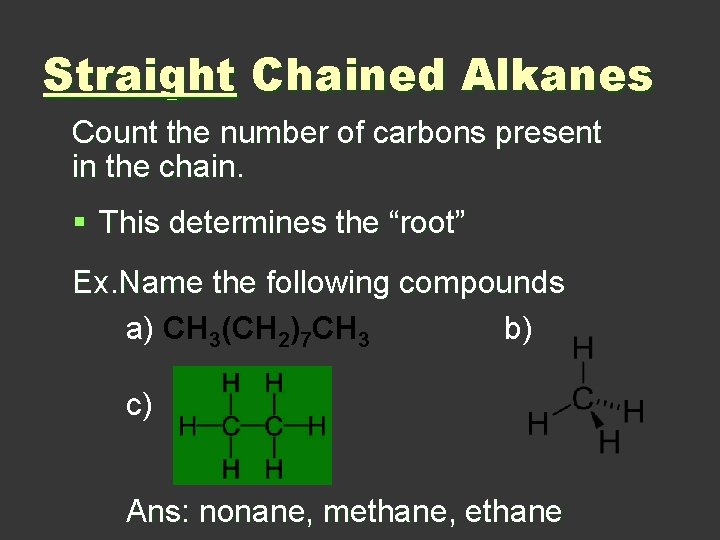
Straight Chained Alkanes Count the number of carbons present in the chain. § This determines the “root” Ex. Name the following compounds a) CH 3(CH 2)7 CH 3 b) c) Ans: nonane, methane, ethane

Straight Chained Alkanes Ex. Draw the following § a) Propane (complete structural diagram) § b) Decane (condensed structural diagram) § c) Pentane (line structural diagram)
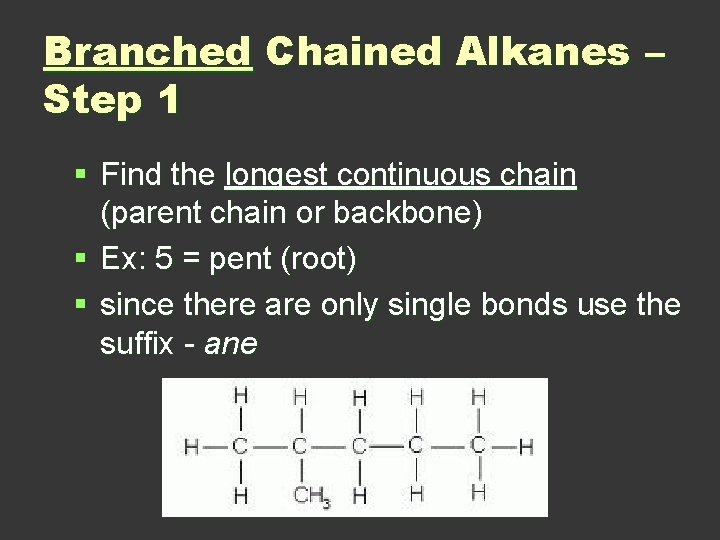
Branched Chained Alkanes – Step 1 § Find the longest continuous chain (parent chain or backbone) § Ex: 5 = pent (root) § since there are only single bonds use the suffix - ane
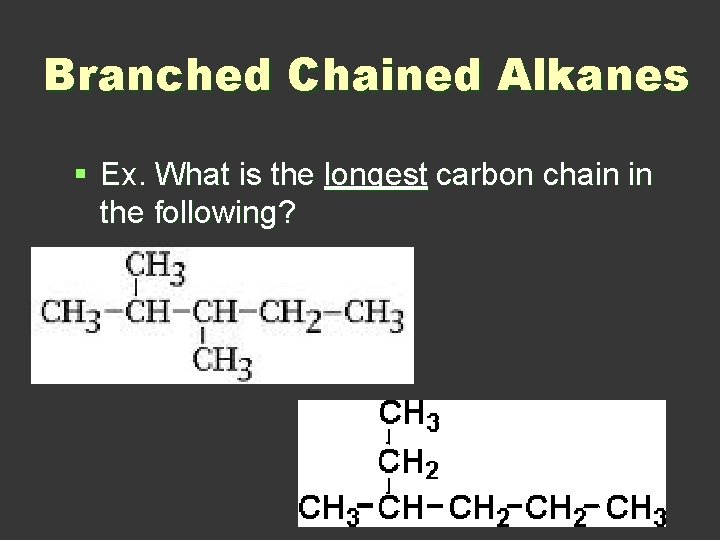
Branched Chained Alkanes § Ex. What is the longest carbon chain in the following?
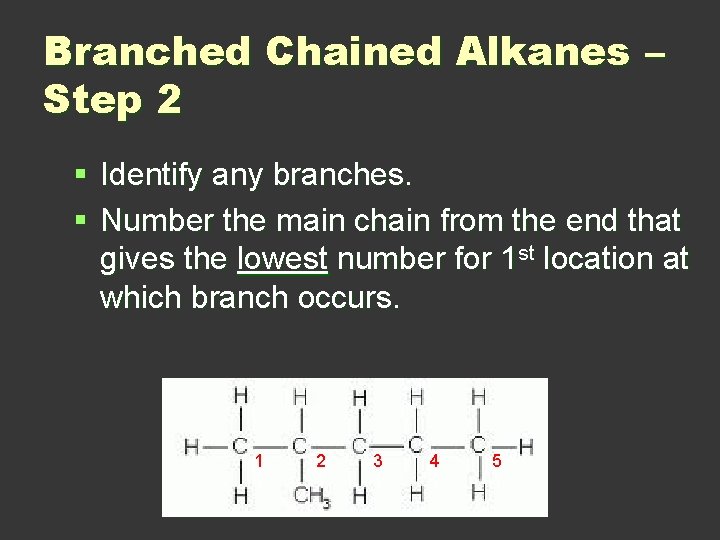
Branched Chained Alkanes – Step 2 § Identify any branches. § Number the main chain from the end that gives the lowest number for 1 st location at which branch occurs. 1 1 2 1 3 1 4 1 5 1

Branched Chained Alkanes – Step 3 § Alkyl group: hydrocarbon group from an alkane by removing a hydrogen § Name branch using root name (table 2 –p 12) and change the ending to –yl § Ex. –CH 3 methyl
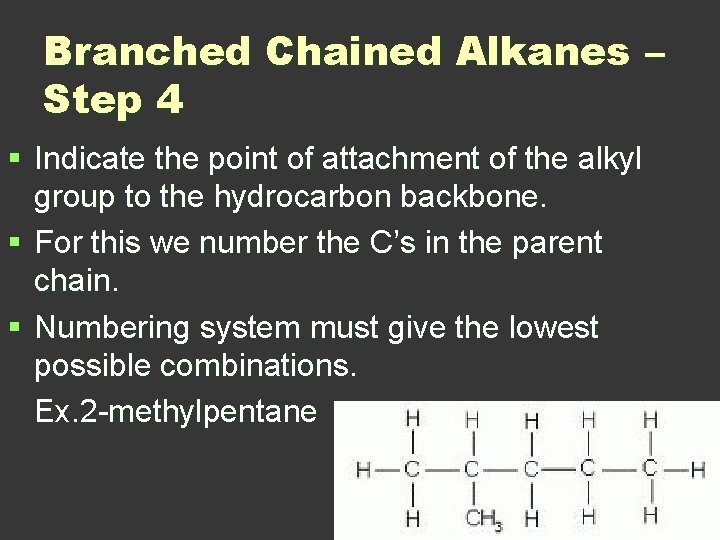
Branched Chained Alkanes – Step 4 § Indicate the point of attachment of the alkyl group to the hydrocarbon backbone. § For this we number the C’s in the parent chain. § Numbering system must give the lowest possible combinations. Ex. 2 -methylpentane

Branched Chained Alkanes § Ex. Draw the following § 3 -ethylhexane

Branched Chained Alkanes We separate…. § numbers from numbers by a comma (, ) and § numbers from letters with a hyphen (-) Spelling Counts!

Branched Chained Alkanes § When multiple branches of the same type are present: § Use the suffix di, tri, tetra etc. before the alkyl group Ex. a) 2, 3 -dimethylhexane b) 2, 3, 3 -trimethylpentane

Branched Chained Alkanes § When multiple branches of the different type are present: § Name the alkyl groups, with number indicating position on parent chain, in alphabetical order. § Note: Some textbooks may be different. § Ex. a) 3 -ethyl-5 -methylheptane b) 4 -ethyl-2, 3 -dimethyloctane § Recall: alphabetical order for alkyl groups, excluding prefix

Branched Chained Alkanes Ex. Name the following: CH 2 CH 3 -CH-CH 2 -CH-CH 3 CH 2 CH 3

Isomers § Isomers have the same molecular formula but different structures. § They have different properties (ex. melting/boiling points, density, reactivity )
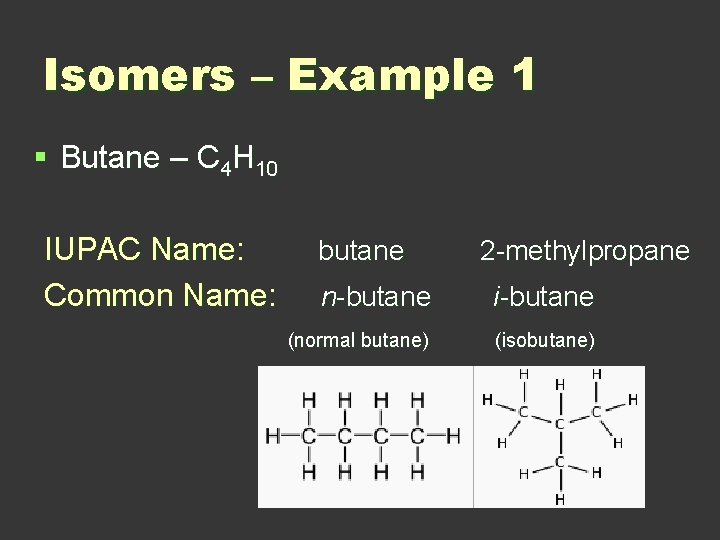
Isomers – Example 1 § Butane – C 4 H 10 IUPAC Name: Common Name: butane 2 -methylpropane n-butane i-butane (normal butane) (isobutane)

Isomers – Example 2 § Pentane - C 5 H 12 Draw all of the ways this compound could be structured. Then name the isomers! (Hint – there are 3 isomers) Ans: pentane, 2 -methylbutane, 2, 2 -dimethylpropane

Isomers of Alkyl Groups When we consider potential attachment points, there are 2 propyl isomers (-C 3 H 7): p 13 Figure 4 Note: The coloured bond indicates where the group is attached to the larger molecule
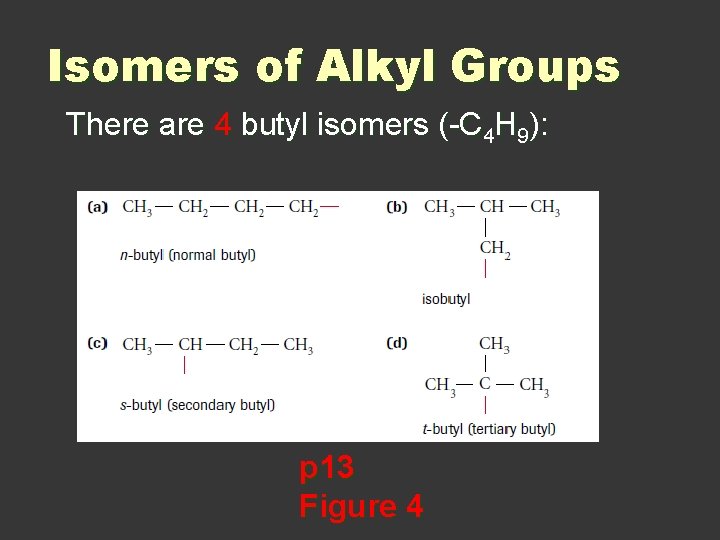
Isomers of Alkyl Groups There are 4 butyl isomers (-C 4 H 9): p 13 Figure 4
- Slides: 23