1 0 Atomic structure Lister p 4 20




















- Slides: 49

1. 0 Atomic structure Lister p 4 - 20

AQA AS Specification Lessons 1 -2 3 Topics Fundamental particles • be able to describe the properties of protons, neutrons and electrons in terms of relative charge and relative mass Electron arrangement • know that early models of atomic structure predicted that atoms and ions with noble gas electron-arrangements should be stable 4 -7 Mass number and isotopes • be able to recall the meaning of mass number (A) and atomic(proton) number (Z) • be able to explain the existence of isotopes • understand the principles of a simple mass spec, limited to ionisation, acceleration, deflection and detection, and its use for identifying elements and RMM 8 -11 Electron arrangement • know the electron configurations of atoms and ions up to Z = 36 in terms of levels and sub-levels (orbitals) s, p and d • know the meaning of the term ionisation energy. • understand how ionisation energies in Period 3 (Na – Ar) and in. Group 2 (Be – Ba) give evidence for electron arrangement in sub-levels and in levels

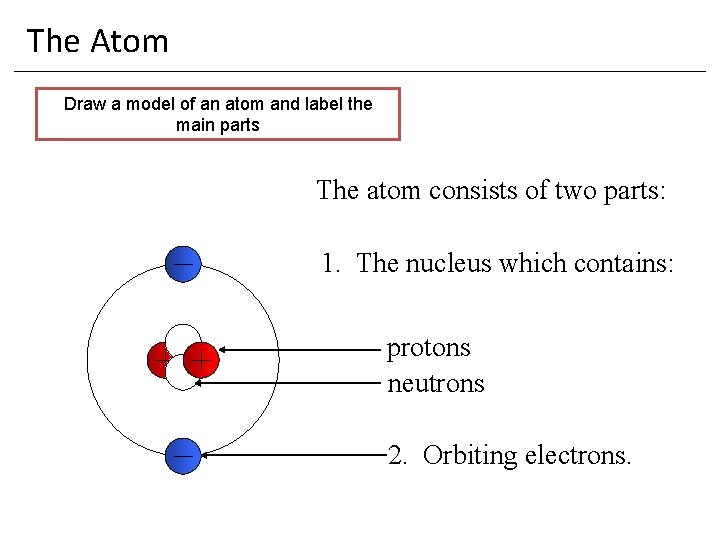
The Atom Draw a model of an atom and label the main parts The atom consists of two parts: 1. The nucleus which contains: protons neutrons 2. Orbiting electrons.

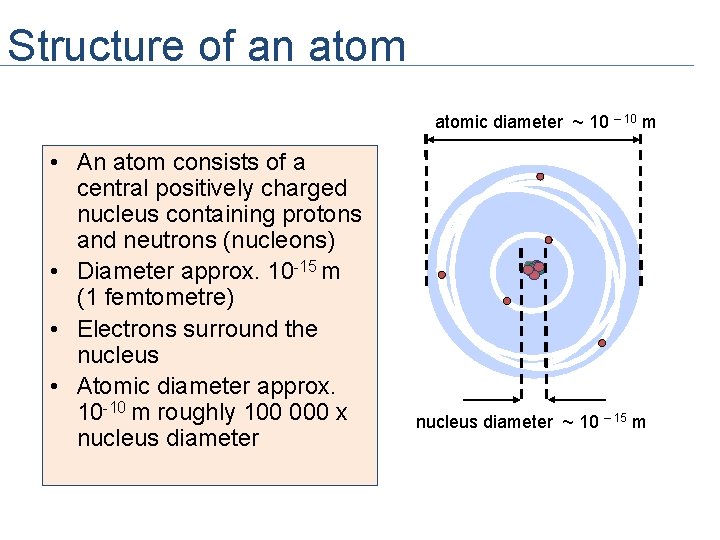
Structure of an atomic diameter ~ 10 – 10 m • An atom consists of a central positively charged nucleus containing protons and neutrons (nucleons) • Diameter approx. 10 -15 m (1 femtometre) • Electrons surround the nucleus • Atomic diameter approx. 10 -10 m roughly 100 000 x nucleus diameter ~ 10 – 15 m

Atoms: How small? If a helium atom was the size of a full stop, then the average gerbil would be the size of the Earth.

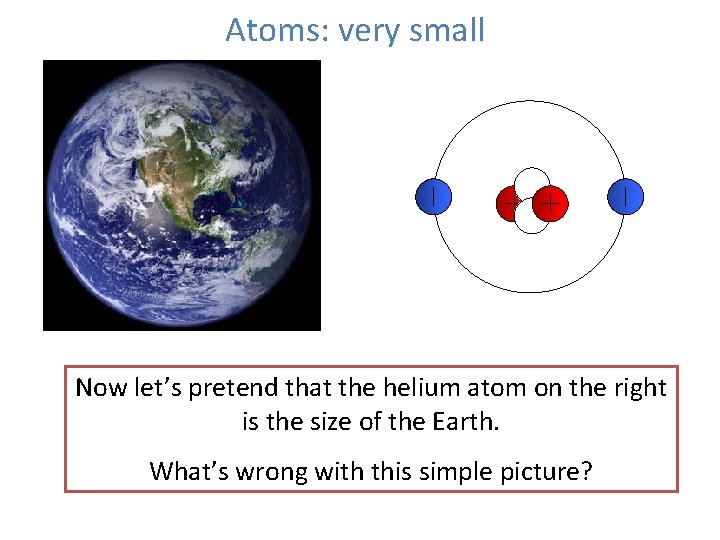
Atoms: very small Now let’s pretend that the helium atom on the right is the size of the Earth. What’s wrong with this simple picture?

How big is a nucleus? The helium atom is not in the right proportions. The three subatomic particles are wrongly enormous in comparison to the atom’s radius.

Most of the atom is empty space! If you imagine an atom being the size of Wembley stadium, the nucleus would be about the size of a football on the centre spot. The electrons would be two peas flying around the whole stadium. The rest of it: emptiness.

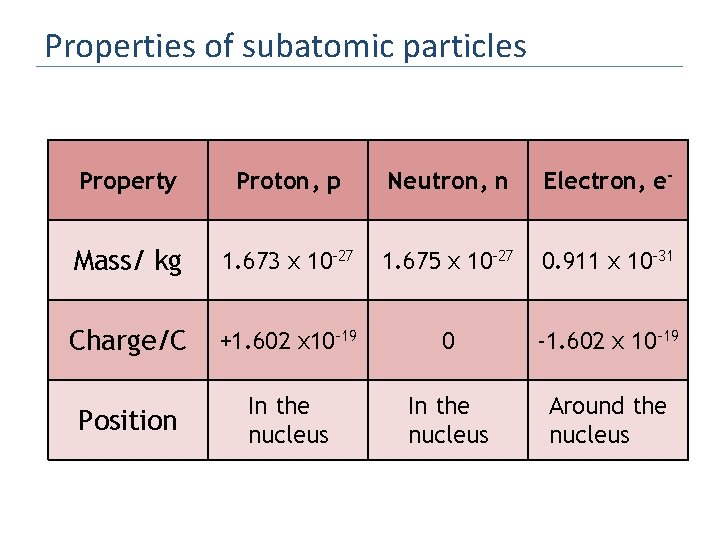
Properties of subatomic particles Property Proton, p Neutron, n Electron, e- Mass/ kg 1. 673 x 10 -27 1. 675 x 10 -27 0. 911 x 10 -31 Charge/C +1. 602 x 10 -19 0 -1. 602 x 10 -19 Position In the nucleus Around the nucleus

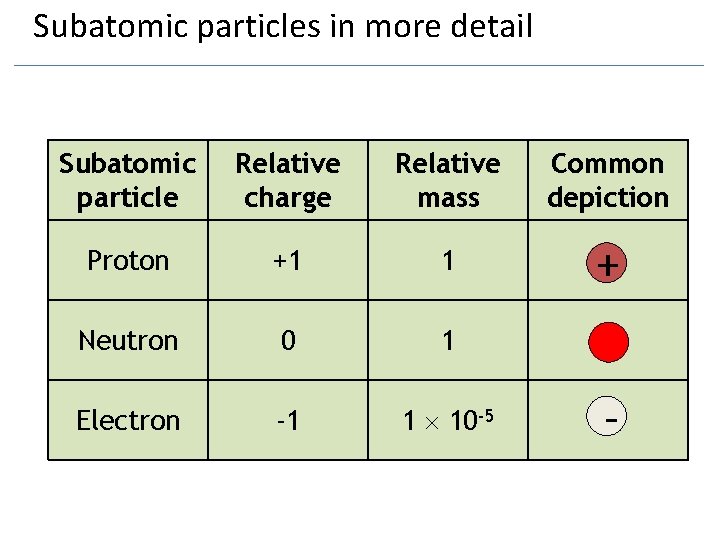
Subatomic particles in more detail Subatomic particle Relative charge Relative mass Common depiction Proton +1 1 + Neutron 0 1 Electron -1 1 10 -5 -

Subatomic particles in more detail Subatomic particle Relative charge Relative mass Common depiction Proton +1 1 + Neutron 0 1 Electron -1 1 10 -5 -

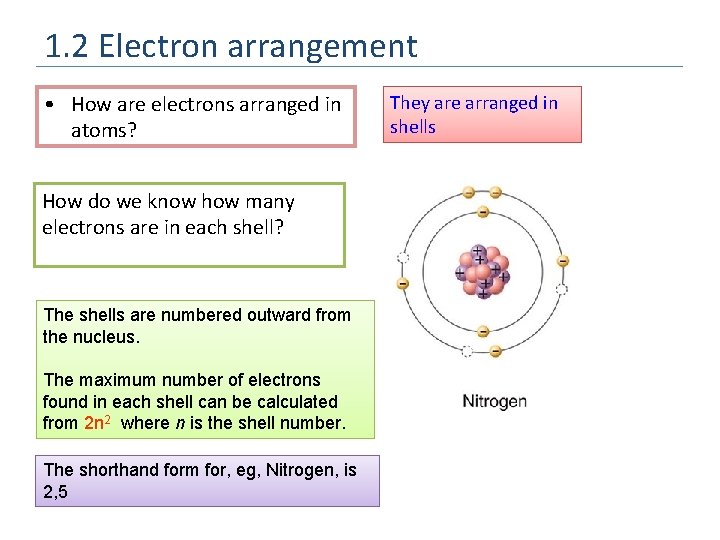
1. 2 Electron arrangement • How are electrons arranged in atoms? How do we know how many electrons are in each shell? The shells are numbered outward from the nucleus. The maximum number of electrons found in each shell can be calculated from 2 n 2 where n is the shell number. The shorthand form for, eg, Nitrogen, is 2, 5 They are arranged in shells

Task Complete the following table: Shell Number Maximum number of electrons 1 2 x 12 =2 x 1 =2 2 2 x 22 =2 x 4 =8 3 2 x 32 =2 x 9 = 18 4 2 x 42 = 2 x 16 = 32 5 2 x 52 = 2 x 25 = 50 Now complete worksheet 1. 1

1. 3 Mass number, atomic number and isotopes How can we describe an atom in terms of it’s subatomic structure? What information do we need to know? The number of protons is called the Atomic number. What is significant about the number of protons in the nucleus? The number of nucelons is called the Mass number. What information can we get from this? The number of protons The number of neutrons The number of electrons It tells us what the element is and how many electrons are present in the neutral atom We can find out the number of neutrons

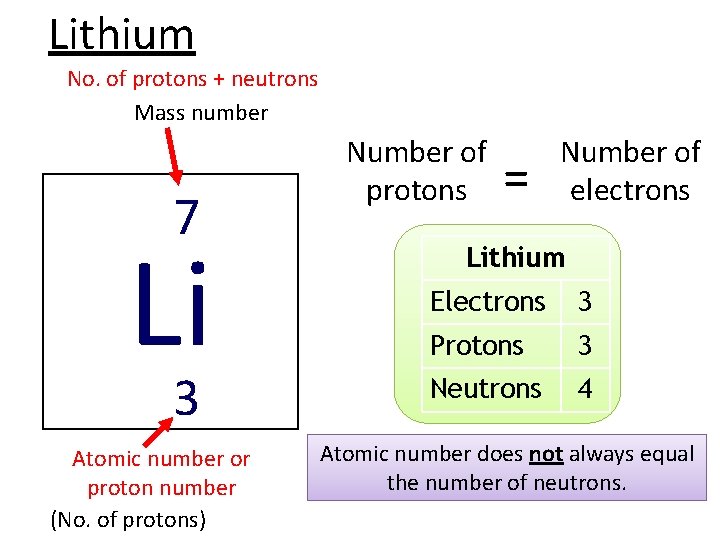
Lithium No. of protons + neutrons Mass number 7 Li 3 Atomic number or proton number (No. of protons) Number of protons = Number of electrons Lithium Electrons 3 Protons 3 Neutrons 4 Atomic number does not always equal the number of neutrons.

Lithium Number of protons = Number of electrons Lithium This is because the atom is neutral. The charges balance out Electrons 3 -3 charge Protons 3 +3 charge Neutrons 4 No charge But atoms can gain and lose electrons (they become ions). This changes the overall charge on the atom.

Isotopes Some isotopes of lithium: 4 Li 6 Li 4 -Lithium 6 -Lithium The number of protons “defines” an element – nothing else! 3 protons, 1 neutron 3 protons, 3 neutrons 3 protons, 7 neutrons 10 Li 10 -Lithium 11 Li 3 protons, 11 -Lithium 8 neutrons 7 -Lithium (7 Li) Electrons 3 Protons 3 Neutrons 4 Lithium: always 3 protons! Atomic number does not always equal the number of neutrons. This can change, even in atoms of the same element. These are called isotopes.

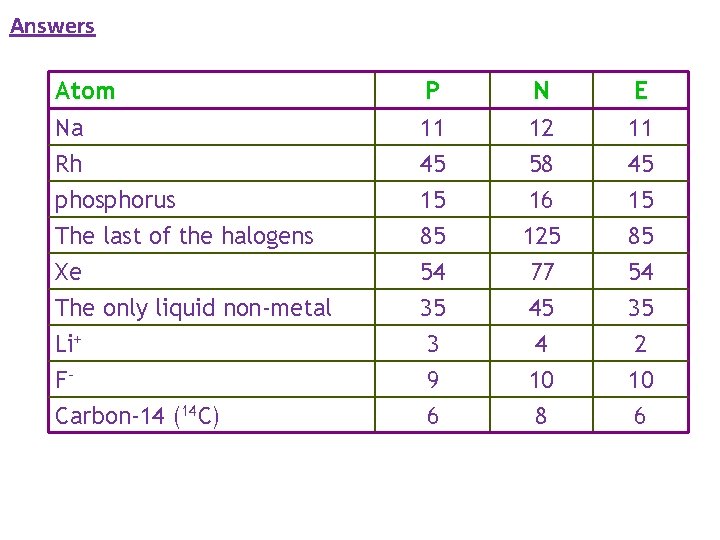
Complete: Atom Na Rh phosphorus The last of the halogens Xe The only liquid non-metal Li+ FCarbon-14 (14 C) 4 2 He A helium atom P n e- 2 protons 2 electrons 2 neutrons

Answers Atom Na Rh phosphorus The last of the halogens Xe The only liquid non-metal Li+ FCarbon-14 (14 C) P 11 45 15 85 54 35 3 9 6 N 12 58 16 125 77 45 4 10 8 E 11 45 15 85 54 35 2 10 6

Chemical properties of isotopes Would you expect the isotopes of lithium to have the same chemical properties? What is the Mass number, Z, of Chlorine? How can you get a fraction of a nucleon? Yes – chemistry is about the movement of electrons 35. 5 The relative abundance of two chlorine isotopes is similar, hence the mass number on the PT is an average number determined by the abundances of the isotopes

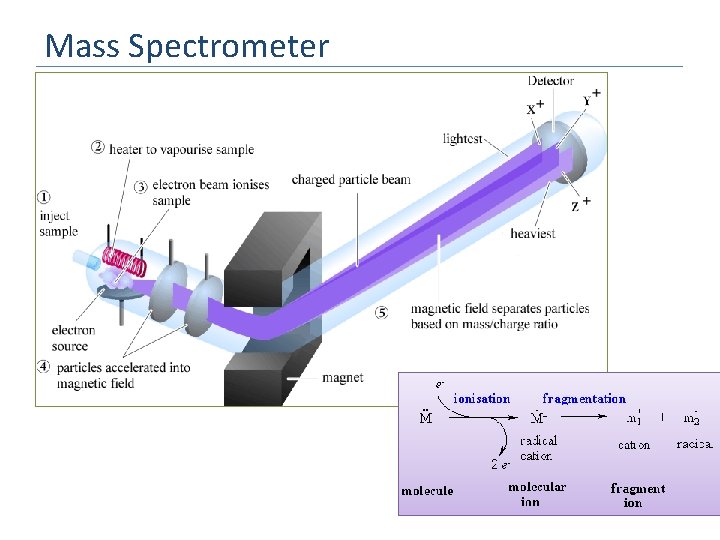
1. 4 Mass spectrometry What does a mass spectrometer do? Why is it important that the instrument is under vacuum? How are samples put into the machine? It ionizes atoms and then sends them through an em field where they become deflected on the basis of their mass and charge To prevent collisions of the ions with gas molecules Volatile liquids and gases can be injected directly, solids must be vapourised. http: //www. youtube. com/watch? v=Jwao 0 O 0_q. M&feature=related

Mass Spectrometer

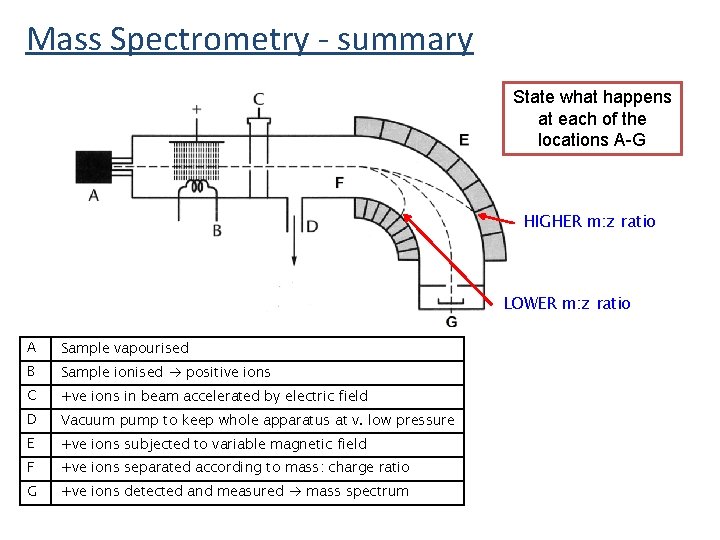
Mass Spectrometry - summary State what happens at each of the locations A-G HIGHER m: z ratio LOWER m: z ratio A Sample vapourised B Sample ionised positive ions C +ve ions in beam accelerated by electric field D Vacuum pump to keep whole apparatus at v. low pressure E +ve ions subjected to variable magnetic field F +ve ions separated according to mass: charge ratio G +ve ions detected and measured mass spectrum

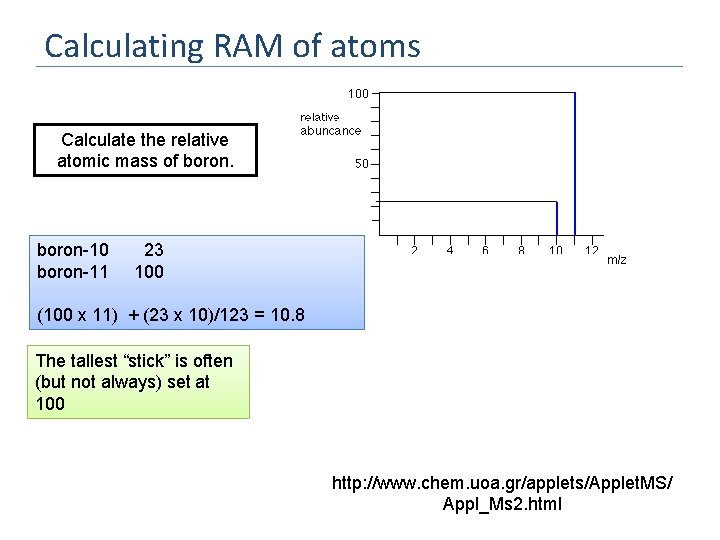
Calculating RAM of atoms Calculate the relative atomic mass of boron-10 boron-11 23 100 (100 x 11) + (23 x 10)/123 = 10. 8 The tallest “stick” is often (but not always) set at 100 http: //www. chem. uoa. gr/applets/Applet. MS/ Appl_Ms 2. html

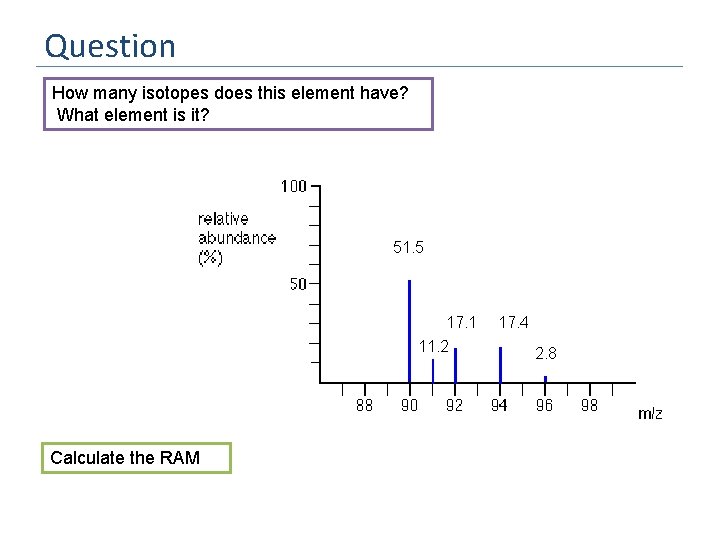
Question How many isotopes does this element have? What element is it? 51. 5 17. 1 11. 2 Calculate the RAM 17. 4 2. 8

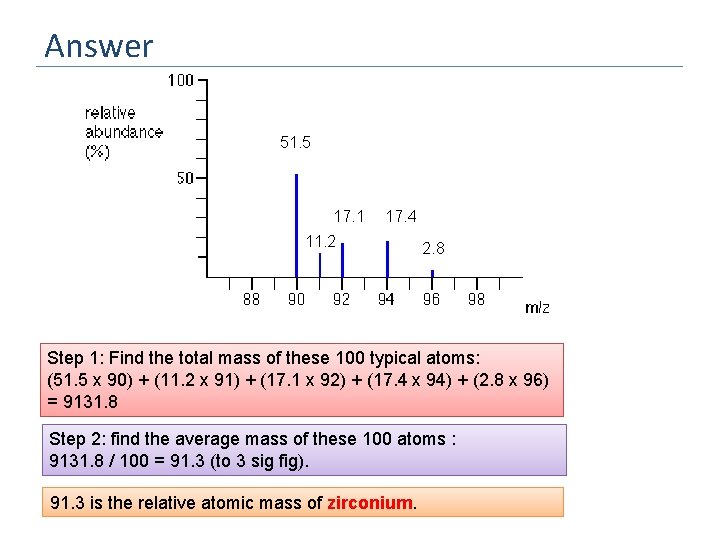
Answer 51. 5 17. 1 17. 4 11. 2 2. 8 Step 1: Find the total mass of these 100 typical atoms: (51. 5 x 90) + (11. 2 x 91) + (17. 1 x 92) + (17. 4 x 94) + (2. 8 x 96) = 9131. 8 Step 2: find the average mass of these 100 atoms : 9131. 8 / 100 = 91. 3 (to 3 sig fig). 91. 3 is the relative atomic mass of zirconium.

Question The mass spectrum of uranium has 3 peaks: at 234 m/z, 235 m/z and 238 m/z. The abundance of the isotopes is 0. 006%, 0. 72% and 99. 2% respectively. What is the average relative atomic mass of uranium? 240 237. 8 238

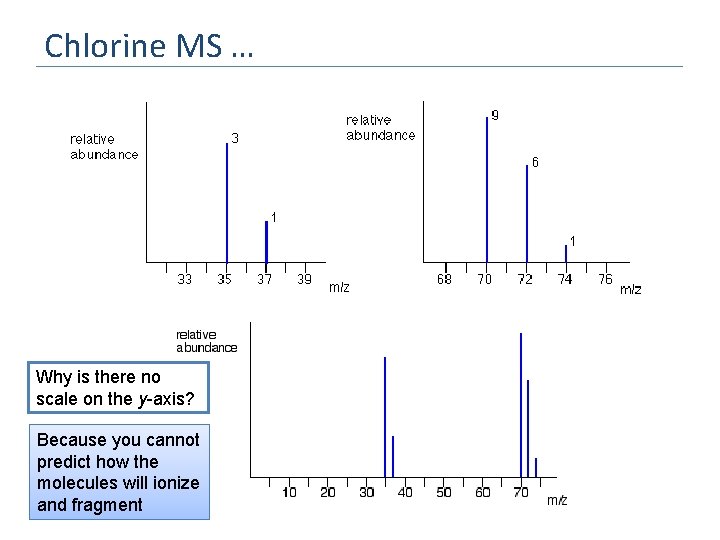
Question Chlorine has two isotopes, 35 Cl and 37 Cl, in the approximate ratio of 3 atoms of 35 Cl to 1 atom of 37 Cl. Draw the stick diagram for Chlorine Wrong! Why? The problem is that chlorine consists of molecules, not individual atoms. When chlorine is passed into the ionisation chamber, an electron is knocked off the molecule to give a molecular ion, Cl 2+. Doubly charges ions can also form. These ions aren’t very stable, and some will fall apart to give a chlorine atom and a Cl+ ion. The term for this is fragmentation

Chlorine MS What can molecular chlorine ions (Cl 2+ ) fragment into? Cl 2+ Cl + Cl+ What happens to the Cl atom? What are the possible combinations of chlorine-35 and chlorine-37 atoms in a Cl 2+ ion? What would the MS look like? If it doesn’t acquire a charge in the ionization chamber then it gets “lost” in the MS Both atoms could be 35 Cl, both atoms could be 37 Cl, or you could have one of each sort. Masses of the Cl 2+ ion: 35 + 35 = 70 35 + 37 = 72 37 + 37 = 74

Chlorine MS … Why is there no scale on the y-axis? Because you cannot predict how the molecules will ionize and fragment

1. 5 Electron configurations Why is the periodic table broken up into sections? What links each of these sections? The distribution of electrons within the shells is, in most cases, more complicated than simple spheres. The regions within the PT closely follow the patterns of these distributions – or probabilities of electron density The shells represent energy levels in atoms. Electrons can move between these levels, gaining or losing energy in the process. http: //www. yellowtang. org/images/electron s_atoms_pos_c_la_784. jpg

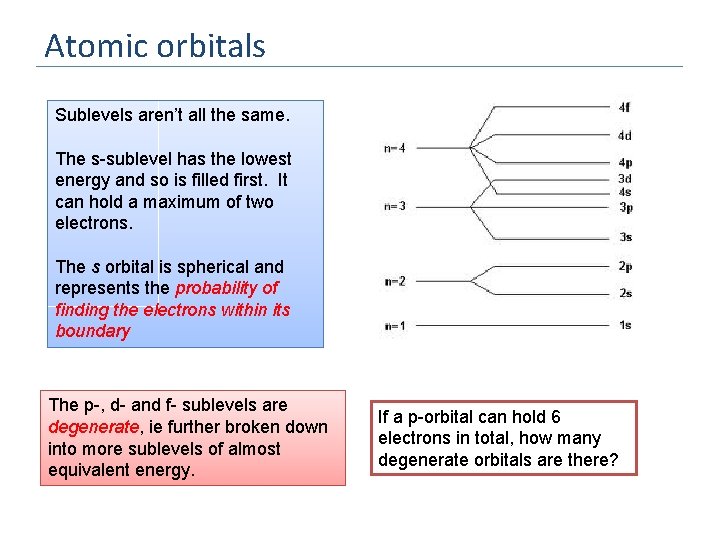
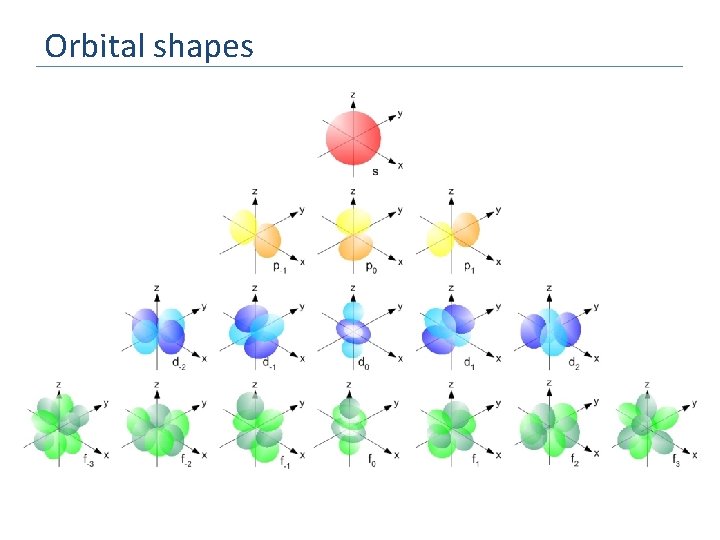
Sublevels Each energy level is divided into one or more sublevels. These sublevels have energies that differ slightly from that of the shell energy. What is the significance of the order of the subshells? E 3 How many types of sublevel are there? 2 (hint – think about he number of regions in the PT) 1 There are 4: s. p. d. f The “s-block” comprises Groups 1 and 2 The “p- block” comprises Groups 3 - 8 d p s How many electrons can an s sublevel have in it? How many electrons can a p sublevel have in it?

Atomic orbitals Sublevels aren’t all the same. The s-sublevel has the lowest energy and so is filled first. It can hold a maximum of two electrons. The s orbital is spherical and represents the probability of finding the electrons within its boundary The p-, d- and f- sublevels are degenerate, ie further broken down into more sublevels of almost equivalent energy. If a p-orbital can hold 6 electrons in total, how many degenerate orbitals are there?

Orbital shapes

Spin Electrons have a property called “spin”. This determines the way in which the degenerate levels are populated. Electron s are either spin up, or spin down – ie clockwise or anticlockwise. (corresponding to a spin quantum number of +1/2 and -1/2) Degenerate orbitals of the same energy fill up first. Parallel spins go in first followed by antiparallel spin. 1 s 2 s 2 px 2 py 2 pz Nomenclature: the number of electrons in a particular orbital is denoted by superscript. e. g. 1 s 2 2 s 2 3 p 2

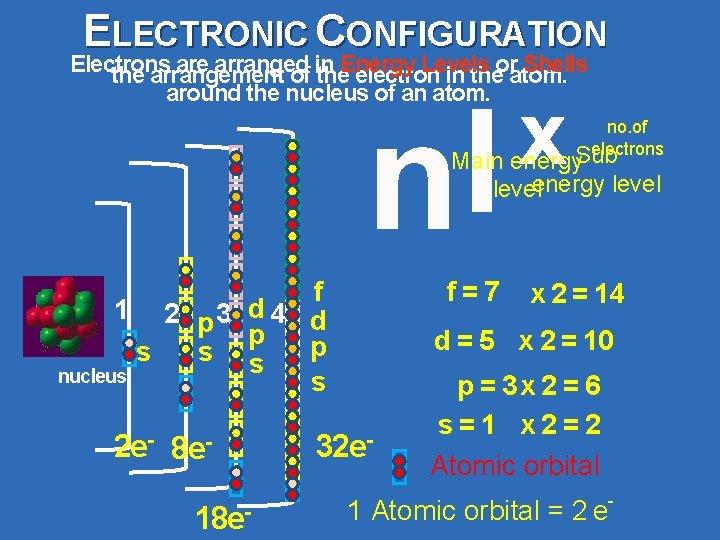
ELECTRONIC CONFIGURATION Electrons are arranged Levels Shells the arrangement of in the. Energy electron in theoratom. around the nucleus of an atom. l n x no. of electrons Sub Main energy level f 1 2 p 3 d 4 d p p s s s nucleus s 2 e- 8 e 18 e- f=7 x 2 = 14 d = 5 x 2 = 10 32 e- p = 3 x 2 = 6 s=1 x 2=2 Atomic orbital - 1 Atomic orbital = 2 e

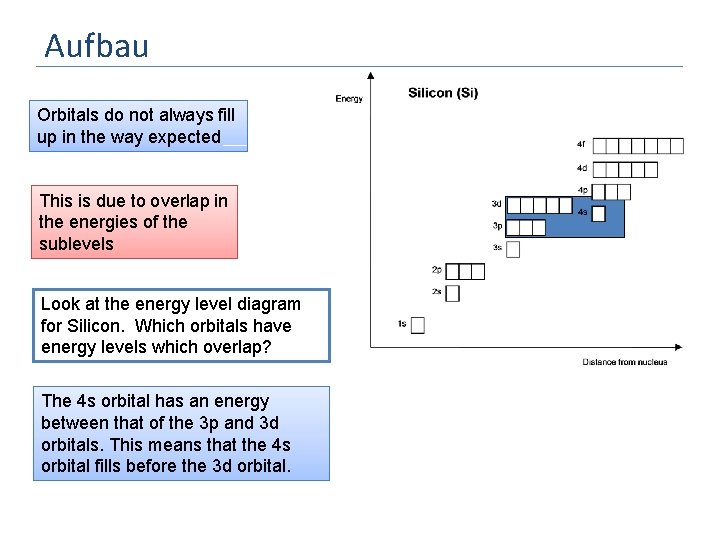
Aufbau Orbitals do not always fill up in the way expected This is due to overlap in the energies of the sublevels Look at the energy level diagram for Silicon. Which orbitals have energy levels which overlap? The 4 s orbital has an energy between that of the 3 p and 3 d orbitals. This means that the 4 s orbital fills before the 3 d orbital.

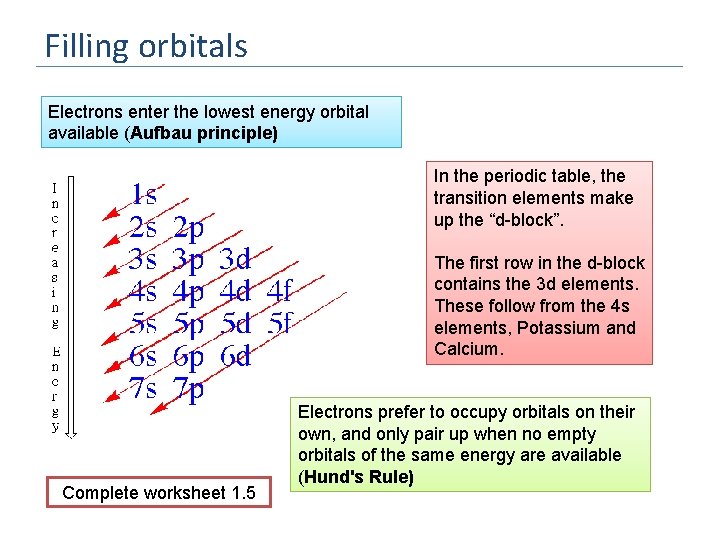
Filling orbitals Electrons enter the lowest energy orbital available (Aufbau principle) In the periodic table, the transition elements make up the “d-block”. The first row in the d-block contains the 3 d elements. These follow from the 4 s elements, Potassium and Calcium. Complete worksheet 1. 5 Electrons prefer to occupy orbitals on their own, and only pair up when no empty orbitals of the same energy are available (Hund's Rule)

Questions 1. Which orbital would the electrons fill first? The 2 s or 2 p orbital? 2. Can you have an electron in between two orbitals? 3. How many d orbitals are there in the d subshell? 4. How many electrons can the p orbital hold? 5. Why can two electrons occupy the same orbital? 1. The 2 s orbital would be filled before the 2 p orbital because orbitals that are lower in energy are filled first and the 2 s orbital is lower in energy than the 2 p orbital. 2. You cannot have an electron in between two orbitals. The electron will either be in one orbital or the next. 3. There are 5 d orbitals in the d subshell. 4. A p orbital can hold 6 electrons. 5. Two electrons can occupy the same orbital because they each have a different spin. There cannot be two electrons that have the same exact orbital configuration and spin.

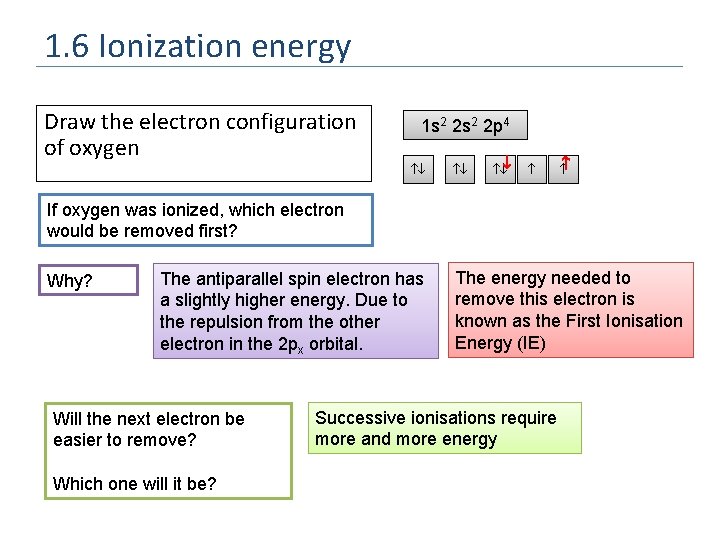
1. 6 Ionization energy Draw the electron configuration of oxygen 1 s 2 2 p 4 If oxygen was ionized, which electron would be removed first? Why? The antiparallel spin electron has a slightly higher energy. Due to the repulsion from the other electron in the 2 px orbital. Will the next electron be easier to remove? Which one will it be? The energy needed to remove this electron is known as the First Ionisation Energy (IE) Successive ionisations require more and more energy

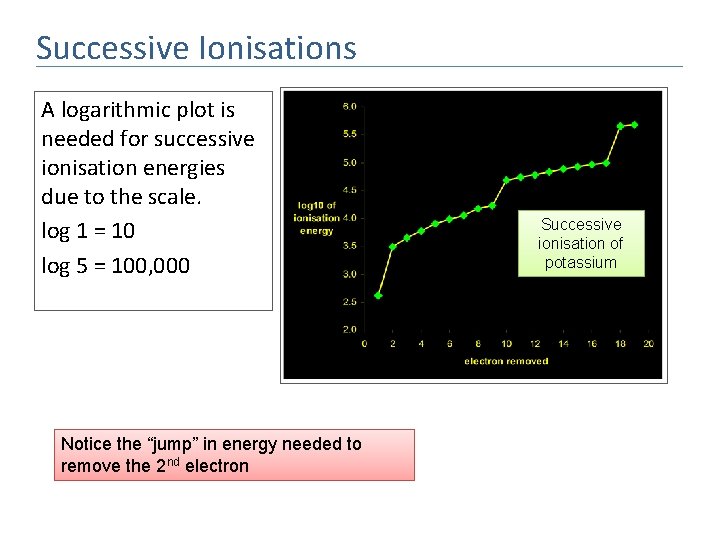
Successive Ionisations A logarithmic plot is needed for successive ionisation energies due to the scale. log 1 = 10 log 5 = 100, 000 Notice the “jump” in energy needed to remove the 2 nd electron Successive ionisation of potassium

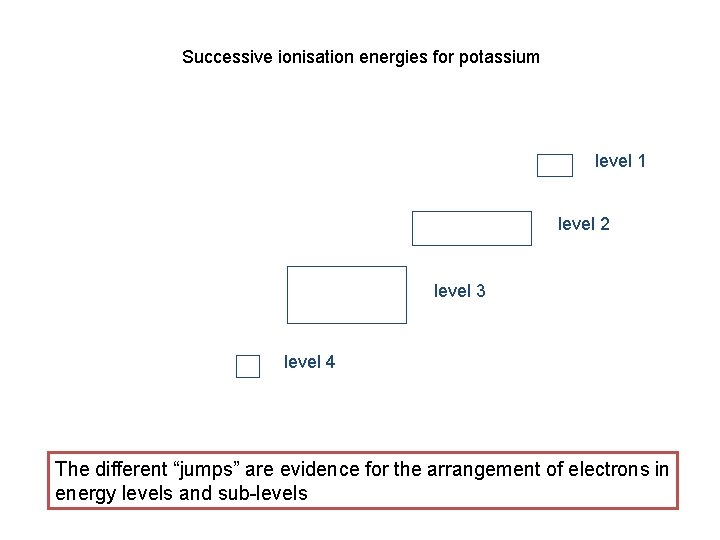
Successive ionisation energies for potassium level 1 level 2 level 3 level 4 The different “jumps” are evidence for the arrangement of electrons in energy levels and sub-levels

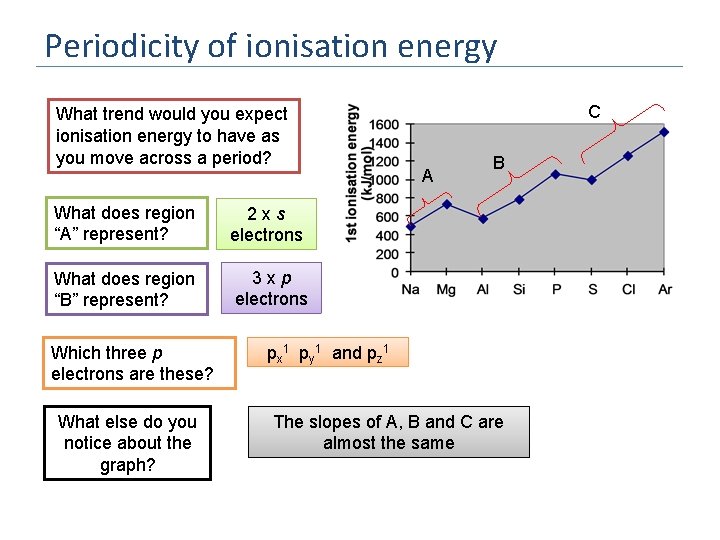
Periodicity of ionisation energy What trend would you expect ionisation energy to have as you move across a period? What does region “A” represent? 2 xs electrons What does region “B” represent? 3 xp electrons Which three p electrons are these? What else do you notice about the graph? C A B px 1 py 1 and pz 1 The slopes of A, B and C are almost the same

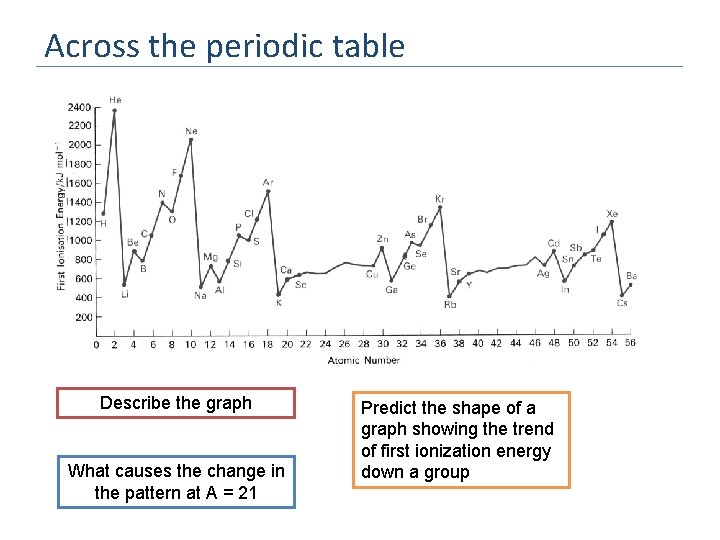
Across the periodic table Describe the graph What causes the change in the pattern at A = 21 Predict the shape of a graph showing the trend of first ionization energy down a group

Trends of first ionization energy in groups Group 2 Describe the graph The initial decrease is steep, but then the graph flattens out Explain why the first ionisation energy decreases as you move down a group

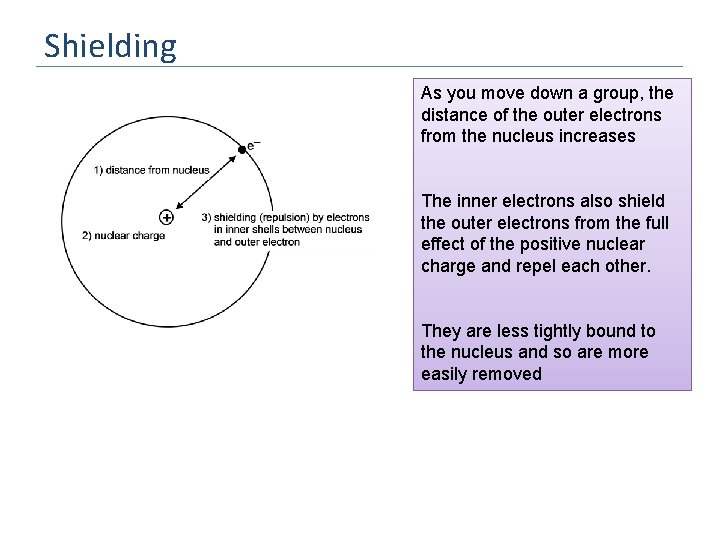
Shielding As you move down a group, the distance of the outer electrons from the nucleus increases The inner electrons also shield the outer electrons from the full effect of the positive nuclear charge and repel each other. They are less tightly bound to the nucleus and so are more easily removed

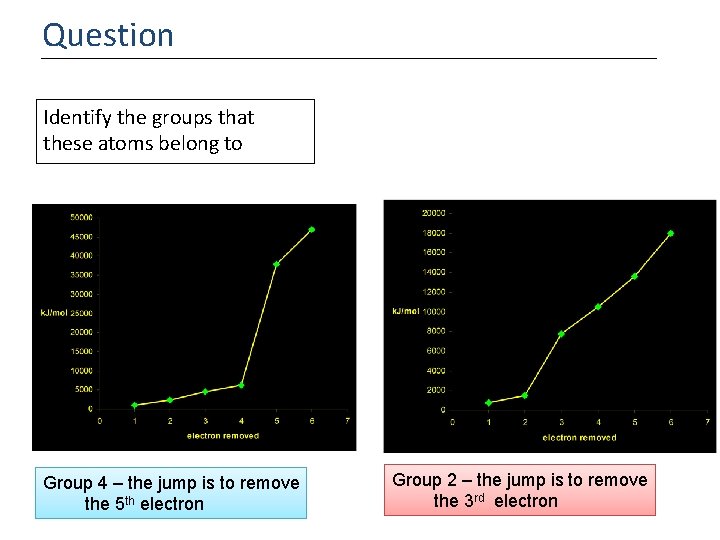
Question Identify the groups that these atoms belong to Group 4 – the jump is to remove the 5 th electron Group 2 – the jump is to remove the 3 rd electron

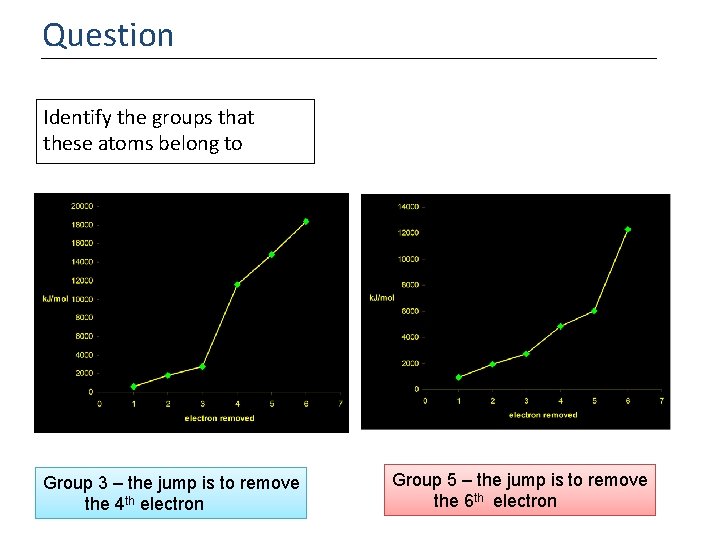
Question Identify the groups that these atoms belong to Group 3 – the jump is to remove the 4 th electron Group 5 – the jump is to remove the 6 th electron

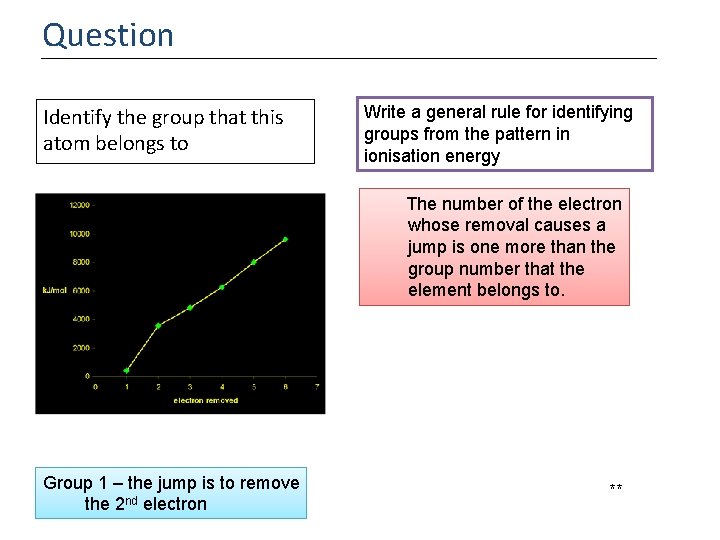
Question Identify the group that this atom belongs to Write a general rule for identifying groups from the pattern in ionisation energy The number of the electron whose removal causes a jump is one more than the group number that the element belongs to. Group 1 – the jump is to remove the 2 nd electron **