08 09 Chemistry Report Determine the calcium carbonate

08 -09 Chemistry Report Determine the calcium carbonate content of a sample of an egg shell Members : Ho chi nam Tong wing kwan Chan siu sing Chan sing yeung Lee cheung fat

Introduction • The pesticide DDT has caused significant damage to the environment and its wildlife. • Birds are especially affected because the DDT weakens the shells of their eggs which would break before hatching. • This caused certain bird species to become endangered (i. e. the American bald eagle). • One method of monitoring the strength of the egg is by determining the percentage of calcium carbonate in eggshell

Principle • During this experiment, the percentage of Ca. CO 3 in an eggshell is determined by reacting the eggshell with hydrochloric acid. • 2 HCl(aq)+Ca. CO 3(s)→Ca 2+(aq)+CO 2(g)+H 2 O(l)+2 Cl-(aq)

Back titration • This reaction cannot be used directly to titrate the Ca. CO 3 because it is dissolved in water. Thus , we must use back titration. • Back titration is an analytical chemistry technique that allows the user to find the concentration of a reactant of unknown concentration by reacting it with an excess volume of another reactant of known concentration.

• The resulting mixture is then titrated back, taking into account the molarity of the excess that was added. • Through to find out the molarity of resulting mixture, we can calculate the abundant of an unknown sample

• In this experiment, excess of standard hydrochloric acid is added to dissolve the eggshell. • Ethyl alcohol is added to eggshell as a wetting agent to help the hydrochloric acid dissolve the Ca. CO 3. • Wetting agents are chemicals that increase the spreading and penetrating properties of a liquid by lowering its surface tension – that is, the tendency of its molecules to adhere to each other.

• The remaining acid is titrated with Na. OH solution. • determine the amount of acid that did not react with the eggshell HCl(aq)+Na. OH(aq)→H 2 O(l)+Na+(aq)+Cl-(aq) • Through to find out the amount of acid , we can calculate the abundant of Ca. CO 3.

Experimental apparatus • • • • Wash bottle x 1 Conical flask x 4 Mortar and Pestle x 1 Filter funnel x 1 Electronic balance x 1 50. 00 ml Burette x 1 White tile x 1 25. 00 ml pipette 250. 00 ml beaker x 2 Oven x 1 Bunsen burner x 2 Tripod x 2 250. 00 ml volumetric flask x 2

Experimental Chemical 1. 2. 3. 4. 5. 6. 7. Phenolphthalein 0. 101188 M Na. OH 0. 2175 M HCl Enthanol Distilled water Paper towel Egg shell x 2 (import form USA and Malaysia respectively)

Procedure Preparing fine egg shell powder: 1. Eggs were put into boiling water for 5 minutes. 2. Eggshells of 2 eggs were washed with distilled water. 3. Eggshells of 2 eggs were obtained by break the eggs shell.

1. The inside membrane was removed. 2. The eggshell of 2 eggs was put into oven for 15 minutes. 3. The eggshell powder of 2 eggs were ground by using mortar and pestle.

Preparation 1. 2. 3. 4. 0. 20 g of dried eggshells powder was weighed into each of 4 conical flask accurately. 25. 00 cm 3 of 0. 2175 M HCl solution was pipetted into each flask. 5. 00 cm 3 of ethanol was added to each flask. The flasks were swirled to wet all of the solid. Any excess HCl should be disposed in the sink by diluting with water.

5. The solutions in the flasks were heated until they began to boil and allow to cool. The walls of the flasks were rinsed with distilled water from the wash bottle. 6. 3 drops of phenolphthalein was added to each flask.

1. 7. A funnel was used to partly fill a clean burette with Na. OH solution to rinse it. 2. 8. The burette was filled with Na. OH solution by using a funnel, and some solution were run out to remove all bubbles from the tip. 3. 9. The initial volume was recorded.

10. Titrate the first sample to get a reference. 11. Titrate other three samples and record the results. 12. Calculate the percentage of calcium carbonate in each sample and the mean value. 13. Calculate the average deviation from the mean.
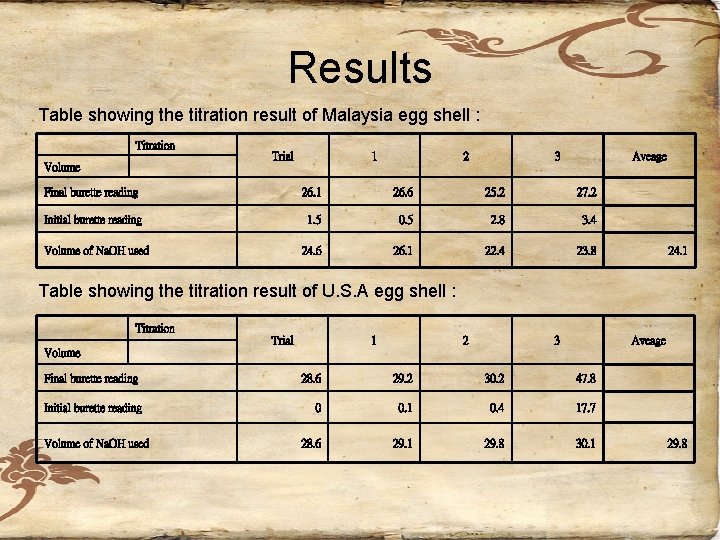
Results Table showing the titration result of Malaysia egg shell : Titration Volume Trial 1 2 3 Aveage Final burette reading 26. 1 26. 6 25. 2 27. 2 Initial burette reading 1. 5 0. 5 2. 8 3. 4 Volume of Na. OH used 24. 6 26. 1 22. 4 23. 8 24. 1 Table showing the titration result of U. S. A egg shell : Titration Volume Trial Final burette reading Initial burette reading Volume of Na. OH used 1 2 3 Aveage 28. 6 29. 2 30. 2 47. 8 0 0. 1 0. 4 17. 7 28. 6 29. 1 29. 8 30. 1 29. 8

Calculations the initial moles of HCl = 0. 2175 M x 25 cm 3/1000 cm 3 = 5. 4375 x 10 -3 mol HCl (aq) + Na. OH (aq)→Na. CL (aq) + H 2 O (l) 1 mole of HCl (aq) react with 1 mole of Na. OH (aq) the moles of HCl react with Na. OH = the moles of Na. OH used =0. 101188 x 24. 1 cm 3/1000 cm 3 =2. 4386308 x 10 -3 mol

The moles of HCL react with Ca. CO 3 = the Initial moles of HCl – the moles of HCl reacted with Na. OH = 5. 1875 x 10 -3 mol – 2. 4386308 x 10 -3 mol =2. 9988692 x 10 -3 mol

Ca. CO 3 (s) + 2 HCl (aq) →Ca. Cl (aq) + H 2 O (l) + CO 2 (g) 1 mole of Ca. CO 3 react with 2 moles HCl The moles of Ca. CO 3 = 2. 9988692 x 103 mol / 2 = 1. 4994346 x 10 -3 mol Percentage of Ca. CO 3 in eggshell = 1. 4994346 x 10 -3 mol x(40. 1+12+16 x 3)gmol/0. 2 g x 100 = 75. 05%

• the initial moles of HCl = 0. 2175 M x 25 cm 3/1000 cm 3 = 5. 4375 x 10 -3 mol HCl (aq) + Na. OH (aq)→Na. CL (aq) + H 2 O (l) • 1 mole of HCl (aq) react with 1 mole of Na. OH (aq) • the moles of HCl react with Na. OH = the moles of Na. OH used =0. 101188 x 29. 8 cm 3/1000 cm 3 =3. 015164 x 10 -3 mol

• The moles of HCL react with Ca. CO 3 = the Initial moles of HCl – the moles of HCl reacted with Na. OH = 5. 4375 x 10 -3 mol – 3. 015164 x 10 -3 mol =2. 4220976 x 10 -3 mol

• Ca. CO 3 (s) + 2 HCl (aq) →Ca. Cl (aq) + H 2 O (l) + CO 2 (g) • 1 mole of Ca. CO 3 react with 2 moles HCl • The moles of Ca. CO 3 = 2. 4220976 x 10 -3 mol / 2 = 1. 2110488 x 10 -3 mol • Percentage of Ca. CO 3 in eggshell =1. 2110488 x 10 -3 mol x(40. 1+12+16 x 3)g-mol/0. 2 g x 100 =60. 61%

Observations • Bubbles appeared when dissolving egg shell powder by HCl(aq). • Eggshell powder could not completely dissolve in HCl. • The sodium hydroxide solution was colourless. • When the sodium hydroxide solution was added to the mixture of eggshell and hydrochloride, the mixture turned pink.

Remarks • Ethanol must be added before the boiling of mixture of eggshell and hydrochloride. • The ethanol should be stored in a closed vessel and was pipette only when it was used. • When the mixture heated to 80 o. C, the titration was started immediately. • Shark flask continuously during the titration. • Make sure that the eggshell powder was dry. • The conical flask should be heated by air bath because the mixture contain ethanol which is flammable.

Source of error • Variation in visual judgement at the end point. • Eggshell powder could not dissolve completely in HCl. • The inside membrane was not completely removed. • The mixture was not heated enough. • Although after through rinsing, not all the eggshell powder is transferred to the volumetric flask.

Precautions • The hydrochloride acid and Sodium hydroxide solution are corrosiveness. • During the heating, the safety goggles must be weared. • Label the containers of each solution because they are colorless solution. • We should wash our hands after the experiment.

Conclusion Though the experiment, the egg of Malaysia was found that the percentage of Ca. CO 3 was 75. 05%. The egg of U. S. A. was found that the percentage of Ca. CO 3 was 60. 61%. The egg of Malaysia contain more Ca. CO 3 than the egg of U. S. A. It reflect that the pollution of DDT is more serious in U. S. A
- Slides: 27